Showing Spotlights 73 - 80 of 152 in category All (newest first):
 Polyurethane (PU) foam is an extremely versatile material that commonly is used in bedding, upholstery and building insulation. However, PU foam is very flammable, often resulting in dripping of melted material that enhances flame spread through the formation of a pool fire under the burning object. Brominated flame retardant compounds (e.g. pentabromodiphenyl ether) have been used to reduce foam flammability but there is growing evidence that these chemicals are toxic to the environment and living organisms. Replacing brominated flame retardants in polymer formulations with safer and more environmentally-friendly alternatives has also sparked the interest of nanoscientists. One recent effort to create an environmentally-friendly flame retardant system involves the layer-by-layer assembly of thin films using materials obtained from completely renewable sources.
Polyurethane (PU) foam is an extremely versatile material that commonly is used in bedding, upholstery and building insulation. However, PU foam is very flammable, often resulting in dripping of melted material that enhances flame spread through the formation of a pool fire under the burning object. Brominated flame retardant compounds (e.g. pentabromodiphenyl ether) have been used to reduce foam flammability but there is growing evidence that these chemicals are toxic to the environment and living organisms. Replacing brominated flame retardants in polymer formulations with safer and more environmentally-friendly alternatives has also sparked the interest of nanoscientists. One recent effort to create an environmentally-friendly flame retardant system involves the layer-by-layer assembly of thin films using materials obtained from completely renewable sources.
Mar 9th, 2012
 Technological advances have made desalination and demineralization of seawater feasible, albeit expensive, solutions for increasing the world's supply of freshwater. Among various desalination technologies, reverse osmosis membranes have been widely used for water reclamation. However, external energy required and high operational pressure used (above 75 bar for reverse osmosis desalination and 25 bar for reverse osmosis water recovery from wastewater) make reverse osmosis membrane water reclamation processes energy intensive - not exactly an advantage given the rising cost of energy and the negative climate impact of fossil fuels. Researchers have now demonstrate the novel concept of a "desalination battery", which operates by performing cycles in reverse on our previously reported mixing entropy battery.
Technological advances have made desalination and demineralization of seawater feasible, albeit expensive, solutions for increasing the world's supply of freshwater. Among various desalination technologies, reverse osmosis membranes have been widely used for water reclamation. However, external energy required and high operational pressure used (above 75 bar for reverse osmosis desalination and 25 bar for reverse osmosis water recovery from wastewater) make reverse osmosis membrane water reclamation processes energy intensive - not exactly an advantage given the rising cost of energy and the negative climate impact of fossil fuels. Researchers have now demonstrate the novel concept of a "desalination battery", which operates by performing cycles in reverse on our previously reported mixing entropy battery.
Feb 7th, 2012
 Many batteries still contain heavy metals such as mercury, lead, cadmium, and nickel, which can contaminate the environment and pose a potential threat to human health when batteries are improperly disposed of. Not only do the billions upon billions of batteries in landfills pose an environmental problem, they also are a complete waste of a potential and cheap raw material. Unfortunately, current recycling methods for many battery types, especially the small consumer type ones, don't make sense from an economical point of view since the recycling costs exceed the recoverable metals value. Researchers in India have carried out research to address the recycling of consumer-type batteries. They report the recovery of pure zinc oxide nanoparticles from spent Zn-Mn dry alkaline batteries.
Many batteries still contain heavy metals such as mercury, lead, cadmium, and nickel, which can contaminate the environment and pose a potential threat to human health when batteries are improperly disposed of. Not only do the billions upon billions of batteries in landfills pose an environmental problem, they also are a complete waste of a potential and cheap raw material. Unfortunately, current recycling methods for many battery types, especially the small consumer type ones, don't make sense from an economical point of view since the recycling costs exceed the recoverable metals value. Researchers in India have carried out research to address the recycling of consumer-type batteries. They report the recovery of pure zinc oxide nanoparticles from spent Zn-Mn dry alkaline batteries.
