Showing Spotlights 1 - 8 of 15 in category All (newest first):
 Molecular separations are extremely important in a wide range of technologies, from conventional proteomics to pathogen detection and DNA fingerprinting. A complication arises from the fact that molecular components in mixtures can span an enormous range of concentration. Conventional approaches such as antibody depletion are not sensitive enough to detect numerous medically significant biomarkers, whose incidence in blood could be as much as a trillion times less abundant than the most plentiful protein, albumin. New research shows a new path in miniaturized molecular separations. It describes a new device that demonstrates simultaneous concentration and separation of proteins by conductivity gradient focusing. Concentration and separation take place in an electric-field-driven 120 nm deep nanochannel that supports a stable salt and conductivity gradient. The results show that relevant proteins can be concentrated to detectable levels.
Molecular separations are extremely important in a wide range of technologies, from conventional proteomics to pathogen detection and DNA fingerprinting. A complication arises from the fact that molecular components in mixtures can span an enormous range of concentration. Conventional approaches such as antibody depletion are not sensitive enough to detect numerous medically significant biomarkers, whose incidence in blood could be as much as a trillion times less abundant than the most plentiful protein, albumin. New research shows a new path in miniaturized molecular separations. It describes a new device that demonstrates simultaneous concentration and separation of proteins by conductivity gradient focusing. Concentration and separation take place in an electric-field-driven 120 nm deep nanochannel that supports a stable salt and conductivity gradient. The results show that relevant proteins can be concentrated to detectable levels.
Aug 18th, 2011
 Researchers in Finland and Israel have explored the feasibility of using superhydrophobicity for guided transport of water droplets in microfluidic devices. They demonstrate a new, simple and general approach for transportation of water droplets based on superhydrophobic technology. Water droplets are transported at high velocity in almost totally water-repellent tracks with vertical walls. Drops move in open tracks, machined in metal or silicon wafers, using gravity or using electrostatic charge. Usually, in digital microfluidics, droplets are formed in a system of two immiscible liquids, for example, water in oil, and the liquid volume is transported in microchannels using pumps and valves. In this new approach droplets in air can be used. Employing superhydrophobic tracks, droplets can be transported with low friction and high efficiency.
Researchers in Finland and Israel have explored the feasibility of using superhydrophobicity for guided transport of water droplets in microfluidic devices. They demonstrate a new, simple and general approach for transportation of water droplets based on superhydrophobic technology. Water droplets are transported at high velocity in almost totally water-repellent tracks with vertical walls. Drops move in open tracks, machined in metal or silicon wafers, using gravity or using electrostatic charge. Usually, in digital microfluidics, droplets are formed in a system of two immiscible liquids, for example, water in oil, and the liquid volume is transported in microchannels using pumps and valves. In this new approach droplets in air can be used. Employing superhydrophobic tracks, droplets can be transported with low friction and high efficiency.
Aug 9th, 2011
 Structural color - where the color results from interferences, not pigments - is a natural fit for new display technologies and writing substrates since it can provide vivid colors in environments of high intensity light (e.g. sunlight) without the need for additional illuminating power. Most existing display technologies such as LCDs and LEDs require power to actively project colors and often suffer from performance degradations in these reflective environments. Recognizing this, researchers have developed a number of techniques for dynamically and spatially modulating the colors reflected off of photonic crystal-like substrates. In new work, researchers have demonstrated an electrical-power-free infusion technique that allows them to draw multi-color images on colloidal photonic crystal substrates using transparent imaging oils.
Structural color - where the color results from interferences, not pigments - is a natural fit for new display technologies and writing substrates since it can provide vivid colors in environments of high intensity light (e.g. sunlight) without the need for additional illuminating power. Most existing display technologies such as LCDs and LEDs require power to actively project colors and often suffer from performance degradations in these reflective environments. Recognizing this, researchers have developed a number of techniques for dynamically and spatially modulating the colors reflected off of photonic crystal-like substrates. In new work, researchers have demonstrated an electrical-power-free infusion technique that allows them to draw multi-color images on colloidal photonic crystal substrates using transparent imaging oils.
