Human Brain Project specifies its research goals
 The European Human Brain Project (HBP) presented its new strategic objectives at its annual meeting.
The European Human Brain Project (HBP) presented its new strategic objectives at its annual meeting.
Nov 14th, 2016
Read more
 Subscribe to our Biotechnology News feed
Subscribe to our Biotechnology News feed
 The European Human Brain Project (HBP) presented its new strategic objectives at its annual meeting.
The European Human Brain Project (HBP) presented its new strategic objectives at its annual meeting.
Nov 14th, 2016
Read more Encapsulating molecular components in artificial membranes offers more flexibility in designing circuits.
Encapsulating molecular components in artificial membranes offers more flexibility in designing circuits.
Nov 14th, 2016
Read more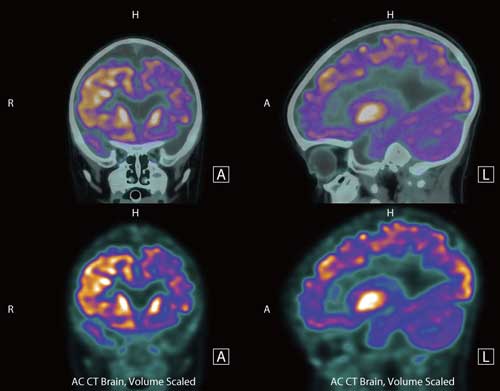 PET imaging of new neurons in the brain promises to advance our understanding and treatment of depression.
PET imaging of new neurons in the brain promises to advance our understanding and treatment of depression.
Nov 11th, 2016
Read more Researchers develop a combination of software and hardware for adaptive live imaging of large living organisms.
Researchers develop a combination of software and hardware for adaptive live imaging of large living organisms.
Nov 10th, 2016
Read more Scientists will study potential uses of the carbon contained in biomass and of the carbon dioxide arising as a by-product in industrial production. The project is aimed at developing a novel process chain for the synthesis of the basic chemical substance methanol.
Scientists will study potential uses of the carbon contained in biomass and of the carbon dioxide arising as a by-product in industrial production. The project is aimed at developing a novel process chain for the synthesis of the basic chemical substance methanol.
Nov 10th, 2016
Read more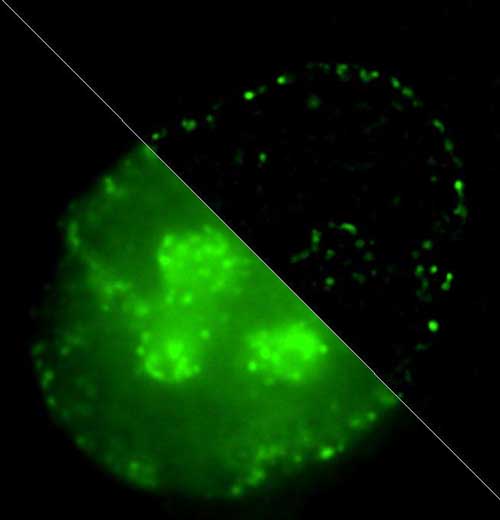 Scientists have conducted a research on DNA packaging in the cell nucleus and its alteration in the process of DNA replication. DNA packaging is supposed to have strong influence on genes' activity. It's also one of the mechanisms of epigenetic control of gene expression.
Scientists have conducted a research on DNA packaging in the cell nucleus and its alteration in the process of DNA replication. DNA packaging is supposed to have strong influence on genes' activity. It's also one of the mechanisms of epigenetic control of gene expression.
Nov 8th, 2016
Read more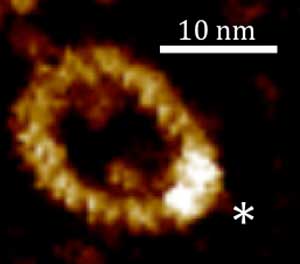 Researchers have studied and visualised the formation of quadruplexes in looped DNA that may be a better representation of DNA in the living cell than in the short and linear pieces of DNA that are more commonly used for such investigations.
Researchers have studied and visualised the formation of quadruplexes in looped DNA that may be a better representation of DNA in the living cell than in the short and linear pieces of DNA that are more commonly used for such investigations.
Nov 7th, 2016
Read moreUsing nucleophilic probes, researchers discover a novel electrophilic protein modification.
Nov 1st, 2016
Read more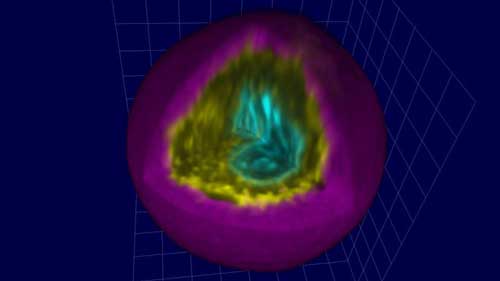 A new method for encapsulating single cells within tunable microgels could boost efficacy of cell-based therapies and tissue engineering.
