Showing Spotlights 161 - 168 of 203 in category All (newest first):
 The catalytic conversion of chemical to mechanical energy, which is ubiquitous in biological systems, also is the basis for many of the engine systems that nanotechnology researchers are developing. Catalytic 'engines' will be key components of active micron- and sub-micron scale systems for controlled movement, particle assembly, and separations. So far, most of these catalytic micro- and nanomotors use hydrogen peroxide as the fuel. The major problem associated with this is that the produced oxygen bubbles make the observation and detailed study of these motors difficult. Researchers at the Pennsylvania State University have now introduced a new bubble-free, high efficient nanomotor system that involves the operation of a miniaturized copper-platinum nanobattery.
The catalytic conversion of chemical to mechanical energy, which is ubiquitous in biological systems, also is the basis for many of the engine systems that nanotechnology researchers are developing. Catalytic 'engines' will be key components of active micron- and sub-micron scale systems for controlled movement, particle assembly, and separations. So far, most of these catalytic micro- and nanomotors use hydrogen peroxide as the fuel. The major problem associated with this is that the produced oxygen bubbles make the observation and detailed study of these motors difficult. Researchers at the Pennsylvania State University have now introduced a new bubble-free, high efficient nanomotor system that involves the operation of a miniaturized copper-platinum nanobattery.
Oct 17th, 2011
 Harvesting unexploited energy in the living environment to power small electronic devices and systems is increasingly attracting the attention of research groups around the world. As device sizes shrink to the micro- and even nanoscale, power consumption also decreases to ever lower levels, i.e. microwatts to milliwatts range. And as research findings have shown over the past few years, it is entirely possible to drive such minuscule devices by directly scavenging energy from their working environment. A recent example is the integration of nanogenerators into the inner surface of tires, demonstrating the possibility of energy harvesting from the motion of automobiles.
Harvesting unexploited energy in the living environment to power small electronic devices and systems is increasingly attracting the attention of research groups around the world. As device sizes shrink to the micro- and even nanoscale, power consumption also decreases to ever lower levels, i.e. microwatts to milliwatts range. And as research findings have shown over the past few years, it is entirely possible to drive such minuscule devices by directly scavenging energy from their working environment. A recent example is the integration of nanogenerators into the inner surface of tires, demonstrating the possibility of energy harvesting from the motion of automobiles.
Sep 14th, 2011
 Clean and affordable energy generation and storage is one of the most significant challenges that our world is facing in the 21st century. Materials are going to play a crucial role in generation and storage of renewable energy. While searching for new materials for electrical energy storage, materials scientists have discovered a new family of two-dimensional compounds proposed to have unique properties that may lead to ground-breaking advances in energy storage technology. Researchers transformed three dimensional titanium-aluminum carbide into a two dimensional structure with greatly different properties. This work opens the door for a wide range of metal carbide and/or nitride compositions in form of 2-D sheets.
Clean and affordable energy generation and storage is one of the most significant challenges that our world is facing in the 21st century. Materials are going to play a crucial role in generation and storage of renewable energy. While searching for new materials for electrical energy storage, materials scientists have discovered a new family of two-dimensional compounds proposed to have unique properties that may lead to ground-breaking advances in energy storage technology. Researchers transformed three dimensional titanium-aluminum carbide into a two dimensional structure with greatly different properties. This work opens the door for a wide range of metal carbide and/or nitride compositions in form of 2-D sheets.
Aug 29th, 2011
 Surface energy is ubiquitous in nature and it plays an important role in many scientific areas such as for instance surface physics, biophysics, surface chemistry, or catalysis. Prior to the area of nanotechnology it has been impractical to consider utilizing surface energy as an energy source because there are few molecules or atoms involved in the surface interaction and the density of surface energy is low. Now, however, due to the lower power consumption requirements of nanoscale devices and the higher specific surface area for nanomaterials it appears attractive to use surface energy at the nanoscale. In new work, researchers have investigated how the flow of water over surfaces coated with graphene could generate small amounts of electricity.
Surface energy is ubiquitous in nature and it plays an important role in many scientific areas such as for instance surface physics, biophysics, surface chemistry, or catalysis. Prior to the area of nanotechnology it has been impractical to consider utilizing surface energy as an energy source because there are few molecules or atoms involved in the surface interaction and the density of surface energy is low. Now, however, due to the lower power consumption requirements of nanoscale devices and the higher specific surface area for nanomaterials it appears attractive to use surface energy at the nanoscale. In new work, researchers have investigated how the flow of water over surfaces coated with graphene could generate small amounts of electricity.
Jul 25th, 2011
 Energy generation and storage is an important issue at the nanoscale. For tiny devices such as nano- and micro-electromechanical systems, autonomous power sources are crucial for practical applications. Progress is being made in designing and fabricating nanoscale power generators. But, as with the large, macroscale systems of future smart grids, there might be times when powered nanodevices need to bridge a slump in power generation/harvesting or they might be designed to run on stored energy altogether for a limited period of time. Researchers in China are now proposing that the high energy density and power density of carbon nanotubes makes them promising materials for the storage of mechanical energy. The team provides a structural model towards mechanical energy storage for nanodevices and also demonstrates a method to characterize and manipulate ultralong CNTs.
Energy generation and storage is an important issue at the nanoscale. For tiny devices such as nano- and micro-electromechanical systems, autonomous power sources are crucial for practical applications. Progress is being made in designing and fabricating nanoscale power generators. But, as with the large, macroscale systems of future smart grids, there might be times when powered nanodevices need to bridge a slump in power generation/harvesting or they might be designed to run on stored energy altogether for a limited period of time. Researchers in China are now proposing that the high energy density and power density of carbon nanotubes makes them promising materials for the storage of mechanical energy. The team provides a structural model towards mechanical energy storage for nanodevices and also demonstrates a method to characterize and manipulate ultralong CNTs.
