Showing Spotlights 9 - 15 of 15 in category All (newest first):
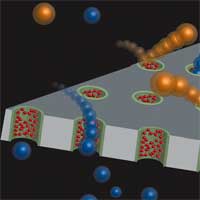 Nanopores function as membrane channels in all living systems, where they serve as sensitive electro-mechanical devices that regulate electrical potential, ionic flow, and molecular transport through the cell membrane. Scientists are studying nanopore construction with the goal of making man-made cell membranes and single molecule detectors. So far, nanoporous membrane-based separations simply use the difference in size of the analytes relative to pore size in the membrane. Here, for a nanopore to be useful as a single molecule detector, its diameter must not be much larger than the size of the molecule to be detected. When a single molecule enters a nanopore in an insulating membrane, it causes changes in the nanopore's electrical properties, which then can be detected and measured. In order to bring about selectivity beyond size, it is necessary that methods for functionalizing the membrane pores are available. To that end, researchers have developed a very simple, but versatile, method for decorating the nanoporous membranes with functional groups. By uniformly modifying the internal cavities of nanopores with various polymers they were able to demonstrate selectivities based on size, charge and hydrophobicity of the molecule passing through the nanopore.
Nanopores function as membrane channels in all living systems, where they serve as sensitive electro-mechanical devices that regulate electrical potential, ionic flow, and molecular transport through the cell membrane. Scientists are studying nanopore construction with the goal of making man-made cell membranes and single molecule detectors. So far, nanoporous membrane-based separations simply use the difference in size of the analytes relative to pore size in the membrane. Here, for a nanopore to be useful as a single molecule detector, its diameter must not be much larger than the size of the molecule to be detected. When a single molecule enters a nanopore in an insulating membrane, it causes changes in the nanopore's electrical properties, which then can be detected and measured. In order to bring about selectivity beyond size, it is necessary that methods for functionalizing the membrane pores are available. To that end, researchers have developed a very simple, but versatile, method for decorating the nanoporous membranes with functional groups. By uniformly modifying the internal cavities of nanopores with various polymers they were able to demonstrate selectivities based on size, charge and hydrophobicity of the molecule passing through the nanopore.
Feb 6th, 2008
 Pipettes (from the French: little pipe) are one of those ubiquitous tools you find in every chemical, medical or biology lab. The original pipette is made of glass and it works by creating a vacuum above the liquid-holding chamber and selectively releasing this vacuum to draw up and dispense liquid. With the advance of molecular biology, researchers required pipettes that were able to confine smaller and smaller amounts of specimen, not only for probing but also for injecting drugs, DNA etc. into cells - without damaging the cells, of course. Just a few months ago, researchers at the Brookhaven National Laboratory have developed what is thought to be the world's smallest pipette: made of a carbon-coated germanium nanowire it can hold a volume of only a few zeptoliters (a billionth of a trillionth of a liter, or one thousand cubic nanometers). Although today it even is possible to process the tip of a glass pipette to have an inner diameter as small as several tens of nanometers, this involves considerable problems in terms of the processing accuracy and the operability of the pipette, particularly in terms of locating the tip. It is generally considered that probes made from carbon nanotubes (CNTs) and nanoscale carbon pipes offer an attractive alternative to glass pipettes because of their small size, high mechanical strength, and high electrical conductivity. Researchers at the University of Pennsylvania have developed a manufacturing technique for carbon nanopipettes (CNPs) that does not require cumbersome nanoassembly and is amenable to mass production.
Pipettes (from the French: little pipe) are one of those ubiquitous tools you find in every chemical, medical or biology lab. The original pipette is made of glass and it works by creating a vacuum above the liquid-holding chamber and selectively releasing this vacuum to draw up and dispense liquid. With the advance of molecular biology, researchers required pipettes that were able to confine smaller and smaller amounts of specimen, not only for probing but also for injecting drugs, DNA etc. into cells - without damaging the cells, of course. Just a few months ago, researchers at the Brookhaven National Laboratory have developed what is thought to be the world's smallest pipette: made of a carbon-coated germanium nanowire it can hold a volume of only a few zeptoliters (a billionth of a trillionth of a liter, or one thousand cubic nanometers). Although today it even is possible to process the tip of a glass pipette to have an inner diameter as small as several tens of nanometers, this involves considerable problems in terms of the processing accuracy and the operability of the pipette, particularly in terms of locating the tip. It is generally considered that probes made from carbon nanotubes (CNTs) and nanoscale carbon pipes offer an attractive alternative to glass pipettes because of their small size, high mechanical strength, and high electrical conductivity. Researchers at the University of Pennsylvania have developed a manufacturing technique for carbon nanopipettes (CNPs) that does not require cumbersome nanoassembly and is amenable to mass production.
