Showing Spotlights 265 - 272 of 544 in category All (newest first):
 Carbon nanomaterials such as nanotubes or graphene not only are widely researched for their potential uses in industrial applications, they also are of great interest to biomedical engineers working on nanotechnology applications. These researchers found that incorporating carbon-based nanomaterials is effective not only as injectable nanoscale devices but also as components to enhance the function of existing biomaterials significantly. A recent article highlights different types of carbon-based nanomaterials currently used in biomedical applications.
Carbon nanomaterials such as nanotubes or graphene not only are widely researched for their potential uses in industrial applications, they also are of great interest to biomedical engineers working on nanotechnology applications. These researchers found that incorporating carbon-based nanomaterials is effective not only as injectable nanoscale devices but also as components to enhance the function of existing biomaterials significantly. A recent article highlights different types of carbon-based nanomaterials currently used in biomedical applications.
Apr 25th, 2013
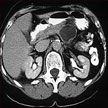 Computed tomography (CT) scans are an imaging method that use x-rays to create pictures of cross-sections of the body. Many CT scans involve intravenously injected iodinated contrast agents - but they have drawbacks such as fast clearance and side effects. Due to these limitations, nano-sized iodinated CT contrast agents have been developed that can increase the circulation time and decrease the adverse effects. In addition to iodine, nanoparticles based on heavy atoms such as gold, lanthanides, and tantalum are used as more efficient CT contrast agents.
Computed tomography (CT) scans are an imaging method that use x-rays to create pictures of cross-sections of the body. Many CT scans involve intravenously injected iodinated contrast agents - but they have drawbacks such as fast clearance and side effects. Due to these limitations, nano-sized iodinated CT contrast agents have been developed that can increase the circulation time and decrease the adverse effects. In addition to iodine, nanoparticles based on heavy atoms such as gold, lanthanides, and tantalum are used as more efficient CT contrast agents.
Apr 10th, 2013
 A comprehensive understanding of the brain remains an elusive, distant frontier. To arrive at a general theory of brain function would be an historic event, comparable to inferring quantum theory from huge sets of complex spectra and inferring evolutionary theory from vast biological field work. The proposed Brain Activity Map is a project that will tap the hive mind of experts to make headway in the understanding of the field. Engineers and nanotechnologists will be needed to help build ever smaller devices for measuring the activity of individual neurons and, later, to control how those neurons function. Recent developments in nanoscale analysis tools and in the design and synthesis of nanomaterials have generated optical, electrical, and chemical methods that can readily be adapted for use in neuroscience.
A comprehensive understanding of the brain remains an elusive, distant frontier. To arrive at a general theory of brain function would be an historic event, comparable to inferring quantum theory from huge sets of complex spectra and inferring evolutionary theory from vast biological field work. The proposed Brain Activity Map is a project that will tap the hive mind of experts to make headway in the understanding of the field. Engineers and nanotechnologists will be needed to help build ever smaller devices for measuring the activity of individual neurons and, later, to control how those neurons function. Recent developments in nanoscale analysis tools and in the design and synthesis of nanomaterials have generated optical, electrical, and chemical methods that can readily be adapted for use in neuroscience.
Mar 27th, 2013
 Currently developed nanotechnology-based drug delivery systems, either 'passively targeted' or 'actively targeted', do not significantly improve the delivery of drugs to target tumors. Scientists in China now propose a new-generation nanocarrier that integrates various desired functions into a single nanosystem, which can harmonize with the complex physiological environment and display different properties sequentially, thus resulting in an excellent targeting effect and satisfactory biodistribution of drugs.
Currently developed nanotechnology-based drug delivery systems, either 'passively targeted' or 'actively targeted', do not significantly improve the delivery of drugs to target tumors. Scientists in China now propose a new-generation nanocarrier that integrates various desired functions into a single nanosystem, which can harmonize with the complex physiological environment and display different properties sequentially, thus resulting in an excellent targeting effect and satisfactory biodistribution of drugs.