Aug 2nd, 2011
 In order to improve throughput speed of DNA sequencing and reduce its cost, researchers are pursuing real-time solid-state DNA sequencing devices. To that end, electronic functional devices in liquid environments need to be developed, ideally utilizing the compatibility with current complementary metal oxide semiconductor (CMOS) based fabrication technology. In this regard, the combination of electronics and nanofluidics leads to the field of electrofluidics, which utilizes the electrical behaviors of fluids for solid state device applications. In order to explore the ion transport and biomolecule transport through nanochannels, researchers have now reported the fabrication of an electrofluidic platform to study the motion of single molecules, including DNA. The device's nanochannel structures were fabricated with sub-lithographic dimension through top-down based, conventional semiconductor fabrication methods.
In order to improve throughput speed of DNA sequencing and reduce its cost, researchers are pursuing real-time solid-state DNA sequencing devices. To that end, electronic functional devices in liquid environments need to be developed, ideally utilizing the compatibility with current complementary metal oxide semiconductor (CMOS) based fabrication technology. In this regard, the combination of electronics and nanofluidics leads to the field of electrofluidics, which utilizes the electrical behaviors of fluids for solid state device applications. In order to explore the ion transport and biomolecule transport through nanochannels, researchers have now reported the fabrication of an electrofluidic platform to study the motion of single molecules, including DNA. The device's nanochannel structures were fabricated with sub-lithographic dimension through top-down based, conventional semiconductor fabrication methods.
Oct 14th, 2010
 In contrast with microchannel-based fluidics, the manipulation of discrete droplets without using microfluidic channels is a new field. Here, a liquid droplet is not confined to a closed channel and there is no risk of being adsorbed on a channel wall. A liquid marble, a liquid encapsulated by non-wetting powder, could be a new microfluidic device, which is especially useful for handling single liquid droplet. One of the challenges for using liquid marbles as microfluidic devices is the communication between the liquid droplet and the external devices/materials. Researchers in Australia have been trying to develop 'field-responsive smart liquid marbles' which can be opened and closed reversibly on demand, such that the liquid in the marble can be easily taken and other liquid can also be added into the marble easily. The mechanically robust magnetic liquid marble, prepared by coating a water droplet with highly hydrophobic magnetite nanoparticles, can be actuated magnetically.
In contrast with microchannel-based fluidics, the manipulation of discrete droplets without using microfluidic channels is a new field. Here, a liquid droplet is not confined to a closed channel and there is no risk of being adsorbed on a channel wall. A liquid marble, a liquid encapsulated by non-wetting powder, could be a new microfluidic device, which is especially useful for handling single liquid droplet. One of the challenges for using liquid marbles as microfluidic devices is the communication between the liquid droplet and the external devices/materials. Researchers in Australia have been trying to develop 'field-responsive smart liquid marbles' which can be opened and closed reversibly on demand, such that the liquid in the marble can be easily taken and other liquid can also be added into the marble easily. The mechanically robust magnetic liquid marble, prepared by coating a water droplet with highly hydrophobic magnetite nanoparticles, can be actuated magnetically.
Feb 10th, 2010
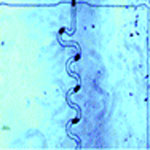 A lot of the scientific knowledge in chemistry and biology comes from experiments on ensembles of molecules by which a vast number of duplicate behaviors are investigated and averaged responses are recorded. Especially the ability to make measurements at the single molecule level provides crucial information about biological and chemical systems. This research depends on molecular manipulation technologies that are able to isolate individual molecules and sequentially transport them for measurement and, potentially, manipulation. To this end, generation and manipulation of small, highly monodisperse droplets have received a lot of attention in the biotech community recently. Using a simple droplet generator chip, scientists can generate millions of droplets in a short amount of time. Each individual droplet is isolated from another droplet, hence, meaning that a million droplets constitute a million individual microreactors running a million reactions simultaneously.
A lot of the scientific knowledge in chemistry and biology comes from experiments on ensembles of molecules by which a vast number of duplicate behaviors are investigated and averaged responses are recorded. Especially the ability to make measurements at the single molecule level provides crucial information about biological and chemical systems. This research depends on molecular manipulation technologies that are able to isolate individual molecules and sequentially transport them for measurement and, potentially, manipulation. To this end, generation and manipulation of small, highly monodisperse droplets have received a lot of attention in the biotech community recently. Using a simple droplet generator chip, scientists can generate millions of droplets in a short amount of time. Each individual droplet is isolated from another droplet, hence, meaning that a million droplets constitute a million individual microreactors running a million reactions simultaneously.
