Showing Spotlights 889 - 896 of 2793 in category All (newest first):
 Caenorhabditis elegans, a free-living soil nematode, has become an important experimental model for biomedical research. This nematode has been successfully employed in genetics, ageing research, behavioral assays, drug screening and (nano)toxicology. In a new study, researchers report for the first time the effective imaging of the nanoscale structure of C. elegans nematodes' cuticle using atomic force microscopy operating in PeakForce Tapping mode. This mode allows imaging C. elegans nematodes both in air and in their native liquid environment.
Caenorhabditis elegans, a free-living soil nematode, has become an important experimental model for biomedical research. This nematode has been successfully employed in genetics, ageing research, behavioral assays, drug screening and (nano)toxicology. In a new study, researchers report for the first time the effective imaging of the nanoscale structure of C. elegans nematodes' cuticle using atomic force microscopy operating in PeakForce Tapping mode. This mode allows imaging C. elegans nematodes both in air and in their native liquid environment.
Nov 16th, 2016
 Scientists have now demonstrated in detail how chemical purity of perovskite precursors can affect the morphology of thin-film perovskite layers. While most studies concentrate on the exploration of processing conditions, the research team has investigated the purity levels of common perovskite precursor solutions and found a number of impurities that are critically important toward controlling the crystallization of perovskites. They observed that impurities that formed during the preparation of perovskite precursor solutions initiate formation of PbHPO3 nanoparticles, which helps to growth larger perovskite crystal grains resulting in higher solar cells efficiency.
Scientists have now demonstrated in detail how chemical purity of perovskite precursors can affect the morphology of thin-film perovskite layers. While most studies concentrate on the exploration of processing conditions, the research team has investigated the purity levels of common perovskite precursor solutions and found a number of impurities that are critically important toward controlling the crystallization of perovskites. They observed that impurities that formed during the preparation of perovskite precursor solutions initiate formation of PbHPO3 nanoparticles, which helps to growth larger perovskite crystal grains resulting in higher solar cells efficiency.
Nov 15th, 2016
 Researchers have demonstrated a fully stretchable energy harvester for thermal waste, which is very simple to fabricate and uses inexpensive substrate materials such polymers or paper. Thermoelectric generators (TEGs) promise a cheap and pragmatic way to obtain energy out of waste heat. The novelty of this work is to effectively integrate the high-performance of inorganic thermoelectric materials with the mechanical advantages of affordable organic materials (polymers, paper) and the use of innovative geometries that can be inherently stretched (spirals, helixes).
Researchers have demonstrated a fully stretchable energy harvester for thermal waste, which is very simple to fabricate and uses inexpensive substrate materials such polymers or paper. Thermoelectric generators (TEGs) promise a cheap and pragmatic way to obtain energy out of waste heat. The novelty of this work is to effectively integrate the high-performance of inorganic thermoelectric materials with the mechanical advantages of affordable organic materials (polymers, paper) and the use of innovative geometries that can be inherently stretched (spirals, helixes).
Nov 14th, 2016
 Researchers have demonstrated that nitric-oxide releasing nanoparticles interfere with Staphylococcus aureus (S. aureus) adhesion and prevent biofilm formation on a rat central venous catheters model of infection. Specifically, they demonstrated that a well studied nitric oxide-releasing nanoparticle platform (NO-np) has the potential to reduce the incidence and/or treat central venous catheter infections. The investigators examined the formation of staphylococcal biofilms by confocal and scanning electron microscopy and found that treatment of staphylococcal biofilms with NO-np significantly reduced biofilm thickness and bacterial number compared to control biofilms.
Researchers have demonstrated that nitric-oxide releasing nanoparticles interfere with Staphylococcus aureus (S. aureus) adhesion and prevent biofilm formation on a rat central venous catheters model of infection. Specifically, they demonstrated that a well studied nitric oxide-releasing nanoparticle platform (NO-np) has the potential to reduce the incidence and/or treat central venous catheter infections. The investigators examined the formation of staphylococcal biofilms by confocal and scanning electron microscopy and found that treatment of staphylococcal biofilms with NO-np significantly reduced biofilm thickness and bacterial number compared to control biofilms.
Nov 11th, 2016
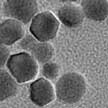 Researchers have, for the first time, used naturally occurring bacterial magnetic nanoparticles (BMPs) - magnetosome extracted from magnetotactic bacteria - to substitute man-made nanoparticles for photothermal cancer therapy. Compared with engineered magnetic nanoparticles, BMPs have specific features such as large-scale production, monodispersity, good biocompatibility, high crystallinity, and close-to-bulk magnetization besides being covered with a lipid bilayer. This layer of biomembrane is particularly useful as it removes the need for a postsynthetic surface modification step for escaping destruction by the body's immune system.