Dec 1st, 2011
 Breath analysis has been recognized as an increasingly accurate diagnostic method to link specific gaseous components in human breath to medical conditions and exposure to chemical compounds. Sampling breath is also much less invasive than testing blood, can be done very quickly, and creates as good as no biohazard waste. A recent review article in Environmental Science and Technology focuses on breath analysis as a tool for assessing environmental exposure and provides a good overview of the current state of diagnostic tools, leading studies in this field, and emerging technologies for hand-held breath analyzers. After describing the basics of breath analysis as a diagnostic tool, the authors discuss emerging chemical sensor technology ('electronic noses'), in particular two nanotechnology-based approaches, that are suitable to identify target analytes in breath.
Breath analysis has been recognized as an increasingly accurate diagnostic method to link specific gaseous components in human breath to medical conditions and exposure to chemical compounds. Sampling breath is also much less invasive than testing blood, can be done very quickly, and creates as good as no biohazard waste. A recent review article in Environmental Science and Technology focuses on breath analysis as a tool for assessing environmental exposure and provides a good overview of the current state of diagnostic tools, leading studies in this field, and emerging technologies for hand-held breath analyzers. After describing the basics of breath analysis as a diagnostic tool, the authors discuss emerging chemical sensor technology ('electronic noses'), in particular two nanotechnology-based approaches, that are suitable to identify target analytes in breath.
Oct 24th, 2011
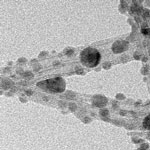 Back in 2008 we reported on nanotechnology solution for radioactive waste cleanup, specifically the use of titanate nanofibers as absorbents for the removal of radioactive ions from water. Now, the same group that developed these nanomaterials reports in a new study that the unique structural properties of titanate nanotubes and nanofibers make them superior materials for removal of radioactive cesium and iodine ions in water. Based on their earlier work, the researchers have now demonstrated a potentially cost-effective method to remediate cesium and iodine ions from contaminated water by using the unique chemistry of titanate nanotubes and nanofibers to chemisorb these ions.
Back in 2008 we reported on nanotechnology solution for radioactive waste cleanup, specifically the use of titanate nanofibers as absorbents for the removal of radioactive ions from water. Now, the same group that developed these nanomaterials reports in a new study that the unique structural properties of titanate nanotubes and nanofibers make them superior materials for removal of radioactive cesium and iodine ions in water. Based on their earlier work, the researchers have now demonstrated a potentially cost-effective method to remediate cesium and iodine ions from contaminated water by using the unique chemistry of titanate nanotubes and nanofibers to chemisorb these ions.
Oct 4th, 2011
 In the last couple of years, there has been particularly growing interest worldwide in exploring ways of finding suitable solutions to clean up oil spills and deal with industrial oily wastewater through use of nanomaterials. Key for the success of these materials is a high separation capacity, with resistance to oil fouling, and that are easily recyclable. Oil/water separation is an interfacial challenge, and novel materials designed to possess special wettability have different interaction and affinity for oil and water, thus can realize the separation. Until now, researches in this field all focus on materials with both hydrophobic and oleophilic properties. However, the oil-removing type of materials is easily fouled even blocked up by oils because of their intrinsic oleophilic property. A novel superhydrophilic and underwater superoleophobic hydrogel coated mesh can selectively separate water from oil/water mixtures effectively and without any extra power.
In the last couple of years, there has been particularly growing interest worldwide in exploring ways of finding suitable solutions to clean up oil spills and deal with industrial oily wastewater through use of nanomaterials. Key for the success of these materials is a high separation capacity, with resistance to oil fouling, and that are easily recyclable. Oil/water separation is an interfacial challenge, and novel materials designed to possess special wettability have different interaction and affinity for oil and water, thus can realize the separation. Until now, researches in this field all focus on materials with both hydrophobic and oleophilic properties. However, the oil-removing type of materials is easily fouled even blocked up by oils because of their intrinsic oleophilic property. A novel superhydrophilic and underwater superoleophobic hydrogel coated mesh can selectively separate water from oil/water mixtures effectively and without any extra power.