Dec 7th, 2007
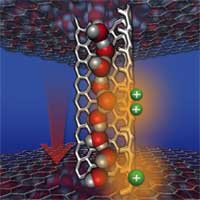 Nanofluidic channels, confining and transporting tiny amounts of fluid, are the pipelines that make the cellular activities of organisms possible. Nanoscale channels carry nutrients into cells and waste from cells and they also transport water into and out of the cell. Body temperature, digestion, reproduction, fluid pressure in the eye, and water conservation in the kidney are only a few of the processes in humans that depend on the proper functioning of cellular water channels. Special proteins called aquaporins can transport water through the cell membrane at a high rate while effectively blocking everything else - even individual protons, the nuclei of hydrogen atoms. The aquaporin channels are so narrow that no molecule larger than water can pass through, effectively forcing them through like beads on a chain. A unique distribution of amino acid residues along the pore wall also accounts for the channel's ability to move water quickly. To keep out molecules smaller than water there is also a chemical filter, formed by the specific orientation and distribution of the amino acid residues lining the pore. Thus water, and only water, flows freely through the aquaporin nanochannels, the direction of flow depending only on changing relative pressure inside and outside the cell. This intriguing mechanism has attracted the attention of nanotechnology researchers who see it as a blueprint for the construction of nanoscale water pumps. A molecular dynamics simulation conducted by Chinese researchers proposes a design for such a molecular pump constructed with a carbon nanotube.
Nanofluidic channels, confining and transporting tiny amounts of fluid, are the pipelines that make the cellular activities of organisms possible. Nanoscale channels carry nutrients into cells and waste from cells and they also transport water into and out of the cell. Body temperature, digestion, reproduction, fluid pressure in the eye, and water conservation in the kidney are only a few of the processes in humans that depend on the proper functioning of cellular water channels. Special proteins called aquaporins can transport water through the cell membrane at a high rate while effectively blocking everything else - even individual protons, the nuclei of hydrogen atoms. The aquaporin channels are so narrow that no molecule larger than water can pass through, effectively forcing them through like beads on a chain. A unique distribution of amino acid residues along the pore wall also accounts for the channel's ability to move water quickly. To keep out molecules smaller than water there is also a chemical filter, formed by the specific orientation and distribution of the amino acid residues lining the pore. Thus water, and only water, flows freely through the aquaporin nanochannels, the direction of flow depending only on changing relative pressure inside and outside the cell. This intriguing mechanism has attracted the attention of nanotechnology researchers who see it as a blueprint for the construction of nanoscale water pumps. A molecular dynamics simulation conducted by Chinese researchers proposes a design for such a molecular pump constructed with a carbon nanotube.
Nov 9th, 2007
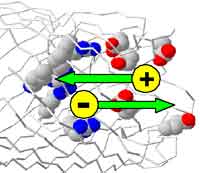 Ion channels are proteins with a hole down their middle that are the gatekeepers for cells. Ion channels control an enormous range of biological function in health and disease. In channels with a diameter greater than 100 nm, the interaction between the channel wall and electrolyte solution hardly affects the flow of ions. When the channel diameter enters the the below-10 nm range, things change dramatically, however. Then, the interaction between the solution and channel wall starts to dominate ionic flow and ion transport through such narrow, nano-scaled channels is dominated by electrostatics. The same is true for biological ion channels where charged amino residues in the selectivity filter determine the ionic flow through the channel, along with the dielectric charge on the channel wall, and the concentrations and potential in the bulk solution. The role electrostatics play in biological pores has been confirmed by numerous mutation studies where amino acids residues in the selectivity filter were replaced by others. Ion channels have simple enough structure that they can be analyzed with the usual tools of physical science. With that analysis in hand, researchers are trying to design practical machines that use ion channels. By exploiting the electrostatics in nanochannels a group of US and Dutch scientists managed to make a diode. Like a solid-state diode allows current flow in one direction, the ionic equivalent they designed can be used to direct the flow of ions across a membrane that separates two electrolyte solutions. Now that they know how to manipulate the ion selectivity in these devices, they hope to be able one day to selectively amplify currents carried by individual chemical species - a stunning prospect for molecular nanoelectronics.
