| Posted: Jun 19, 2007 | |
Nanotechnology's "magic bullet" |
|
| (Nanowerk Spotlight) The ideal drug carrier may be something out of science fiction. In principle, it is injected into the body and transports itself to the correct target, such as a tumor, and delivers the required dose at this target. This idealized concept was first proposed by Paul Ehrlich at the beginning of the 20th century and was nicknamed the “magic bullet” concept. With the advent of nanotechnology and nanomedicine this dream is rapidly becoming a reality. Nanotechnology has already been applied to drug delivery and cosmetics through the use of liposomal technology, and now nanoparticles and nanotubes present an exciting and more promising alternative. | |
| Typically nanoparticles have been used for drug delivery and it has been only recently that nanotubes have gained attention as potential drug delivery vehicles. Due to their precise targeting nature and the protective environment they provide, these nanoscale drug carriers may reduce the adverse side effects which result from the current methods of delivery of anticancer agents. Furthermore, nanocapsules may improve drug delivery methods in many areas other than cancer treatment, such as infections, metabolic diseases, autoimmune diseases, pain treatment and gene therapy. | |
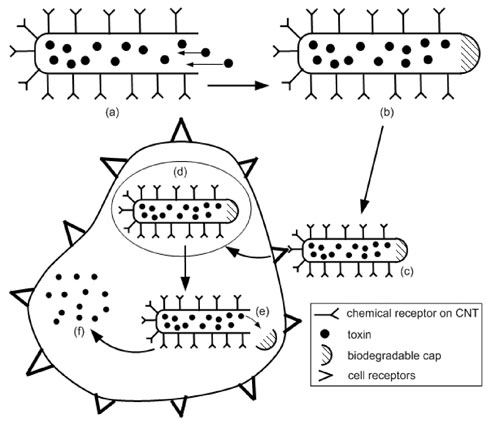
| Carbon nanotubes offer a number of advantages which suggest that they may provide an improved result. They have a larger inner volume which allows more drug molecules to be encapsulated, and this volume is more easily accessible because the end caps can be easily removed, and they have distinct inner and outer surfaces for functionalization. Both nanoparticles and nanotubes have been shown to be readily taken up by cells, and nanotubes have been found to enter cell nuclei suggesting that they may be useful in gene therapy. With reference to the figure below, the general process of drug delivery using a carbon nanotube proceeds as follows: | |
| (a) the carbon nanotube, in this case in the form of a nano test tube ("Corking Nano Test Tubes by Chemical Self-Assembly"), has its surface functionalized with some chemical receptor such in folate-targeting ("Folate receptor-mediated drug targeting: From therapeutics to diagnostics") and the drug molecules are encapsulated through the open end, | |
| (b) the open end is capped with some chemically removable cap, such as biodegradable. Degradation may be sensitive to the environment such as pH, or may be initiated by an external source, | |
| (c) the nanocapsule is then introduced into the body by intravenous injection or orally, whereby it locates to the target site through the use of the chemical receptors. For example often cancer tumors over express folate receptors and thus the nanocapsule selectively binds to these cells, | |
| (d) the cell ingests the nanocapsule, for example by receptor-mediated endocytosis, | |
| (e) the chemically removable cap is triggered and either falls off or biodegrades, | |
| (f) and finally the nanotube spills its contents into the cell and thus the drug is delivered. | |
 |
|
| Outline of the proposed drug delivery process. (a) Nanotube surface is functionalized with a chemical receptor and the drug molecules are encapsulated (b) open end is capped (c) nanoscale carrier is ingested and locates to target site due to functionalized surface (d) cell internalizes the carrier for example by receptor mediated endocytosis (e) cap is removed or biodegrades inside cell (f) drug molecules are released. (Image: Tamsyn Hilder, University of Wollongong) | |
| Ideally we want to engineer a nanotube carrier so that when outside the body it is energetically favorable for the drug molecule to be encapsulated, and once inside the desired cell it is energetically favorable to be ejected, and thus depositing the desired drug at the target site. | |
| Motivated by our recent research into the suction of fullerenes into carbon nanotubes, in our paper, "Modelling the encapsulation of the anticancer drug cisplatin into carbon nanotubes", we investigate the encapsulation behavior of a particular anticancer drug molecule entering a carbon nanotube using applied mathematical modeling techniques and elementary mechanics. This paper determines the minimum radius of nanotube that would accept cisplatin into its interior and similarly determines the tube radius which will provide the maximum drug uptake. It is found that the nanotube radius must be at least 4.785 Å (slightly smaller than a (9, 5) tube) to accept cisplatin into the interior, and that a radius of approximately 5.3 Å (approximately equivalent to a (11, 4) tube) provides the maximum suction energy (or uptake). | |
| The paper draws on a mathematical formulation proposed in an earlier paper, "Continuous versus discrete for interacting carbon nanostructures", by the authors, Tamsyn Hilder and Prof. James Hill, who are part of the Nanomechanics Group at the University of Wollongong. This group attempts to model nanotechnology and nanomedical phenomena using the techniques of classical applied mathematical modeling based on mathematical analysis and elementary mechanical principles. | |
| This technique, which can be extended to any number of drug molecules, may be used to provide overall guidelines for both medical scientists and other researchers to assist in experimental and molecular dynamics studies. Our study presents for the first time a calculation of this nature, and although it represents a first approximation, it constitutes a necessary preliminary calculation. It is hoped that in future work the expulsion behavior of the drug molecule once the nanocapsule is inside the cell may be investigated. However, the expulsion occurs in an extremely complicated environment and as such presents challenges with respect to how best to represent this environment mathematically. | |
| By Tamsyn Hilder, Nanomechanics Group at the University of Wollongong, Australia. Copyright Nanowerk LLC | |
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