Mar 5th, 2013
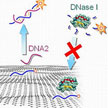
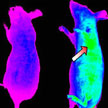
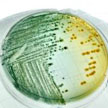 A bacterium which causes disease reacts to the antibiotics used as treatment by becoming resistant to them, sooner or later. This natural process of adaptation, antimicrobial resistance, means that the effective lifespan of antibiotics is limited. Unnecessary use and inappropriate use of antibiotics favors the emergence and spread of resistant bacteria. New research uses a graphene-based photothermal agent to trap and kill bacteria.
A bacterium which causes disease reacts to the antibiotics used as treatment by becoming resistant to them, sooner or later. This natural process of adaptation, antimicrobial resistance, means that the effective lifespan of antibiotics is limited. Unnecessary use and inappropriate use of antibiotics favors the emergence and spread of resistant bacteria. New research uses a graphene-based photothermal agent to trap and kill bacteria.
Feb 19th, 2013
 Carbon nanotubes, like the nervous cells of our brain, are excellent electrical signal conductors and can form intimate mechanical contacts with cellular membranes, thereby establishing a functional link to neuronal structures. There is a growing body of research on using nanomaterials in neural engineering. Carbon nanotube (CNT) synapse circuits are a first step in this direction. In new work, researchers at the University of California, Los Angeles, have developed a CNT synapse with the elementary dynamic logic, learning, and memory functions of a biological synapse.
Carbon nanotubes, like the nervous cells of our brain, are excellent electrical signal conductors and can form intimate mechanical contacts with cellular membranes, thereby establishing a functional link to neuronal structures. There is a growing body of research on using nanomaterials in neural engineering. Carbon nanotube (CNT) synapse circuits are a first step in this direction. In new work, researchers at the University of California, Los Angeles, have developed a CNT synapse with the elementary dynamic logic, learning, and memory functions of a biological synapse.
Feb 12th, 2013
 The majority of current commercial applications of nanotechnology to medicine is geared towards drug delivery to enable new modes of action, as well as better targeting and bioavailability of existing medicinal substances. While most reviews on nanomedicine tend to focus on specific sectors or take a very forward-looking stance and fail to provide a complete perspective on the current landscape, a new review article provides a more comprehensive and contemporary inventory of nanomedicine products.
The majority of current commercial applications of nanotechnology to medicine is geared towards drug delivery to enable new modes of action, as well as better targeting and bioavailability of existing medicinal substances. While most reviews on nanomedicine tend to focus on specific sectors or take a very forward-looking stance and fail to provide a complete perspective on the current landscape, a new review article provides a more comprehensive and contemporary inventory of nanomedicine products.
Jan 17th, 2013
 Metastasis - the process of cancer spreading from its original site to distant tissues - is caused by marauding tumor cells that break off from the primary tumor site and ride in the bloodstream to set up colonies in other parts of the body. Detecting and analyzing these circulating tumor cells (CTCs) can provide critical information for managing the spread of cancer and monitoring the effectiveness of therapies. In new work, researchers have created a Velcro-like platform which can not only capture CTCs with very high efficiency, but also release them in order to biochemically analyze them further.
Metastasis - the process of cancer spreading from its original site to distant tissues - is caused by marauding tumor cells that break off from the primary tumor site and ride in the bloodstream to set up colonies in other parts of the body. Detecting and analyzing these circulating tumor cells (CTCs) can provide critical information for managing the spread of cancer and monitoring the effectiveness of therapies. In new work, researchers have created a Velcro-like platform which can not only capture CTCs with very high efficiency, but also release them in order to biochemically analyze them further.
Jan 8th, 2013
 Carbon nanomaterials such as nanotubes or graphene not only are widely researched for their potential uses in industrial applications, they also are of great interest to biomedical engineers working on nanotechnology applications. These researchers found that incorporating carbon-based nanomaterials is effective not only as injectable nanoscale devices but also as components to enhance the function of existing biomaterials significantly. A recent article highlights different types of carbon-based nanomaterials currently used in biomedical applications.
Carbon nanomaterials such as nanotubes or graphene not only are widely researched for their potential uses in industrial applications, they also are of great interest to biomedical engineers working on nanotechnology applications. These researchers found that incorporating carbon-based nanomaterials is effective not only as injectable nanoscale devices but also as components to enhance the function of existing biomaterials significantly. A recent article highlights different types of carbon-based nanomaterials currently used in biomedical applications.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