A new method for encapsulating single cells within tunable microgels could boost efficacy of cell-based therapies and tissue engineering.
Oct 31st, 2016
Read more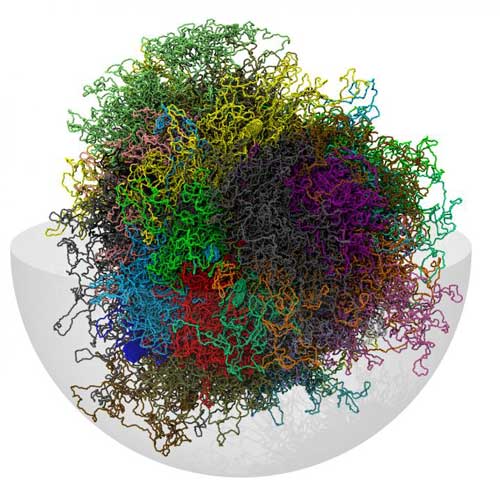 A new method provides a three-dimensional identikit of chromosomes.
A new method provides a three-dimensional identikit of chromosomes.
Oct 27th, 2016
Read more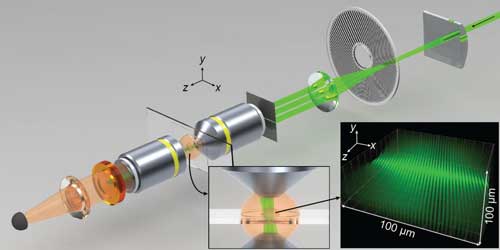 Researchers have designed and built a fluorescence-detection microscope that combines 3-D and high-resolution image processing.
Researchers have designed and built a fluorescence-detection microscope that combines 3-D and high-resolution image processing.
Oct 25th, 2016
Read more Scientists develop a novel method to benchmark and improve the performance of protein measurement techniques.
Scientists develop a novel method to benchmark and improve the performance of protein measurement techniques.
Oct 25th, 2016
Read moreA new device could speed up the process of genetically modifying blood stem cells to treat diseases and expand access to gene therapy worldwide.
Oct 20th, 2016
Read moreTransforming data about the structure of proteins into melodies gives scientists a completely new way of analyzing the molecules that could reveal new insights into how they work - by listening to them. A new study shows how musical sounds can help scientists analyze data using their ears instead of their eyes.
Oct 20th, 2016
Read more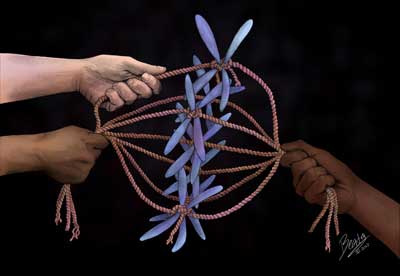 Biologists who study the mechanics of cell division have for years disagreed about how much force is at work when the cell's molecular engines are lining chromosomes up in the cell, preparing to winch copies to opposite poles across a bridge-like structure called the kinetochore to form two new cells. The question is fundamental to understanding how cells divide.
Biologists who study the mechanics of cell division have for years disagreed about how much force is at work when the cell's molecular engines are lining chromosomes up in the cell, preparing to winch copies to opposite poles across a bridge-like structure called the kinetochore to form two new cells. The question is fundamental to understanding how cells divide.
Oct 20th, 2016
Read more Scientists have for the first time precisely described the chemical reactions responsible for the stability of hydrogen generation in an aerobic environment by algal enzymes.
Scientists have for the first time precisely described the chemical reactions responsible for the stability of hydrogen generation in an aerobic environment by algal enzymes.
Oct 19th, 2016
Read more Up until now it had only been possible to create monolayer membranes or vesicles from specially synthesized macromolecules. Vesicles made from a bilayer of naturally occurring proteins that can also be tailored for use in an aqueous or an oil-based environment are something quite new.
Up until now it had only been possible to create monolayer membranes or vesicles from specially synthesized macromolecules. Vesicles made from a bilayer of naturally occurring proteins that can also be tailored for use in an aqueous or an oil-based environment are something quite new.
Oct 17th, 2016
Read more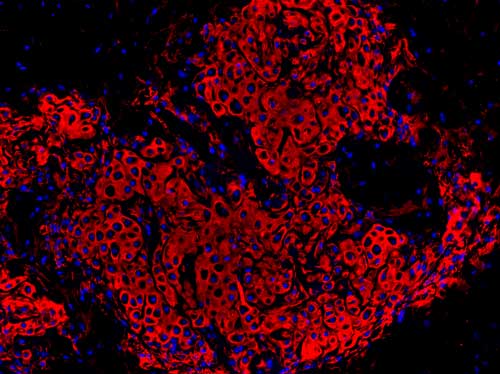 Rsearchers have identified stem cells that can make new cartilage and repair damaged joints.
Rsearchers have identified stem cells that can make new cartilage and repair damaged joints.
Oct 10th, 2016
Read more