| Posted: Nov 10, 2016 | |
Magnetic nanoparticles produced by bacteria could help kill cancer cells |
|
| (Nanowerk Spotlight) Photothermal therapy (PTT) is a form of cancer treatment where a therapeutic agent absorbs energy from photons, usually from near-infrared light, and dissipates it partially in the form of heat. When the therapeutic agents, for instance nanoparticles, are located in close vicinity to the tumor site, the temperature increase can lead to cell damage, i.e. it kills the cancer cell. | |
| For PTT to work effectively, it is most important to design photothermal conversion agents with low toxicity and high therapeutic efficiency. | |
| In new work on nanotechnology to kill cancer cells, published in Biomaterials ("Bacterial magnetic nanoparticles for photothermal therapy of cancer under the guidance of MRI"), a team of Chinese scientists have, for the first time, used naturally occurring bacterial magnetic nanoparticles (BMPs) – magnetosome extracted from magnetotactic bacteria – to substitute man-made nanoparticles for photothermal cancer therapy. | |
| Magnetotactic bacteria are organisms which produce chains of organelles (compartments located inside the bacterial cell wall) called magnetosomes that contain magnetic iron nanoparticles. Magnetosomes allow bacteria to orient themselves along the earth's magnetic field lines in order to migrate to more favorable environments. | |
 |
|
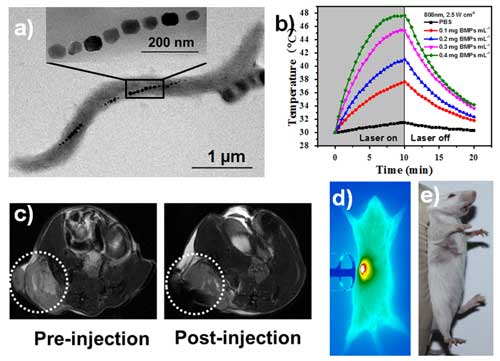
| Magnetic nanoparticles (BMPs) extracted from bacteria help killing cancer cells. a) TEM image of bacteria AMB-1 and BMPs; b) Temperature change with BMPs exposed to NIR laser; c) T2-weighted MRI showing tumor location; d) Infrared Thermal Imaging of tumor tissue under PTT; e) The tumor after PTT has been completely removed. (Image: Beijing Institute of Nanoenergy and Nanosystems) (click on image to enlarge) | |
| "Compared with engineered magnetic nanoparticles, BMPs have specific features such as large-scale production, monodispersity, good biocompatibility, high crystallinity, and close-to-bulk magnetization besides being covered with a lipid bilayer," Prof. Linlin Li from the Beijing Institute of Nanoenergy and Nanosystems, Chinese Academy of Sciences, tells Nanowerk. "In previous reports, BMPs were considered as good materials for the cancer therapy in hyperthermia treatments. With high saturation magnetization, they are also used for MRI in vivo. Up to now, no attention has been paid to the PTT based on BMPs." | |
| BMPs not only act as photothermal conversion agents, explains Li, but also enhance the T2-weighted magnetic resonance imaging (MRI) at the tumor location for guiding the therapy and monitoring the nanoparticle biodistribution. | |
| "Our in vitro and in vivo results all prove that the BMPs have good biocompatibility, high photothermal conversion efficiency, efficient photothermal killing effect on tumors, and T2-weighted MRI," says, Dr. Chuanfang Chen from the Institute of Electrical Engineering, Chinese Academy of Sciences, and a co-author of the paper. "A single dose of therapy could completely remove tumor tissues without recurrence on liver cancer bearing mice." | |
| The layer of biomembrane that covers the BMPs is particularly useful as it removes the need for a postsynthetic surface modification step for escaping destruction by the body's immune system. | |
| Using the BMPs as both photothermal conversion agent and MRI contrast agent opens a new window for highly targeted cancer therapy and cancer theranostics. By integrating active targeting ligands onto the surface of BMPs the team hopes to achieve an even more efficient tumor targeting and therapy. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