Nov 25th, 2009
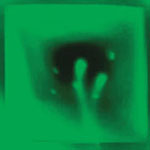 Controlling surface plasmons has become increasingly attractive for optical signal processing, surface enhanced spectroscopy and sensor nanotechnology. For instance, the role of surface plasmon resonance (SPR) on resonant transmission through nanohole arrays has motivated their application as surface-based biosensors. New work by a team of scientists in Canada has combined nanofluidics and nanoplasmonics for SPR sensing using flow-through nanohole arrays. This new format enables rapid transport of reactants to the active sensing surface and the array serves as a sieve. That is, the flow-through array efficiently collects and detects biomarkers from a very small volume of fluid.
Controlling surface plasmons has become increasingly attractive for optical signal processing, surface enhanced spectroscopy and sensor nanotechnology. For instance, the role of surface plasmon resonance (SPR) on resonant transmission through nanohole arrays has motivated their application as surface-based biosensors. New work by a team of scientists in Canada has combined nanofluidics and nanoplasmonics for SPR sensing using flow-through nanohole arrays. This new format enables rapid transport of reactants to the active sensing surface and the array serves as a sieve. That is, the flow-through array efficiently collects and detects biomarkers from a very small volume of fluid.
Jul 17th, 2009
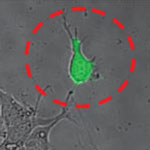 The Atomic Force Microscope (AFM) is a key tool for nanotechnology. This instrument has become the most widely used tool for imaging, measuring and manipulating matter at the nanoscale and in turn has inspired a variety of other scanning probe techniques. Originally the AFM was used to image the topography of surfaces, but by modifying the tip it is possible to measure other quantities (for example, electric and magnetic properties, chemical potentials, friction and so on), and also to perform various types of spectroscopy and analysis. Increasingly, the AFM is also becoming a tool for nanofabrication. Relatively new is the use of AFM in cell biology. Researchers in Switzerland have now demonstrated novel cell biology applications using hollow force-controlled AFM cantilevers - a new device they have called FluidFM.
The Atomic Force Microscope (AFM) is a key tool for nanotechnology. This instrument has become the most widely used tool for imaging, measuring and manipulating matter at the nanoscale and in turn has inspired a variety of other scanning probe techniques. Originally the AFM was used to image the topography of surfaces, but by modifying the tip it is possible to measure other quantities (for example, electric and magnetic properties, chemical potentials, friction and so on), and also to perform various types of spectroscopy and analysis. Increasingly, the AFM is also becoming a tool for nanofabrication. Relatively new is the use of AFM in cell biology. Researchers in Switzerland have now demonstrated novel cell biology applications using hollow force-controlled AFM cantilevers - a new device they have called FluidFM.
Jul 8th, 2009
 Molecular separations are extremely important in a wide range of technologies, from conventional proteomics to pathogen detection and DNA fingerprinting. A complication arises from the fact that molecular components in mixtures can span an enormous range of concentration. Conventional approaches such as antibody depletion are not sensitive enough to detect numerous medically significant biomarkers, whose incidence in blood could be as much as a trillion times less abundant than the most plentiful protein, albumin. New research shows a new path in miniaturized molecular separations. It describes a new device that demonstrates simultaneous concentration and separation of proteins by conductivity gradient focusing. Concentration and separation take place in an electric-field-driven 120 nm deep nanochannel that supports a stable salt and conductivity gradient. The results show that relevant proteins can be concentrated to detectable levels.
Molecular separations are extremely important in a wide range of technologies, from conventional proteomics to pathogen detection and DNA fingerprinting. A complication arises from the fact that molecular components in mixtures can span an enormous range of concentration. Conventional approaches such as antibody depletion are not sensitive enough to detect numerous medically significant biomarkers, whose incidence in blood could be as much as a trillion times less abundant than the most plentiful protein, albumin. New research shows a new path in miniaturized molecular separations. It describes a new device that demonstrates simultaneous concentration and separation of proteins by conductivity gradient focusing. Concentration and separation take place in an electric-field-driven 120 nm deep nanochannel that supports a stable salt and conductivity gradient. The results show that relevant proteins can be concentrated to detectable levels.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