Sep 1st, 2011
 Notwithstanding all the buzz about renewable energy sources, the dirty facts are that coal accounts for 41% of electricity production worldwide. Since, realistically, coal will be a mainstay of electricity generation for many years to come, research into more environmentally friendly use of coal energy is picking up steam. One technology for more efficient power production centers around the solid oxide fuel cell (SOFC). Especially gasified carbon fuel cells offer great prospects for the most efficient utilization of a wide variety of carbonaceous solids fuels, including coal, biomass, and municipal solid waste. Researchers have now developed a self-cleaning technique that could allow solid oxide fuel cells to be powered directly by coal gas at operating temperatures as low as 750 degrees Celsius.
Notwithstanding all the buzz about renewable energy sources, the dirty facts are that coal accounts for 41% of electricity production worldwide. Since, realistically, coal will be a mainstay of electricity generation for many years to come, research into more environmentally friendly use of coal energy is picking up steam. One technology for more efficient power production centers around the solid oxide fuel cell (SOFC). Especially gasified carbon fuel cells offer great prospects for the most efficient utilization of a wide variety of carbonaceous solids fuels, including coal, biomass, and municipal solid waste. Researchers have now developed a self-cleaning technique that could allow solid oxide fuel cells to be powered directly by coal gas at operating temperatures as low as 750 degrees Celsius.
Jun 28th, 2011
 Water treatment is important for human consumption and environmental protection. Non-trivial purification of water involves removal of toxic ions, organic impurities, microbes and their by-products as well as scooping oil spills. The removal of organic contaminants from water is a major industrial concern. The challenging goal here is to detect, decompose and remove contaminants present usually in low concentrations. Towards this end, different types of sorbent materials have been developed to date, the most common being activated carbon. Though the use of activated carbon is still considered to be one of the best method, the disposal of adsorbed contaminants along with the adsorbent is a major concern. Researchers in India have recently come up with an innovative method for organic pollutant removal from waste water.
Water treatment is important for human consumption and environmental protection. Non-trivial purification of water involves removal of toxic ions, organic impurities, microbes and their by-products as well as scooping oil spills. The removal of organic contaminants from water is a major industrial concern. The challenging goal here is to detect, decompose and remove contaminants present usually in low concentrations. Towards this end, different types of sorbent materials have been developed to date, the most common being activated carbon. Though the use of activated carbon is still considered to be one of the best method, the disposal of adsorbed contaminants along with the adsorbent is a major concern. Researchers in India have recently come up with an innovative method for organic pollutant removal from waste water.
Jun 7th, 2011
 Polyurethane (PU) foam is an extremely versatile material that commonly is used in bedding, upholstery and building insulation. However, PU foam is very flammable, often resulting in dripping of melted material that enhances flame spread through the formation of a pool fire under the burning object. Brominated flame retardant compounds (e.g. pentabromodiphenyl ether) have been used to reduce foam flammability but there is growing evidence that these chemicals are toxic to the environment and living organisms. Replacing brominated flame retardants in polymer formulations with safer and more environmentally-friendly alternatives has also sparked the interest of nanoscientists. One recent effort to create an environmentally-friendly flame retardant system involves the layer-by-layer assembly of thin films using materials obtained from completely renewable sources.
Polyurethane (PU) foam is an extremely versatile material that commonly is used in bedding, upholstery and building insulation. However, PU foam is very flammable, often resulting in dripping of melted material that enhances flame spread through the formation of a pool fire under the burning object. Brominated flame retardant compounds (e.g. pentabromodiphenyl ether) have been used to reduce foam flammability but there is growing evidence that these chemicals are toxic to the environment and living organisms. Replacing brominated flame retardants in polymer formulations with safer and more environmentally-friendly alternatives has also sparked the interest of nanoscientists. One recent effort to create an environmentally-friendly flame retardant system involves the layer-by-layer assembly of thin films using materials obtained from completely renewable sources.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