Jul 11th, 2011
 The efforts undertaken in developing renewable energy sources to reduce our dependence on fossil fuels include major research and investment in advanced electricity storage technologies. Among the various existing technologies, lithium batteries are considered as the most competitive power source because of their high energy density, superior power capability, design flexibility and longer lifespan. This article provides an overview of the current patent landscape of rechargeable Li-ion battery, with a focus on the recent developments on nanomaterials and nanotechnologies used for anode, cathode, and electrolyte materials, and the impact of nanomaterials on the performance of rechargeable lithium batteries. Effort has also been taken to identify key players, emerging trends and applications in this area.
The efforts undertaken in developing renewable energy sources to reduce our dependence on fossil fuels include major research and investment in advanced electricity storage technologies. Among the various existing technologies, lithium batteries are considered as the most competitive power source because of their high energy density, superior power capability, design flexibility and longer lifespan. This article provides an overview of the current patent landscape of rechargeable Li-ion battery, with a focus on the recent developments on nanomaterials and nanotechnologies used for anode, cathode, and electrolyte materials, and the impact of nanomaterials on the performance of rechargeable lithium batteries. Effort has also been taken to identify key players, emerging trends and applications in this area.
Jul 1st, 2011
 Notwithstanding all the buzz about renewable energy sources, the dirty facts are that coal accounts for 41% of electricity production worldwide. Since, realistically, coal will be a mainstay of electricity generation for many years to come, research into more environmentally friendly use of coal energy is picking up steam. One technology for more efficient power production centers around the solid oxide fuel cell (SOFC). Especially gasified carbon fuel cells offer great prospects for the most efficient utilization of a wide variety of carbonaceous solids fuels, including coal, biomass, and municipal solid waste. Researchers have now developed a self-cleaning technique that could allow solid oxide fuel cells to be powered directly by coal gas at operating temperatures as low as 750 degrees Celsius.
Notwithstanding all the buzz about renewable energy sources, the dirty facts are that coal accounts for 41% of electricity production worldwide. Since, realistically, coal will be a mainstay of electricity generation for many years to come, research into more environmentally friendly use of coal energy is picking up steam. One technology for more efficient power production centers around the solid oxide fuel cell (SOFC). Especially gasified carbon fuel cells offer great prospects for the most efficient utilization of a wide variety of carbonaceous solids fuels, including coal, biomass, and municipal solid waste. Researchers have now developed a self-cleaning technique that could allow solid oxide fuel cells to be powered directly by coal gas at operating temperatures as low as 750 degrees Celsius.
Jun 28th, 2011
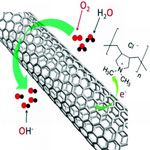 The efficiency of catalyzing the oxygen reduction reaction (ORR) - the process that breaks the bonds of oxygen molecules - to a large degree determines the electrochemical performance of fuel cells. Platinum and platinum-based composites have long been considered as the most efficient ORR catalysts. In their search for practically viable non-precious metal ORR catalysts, researchers have also been investigating vertically-aligned nitrogen-containing carbon nanotubes. Having a strong electron-withdrawing ability, poly(diallyldimethylammonium chloride) (PDDA) was used to create net positive charge for carbon atoms in the nanotube carbon plane via intermolecular charge transfer. The resultant PDDA functionalized/adsorbed carbon nanotubes were demonstrated to act as metal-free catalysts for oxygen reduction reaction in fuel cells with similar performance as platinum catalysts.
The efficiency of catalyzing the oxygen reduction reaction (ORR) - the process that breaks the bonds of oxygen molecules - to a large degree determines the electrochemical performance of fuel cells. Platinum and platinum-based composites have long been considered as the most efficient ORR catalysts. In their search for practically viable non-precious metal ORR catalysts, researchers have also been investigating vertically-aligned nitrogen-containing carbon nanotubes. Having a strong electron-withdrawing ability, poly(diallyldimethylammonium chloride) (PDDA) was used to create net positive charge for carbon atoms in the nanotube carbon plane via intermolecular charge transfer. The resultant PDDA functionalized/adsorbed carbon nanotubes were demonstrated to act as metal-free catalysts for oxygen reduction reaction in fuel cells with similar performance as platinum catalysts.
Mar 21st, 2011
 The catalytic conversion of chemical to mechanical energy, which is ubiquitous in biological systems, also is the basis for many of the engine systems that nanotechnology researchers are developing. Catalytic 'engines' will be key components of active micron- and sub-micron scale systems for controlled movement, particle assembly, and separations. So far, most of these catalytic micro- and nanomotors use hydrogen peroxide as the fuel. The major problem associated with this is that the produced oxygen bubbles make the observation and detailed study of these motors difficult. Researchers at the Pennsylvania State University have now introduced a new bubble-free, high efficient nanomotor system that involves the operation of a miniaturized copper-platinum nanobattery.
The catalytic conversion of chemical to mechanical energy, which is ubiquitous in biological systems, also is the basis for many of the engine systems that nanotechnology researchers are developing. Catalytic 'engines' will be key components of active micron- and sub-micron scale systems for controlled movement, particle assembly, and separations. So far, most of these catalytic micro- and nanomotors use hydrogen peroxide as the fuel. The major problem associated with this is that the produced oxygen bubbles make the observation and detailed study of these motors difficult. Researchers at the Pennsylvania State University have now introduced a new bubble-free, high efficient nanomotor system that involves the operation of a miniaturized copper-platinum nanobattery.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