Ion channels are proteins with a hole down their middle that are the gatekeepers for cells. Ion channels control an enormous range of biological function in health and disease. In channels with a diameter greater than 100 nm, the interaction between the channel wall and electrolyte solution hardly affects the flow of ions. When the channel diameter enters the the below-10 nm range, things change dramatically, however. Then, the interaction between the solution and channel wall starts to dominate ionic flow and ion transport through such narrow, nano-scaled channels is dominated by electrostatics. The same is true for biological ion channels where charged amino residues in the selectivity filter determine the ionic flow through the channel, along with the dielectric charge on the channel wall, and the concentrations and potential in the bulk solution. The role electrostatics play in biological pores has been confirmed by numerous mutation studies where amino acids residues in the selectivity filter were replaced by others. Ion channels have simple enough structure that they can be analyzed with the usual tools of physical science. With that analysis in hand, researchers are trying to design practical machines that use ion channels. By exploiting the electrostatics in nanochannels a group of US and Dutch scientists managed to make a diode. Like a solid-state diode allows current flow in one direction, the ionic equivalent they designed can be used to direct the flow of ions across a membrane that separates two electrolyte solutions. Now that they know how to manipulate the ion selectivity in these devices, they hope to be able one day to selectively amplify currents carried by individual chemical species - a stunning prospect for molecular nanoelectronics.
Aug 21st, 2007
 Nanofluidic channels, confining and transporting tiny amounts of fluid, are the pipelines that make the cellular activities of organisms possible. For instance, nanoscale channels carry nutrients into cells and waste from cells. Researchers are trying to mimic Nature by constructing nanochannels in order to be able to manipulate single molecules in, predominantly biomedical, applications. Although nanochannels adjustable in size are prevalent in Nature, it is challenging to fabricate them artificially because of conflicting requirements for rigid structural integrity (to prevent collapse) on one hand and reconfigurability of nanometer-sized features on the other (to allow adjustability). Recent work at the University of Michigan addresses these issues and introduces methods to rapidly prototype structurally stable yet reconfigurable nanochannels. By fabricating tuneable elastomeric nanochannels for nanofluidic manipulation, the researchers were able to properly balance the need for flexibility and rigidity.
Nanofluidic channels, confining and transporting tiny amounts of fluid, are the pipelines that make the cellular activities of organisms possible. For instance, nanoscale channels carry nutrients into cells and waste from cells. Researchers are trying to mimic Nature by constructing nanochannels in order to be able to manipulate single molecules in, predominantly biomedical, applications. Although nanochannels adjustable in size are prevalent in Nature, it is challenging to fabricate them artificially because of conflicting requirements for rigid structural integrity (to prevent collapse) on one hand and reconfigurability of nanometer-sized features on the other (to allow adjustability). Recent work at the University of Michigan addresses these issues and introduces methods to rapidly prototype structurally stable yet reconfigurable nanochannels. By fabricating tuneable elastomeric nanochannels for nanofluidic manipulation, the researchers were able to properly balance the need for flexibility and rigidity.
May 17th, 2007
 Integrating biochemical analysis with micro- and nano-oelectromechanical systems (MEMS and NEMS) has led to the development of a new class of biomedical analytical devices called lab-on-a-chip. They combine a number of biological functions (such as enzymatic reactions, antigen-antibody conjugation, and DNA probing) with proper micro- or even nanofluidic laboratory components (such as sample dilution, pumping, mixing, metering, incubation, or separation) and detection in micron- and nanometer-sized channels and reservoirs into one single device. In order to reduce the size of these lab-on-chip devices even more, researchers increasingly are finding ways to turn micron-sized components into nanosized components. One problem they have been faced with so far is the issue of illumination. Today, many state-of-the-art lab-on-a-chip devices use external illumination sources, such as lasers or light emitting diodes (LEDs). Being able to fully integrate the excitation and detection mechanisms on lab-on-a-chip devices would allow further size reductions and increase the flexibility for using and handling them. Researchers at Cornell University have electrospun light-emitting nanofibers that, if they can be integrated with micro- and nanofluidic devices, could achieve excitation of light-induced fluorescence and detection within that same device.