Researchers have, for the first time, used naturally occurring bacterial magnetic nanoparticles (BMPs) - magnetosome extracted from magnetotactic bacteria - to substitute man-made nanoparticles for photothermal cancer therapy. Compared with engineered magnetic nanoparticles, BMPs have specific features such as large-scale production, monodispersity, good biocompatibility, high crystallinity, and close-to-bulk magnetization besides being covered with a lipid bilayer. This layer of biomembrane is particularly useful as it removes the need for a postsynthetic surface modification step for escaping destruction by the body's immune system.
Nov 10th, 2016
 Researchers have demonstrated that full-color 3D meta-holography imaging with extended viewing angles can be realized by a single layer of nanostructured metallic surface. In order to overcome the cross-talk among different colors that normally exists in current metasurface holography, the researchers introduced an off-axis illumination method to shift the holographic image in different colors and successfully reconstructed all visible colors in the imaging area. Taking advantages of the achromatic feature of the structure, the team also demonstrated full-color holography based on seven primary colors and 3D holographic imaging.
Researchers have demonstrated that full-color 3D meta-holography imaging with extended viewing angles can be realized by a single layer of nanostructured metallic surface. In order to overcome the cross-talk among different colors that normally exists in current metasurface holography, the researchers introduced an off-axis illumination method to shift the holographic image in different colors and successfully reconstructed all visible colors in the imaging area. Taking advantages of the achromatic feature of the structure, the team also demonstrated full-color holography based on seven primary colors and 3D holographic imaging.
Nov 9th, 2016
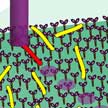 One way to construct useful molecular machines is to combine natural molecules - such as proteins or DNAs in our body - with synthetic molecules in order to control the functions of the natural molecules. Building on previous work that allowed to achieve complete control over on/off switching of the movement of a nanomachine, researchers in Japan have, for the first time, developed a molecular system which allows free control of the motion of single microtubules. The microtubules, tube-like structure with measuring 25 nm in diameter, could potentially serve as carriers of various molecular cargoes in future nano-transportation systems.
One way to construct useful molecular machines is to combine natural molecules - such as proteins or DNAs in our body - with synthetic molecules in order to control the functions of the natural molecules. Building on previous work that allowed to achieve complete control over on/off switching of the movement of a nanomachine, researchers in Japan have, for the first time, developed a molecular system which allows free control of the motion of single microtubules. The microtubules, tube-like structure with measuring 25 nm in diameter, could potentially serve as carriers of various molecular cargoes in future nano-transportation systems.
Nov 8th, 2016
 Carbon nanotubes (CNTs) being highly electrically conductive along the tube axis, have gained great research interests in recent years for connecting two conducting electrodes at the nanoscale - where the CNTs can be integrated into a micro- or nanoelectronic system. Therefore, the orientational control of CNTs has drawn a great deal of research interest in nanotechnology. Researchers now have developed a technique to bridge two electrical conductors by assembling CNTs guided by liquid crystals.
Carbon nanotubes (CNTs) being highly electrically conductive along the tube axis, have gained great research interests in recent years for connecting two conducting electrodes at the nanoscale - where the CNTs can be integrated into a micro- or nanoelectronic system. Therefore, the orientational control of CNTs has drawn a great deal of research interest in nanotechnology. Researchers now have developed a technique to bridge two electrical conductors by assembling CNTs guided by liquid crystals.
Nov 1st, 2016
 Caenorhabditis elegans, a free-living soil nematode, has become an important experimental model for biomedical research. This nematode has been successfully employed in genetics, ageing research, behavioral assays, drug screening and (nano)toxicology. In a new study, researchers report for the first time the effective imaging of the nanoscale structure of C. elegans nematodes' cuticle using atomic force microscopy operating in PeakForce Tapping mode. This mode allows imaging C. elegans nematodes both in air and in their native liquid environment.
Caenorhabditis elegans, a free-living soil nematode, has become an important experimental model for biomedical research. This nematode has been successfully employed in genetics, ageing research, behavioral assays, drug screening and (nano)toxicology. In a new study, researchers report for the first time the effective imaging of the nanoscale structure of C. elegans nematodes' cuticle using atomic force microscopy operating in PeakForce Tapping mode. This mode allows imaging C. elegans nematodes both in air and in their native liquid environment.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