Integrating biochemical analysis with micro- and nano-oelectromechanical systems (MEMS and NEMS) has led to the development of a new class of biomedical analytical devices called lab-on-a-chip. They combine a number of biological functions (such as enzymatic reactions, antigen-antibody conjugation, and DNA probing) with proper micro- or even nanofluidic laboratory components (such as sample dilution, pumping, mixing, metering, incubation, or separation) and detection in micron- and nanometer-sized channels and reservoirs into one single device. In order to reduce the size of these lab-on-chip devices even more, researchers increasingly are finding ways to turn micron-sized components into nanosized components. One problem they have been faced with so far is the issue of illumination. Today, many state-of-the-art lab-on-a-chip devices use external illumination sources, such as lasers or light emitting diodes (LEDs). Being able to fully integrate the excitation and detection mechanisms on lab-on-a-chip devices would allow further size reductions and increase the flexibility for using and handling them. Researchers at Cornell University have electrospun light-emitting nanofibers that, if they can be integrated with micro- and nanofluidic devices, could achieve excitation of light-induced fluorescence and detection within that same device.
May 15th, 2007
 The ability to detect few or individual molecules in solution is at present largely limited to fluorescence techniques, and a comparable method using electrical detection has so far remained elusive. Such a technique would be highly desirable for lab-on-a-chip applications and when labeling with fluorophores is invasive or impossible. More importantly, it would pave the way for fluidic devices in which individual ions are electrically detected and manipulated, allowing a new class of fundamental experiments on nonequilibrium statistical physics, transport at the molecular scale, and a broad range of biophysical systems. Researchers in The Netherlands now have demonstrated a new nanofluidic device for the detection of electrochemical active molecules with an extremely high sensitivity. A prototype device allows detecting fluctuations due to Brownian motion of as few as approx. 70 molecules, a level heretofore unachieved in electrochemical sensors. Ultimately, the researchers hope the device will not only be able to detect single molecules in the device, but also discriminate between various species.
The ability to detect few or individual molecules in solution is at present largely limited to fluorescence techniques, and a comparable method using electrical detection has so far remained elusive. Such a technique would be highly desirable for lab-on-a-chip applications and when labeling with fluorophores is invasive or impossible. More importantly, it would pave the way for fluidic devices in which individual ions are electrically detected and manipulated, allowing a new class of fundamental experiments on nonequilibrium statistical physics, transport at the molecular scale, and a broad range of biophysical systems. Researchers in The Netherlands now have demonstrated a new nanofluidic device for the detection of electrochemical active molecules with an extremely high sensitivity. A prototype device allows detecting fluctuations due to Brownian motion of as few as approx. 70 molecules, a level heretofore unachieved in electrochemical sensors. Ultimately, the researchers hope the device will not only be able to detect single molecules in the device, but also discriminate between various species.
Jan 17th, 2007
 Nanopores function as membrane channels in all living systems, where they serve as sensitive electro-mechanical devices that regulate electrical potential, ionic flow, and molecular transport through the cell membrane. Scientists are studying nanopore construction with the goal of making man-made cell membranes and single molecule detectors. So far, nanoporous membrane-based separations simply use the difference in size of the analytes relative to pore size in the membrane. Here, for a nanopore to be useful as a single molecule detector, its diameter must not be much larger than the size of the molecule to be detected. When a single molecule enters a nanopore in an insulating membrane, it causes changes in the nanopore's electrical properties, which then can be detected and measured. In order to bring about selectivity beyond size, it is necessary that methods for functionalizing the membrane pores are available. To that end, researchers have developed a very simple, but versatile, method for decorating the nanoporous membranes with functional groups. By uniformly modifying the internal cavities of nanopores with various polymers they were able to demonstrate selectivities based on size, charge and hydrophobicity of the molecule passing through the nanopore.
Nanopores function as membrane channels in all living systems, where they serve as sensitive electro-mechanical devices that regulate electrical potential, ionic flow, and molecular transport through the cell membrane. Scientists are studying nanopore construction with the goal of making man-made cell membranes and single molecule detectors. So far, nanoporous membrane-based separations simply use the difference in size of the analytes relative to pore size in the membrane. Here, for a nanopore to be useful as a single molecule detector, its diameter must not be much larger than the size of the molecule to be detected. When a single molecule enters a nanopore in an insulating membrane, it causes changes in the nanopore's electrical properties, which then can be detected and measured. In order to bring about selectivity beyond size, it is necessary that methods for functionalizing the membrane pores are available. To that end, researchers have developed a very simple, but versatile, method for decorating the nanoporous membranes with functional groups. By uniformly modifying the internal cavities of nanopores with various polymers they were able to demonstrate selectivities based on size, charge and hydrophobicity of the molecule passing through the nanopore.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed




