Showing Spotlights 1729 - 1736 of 2782 in category All (newest first):
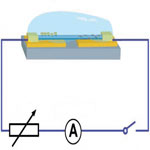 Nanotechnology researchers working on self-powered nanodevices - nanoscale systems that scavenge energy from their surrounding environment - have been experimenting with various power sources ranging from piezoelectric systems to sound. However, the most abundant energy available in biosystems is chemical and biochemical energy, such as glucose. Researchers in China have now reported a nanowire-based biofuel cell based on a single proton conductive polymer nanowire for converting chemical energy from biofluids into electricity, using glucose oxidase and laccase as catalyst. The output of this biofuel cell is sufficient to drive pH, glucose or photon sensors. The high output power, low cost and easy fabrication process, large-scale manufacturability, high 'on-chip' integrability and stability demonstrates its great potential for in vivo biosensing.
Nanotechnology researchers working on self-powered nanodevices - nanoscale systems that scavenge energy from their surrounding environment - have been experimenting with various power sources ranging from piezoelectric systems to sound. However, the most abundant energy available in biosystems is chemical and biochemical energy, such as glucose. Researchers in China have now reported a nanowire-based biofuel cell based on a single proton conductive polymer nanowire for converting chemical energy from biofluids into electricity, using glucose oxidase and laccase as catalyst. The output of this biofuel cell is sufficient to drive pH, glucose or photon sensors. The high output power, low cost and easy fabrication process, large-scale manufacturability, high 'on-chip' integrability and stability demonstrates its great potential for in vivo biosensing.
Jan 3rd, 2011
 Although the literature grows on the use of science to inform decisions on the environmental, health and safety implications of nanotechnology, little has been published by those who make such decisions. In a recent commentary in Nature Nanotechnology, officials of the US Environmental Protection Agency (EPA), the European Commission and the Organisation for Economic Co-operation and Development (OECD), discuss the types of decision facing government regulators, the new considerations nanotechnology brings to decision-making, the role of science in informing decisions, how regulators cooperate internationally on policy issues, and the challenges that lie ahead. The authors provide an overview of key reports and regulations and then discuss the complexity of issues with regard to addressing nanomaterials within the context of existing regulations and the need to weigh nanomaterial risk and benefits.
Although the literature grows on the use of science to inform decisions on the environmental, health and safety implications of nanotechnology, little has been published by those who make such decisions. In a recent commentary in Nature Nanotechnology, officials of the US Environmental Protection Agency (EPA), the European Commission and the Organisation for Economic Co-operation and Development (OECD), discuss the types of decision facing government regulators, the new considerations nanotechnology brings to decision-making, the role of science in informing decisions, how regulators cooperate internationally on policy issues, and the challenges that lie ahead. The authors provide an overview of key reports and regulations and then discuss the complexity of issues with regard to addressing nanomaterials within the context of existing regulations and the need to weigh nanomaterial risk and benefits.
Dec 21st, 2010
 'Nano Textiles' can be produced by a variety of methods. The key difference among them is whether synthetic nanoparticles are integrated into the fibres or the textile, or are applied as a coating on the surface, and/or whether nanoparticles are added to the nanoscale fibres or coating. This article clarifies nano-textile manufacturing processes and application areas, and gives an overview about the potential effects on the environment and health. Many questions remain unanswered, however, there is a need for considerably more research not only for product development but also into the usefulness and risks which nano-textiles give rise to. The open questions have prompted the Swiss Textile Federation to undertake a joint project with the Swiss Federal Laboratories for Materials Science and Technology (EMPA) entitled 'Nanosafe Textiles' and to initiate discussions on the topic.
'Nano Textiles' can be produced by a variety of methods. The key difference among them is whether synthetic nanoparticles are integrated into the fibres or the textile, or are applied as a coating on the surface, and/or whether nanoparticles are added to the nanoscale fibres or coating. This article clarifies nano-textile manufacturing processes and application areas, and gives an overview about the potential effects on the environment and health. Many questions remain unanswered, however, there is a need for considerably more research not only for product development but also into the usefulness and risks which nano-textiles give rise to. The open questions have prompted the Swiss Textile Federation to undertake a joint project with the Swiss Federal Laboratories for Materials Science and Technology (EMPA) entitled 'Nanosafe Textiles' and to initiate discussions on the topic.
Dec 16th, 2010
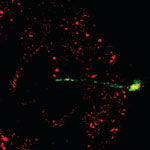 With the advance of nanomedicine, bio-nanotechnology, and molecular biology, researchers require tools that allow them to work on a single cell level. These tools are required to probe individual cells, monitor their processes, and control/alter their functions through nanosurgery procedures and injection of drugs, DNA etc. - all without damaging the cells, of course. Researchers have now developed a multifunctional endoscope-like device, using individual CNTs for prolonged intracellular probing at the single-organelle level, without any recordable disturbance to the metabolism of the cell. These endoscopes can transport attoliter volumes of fluid, record picoampere signals from cells, and can be manipulated magnetically. Furthermore, the tip deflects with submicrometer resolution, and the attachment of gold nanoparticles allows intracellular fingerprinting using surface-enhanced Raman spectroscopy (SERS).
With the advance of nanomedicine, bio-nanotechnology, and molecular biology, researchers require tools that allow them to work on a single cell level. These tools are required to probe individual cells, monitor their processes, and control/alter their functions through nanosurgery procedures and injection of drugs, DNA etc. - all without damaging the cells, of course. Researchers have now developed a multifunctional endoscope-like device, using individual CNTs for prolonged intracellular probing at the single-organelle level, without any recordable disturbance to the metabolism of the cell. These endoscopes can transport attoliter volumes of fluid, record picoampere signals from cells, and can be manipulated magnetically. Furthermore, the tip deflects with submicrometer resolution, and the attachment of gold nanoparticles allows intracellular fingerprinting using surface-enhanced Raman spectroscopy (SERS).
Dec 15th, 2010
 Graphene has attracted a huge amount of attention in recent years with its extraordinarily high electrical and thermal conductivities, mechanical, chemical properties, and large surface area. In order to meet the various demands for a variety of applications, preparation of desired structures of graphene sheets with controlled dimension and architecture are of significant importance. While research on flat graphene oxide structures has become quite common, there have been few reports on the preparation of hollow capsules of graphene through the nanosize, controlled assembly of graphene. Researchers in South Korea have now demonstrated the formation of graphene-based capsules through layer-by-layer (LbL) assembly of surface-functionalized reduced graphene oxide nanosheets of opposite charges onto polystyrene colloidal particles to produce multilayer thin films of graphene nanosheets.
Graphene has attracted a huge amount of attention in recent years with its extraordinarily high electrical and thermal conductivities, mechanical, chemical properties, and large surface area. In order to meet the various demands for a variety of applications, preparation of desired structures of graphene sheets with controlled dimension and architecture are of significant importance. While research on flat graphene oxide structures has become quite common, there have been few reports on the preparation of hollow capsules of graphene through the nanosize, controlled assembly of graphene. Researchers in South Korea have now demonstrated the formation of graphene-based capsules through layer-by-layer (LbL) assembly of surface-functionalized reduced graphene oxide nanosheets of opposite charges onto polystyrene colloidal particles to produce multilayer thin films of graphene nanosheets.
Dec 14th, 2010
 Researchers worldwide are working on fast and low-cost strategies to sequence DNA, that is, to read off the content of our genome. Particularly promising for future genome sequencing are devices that measure single molecules. In this respect, the creation of nanochannels or nanopores in thin membranes has attracted much interest due to the potential to isolate and sense single DNA molecules while they translocate through the highly confined channels. Particularly interesting are techniques that can offer fast and low cost readout of long DNA molecules without the need of DNA labelling or amplification. In very interesting work performed at Imperial College London, researchers have now successfully developed a protocol for the fabrication of a solid state nanopore aligned to a tunneling junction.
Researchers worldwide are working on fast and low-cost strategies to sequence DNA, that is, to read off the content of our genome. Particularly promising for future genome sequencing are devices that measure single molecules. In this respect, the creation of nanochannels or nanopores in thin membranes has attracted much interest due to the potential to isolate and sense single DNA molecules while they translocate through the highly confined channels. Particularly interesting are techniques that can offer fast and low cost readout of long DNA molecules without the need of DNA labelling or amplification. In very interesting work performed at Imperial College London, researchers have now successfully developed a protocol for the fabrication of a solid state nanopore aligned to a tunneling junction.
Dec 13th, 2010
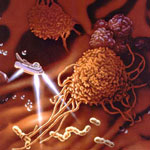 Off-targeting remains a key challenge of researchers working on nanoparticle drug delivery - the majority of intravenously administered therapeutic nanoparticles are also reaching normal tissues, resulting in considerable adverse side effects. Another challenge of nanoparticle drug delivery includes the limited penetration depth of particles into the tumors. While extensive efforts have been devoted for designing therapeutic nanoparticles, a new study - echoing the journey through the human body in Fantastic Voyage - represents the first example of coupling such drug nanocarriers with self-propelled nanoshuttles. The ability of synthetic nanomotors to carry 'cargo' has already been demonstrated; but not in connection to common drug-loaded particles. In a new study, researchers demonstrate that catalytic nanoshuttles can readily pickup common biocompatible and biodegradable drug-loaded particles and liposomes and transport them over predefined routes towards predetermined destination.
Off-targeting remains a key challenge of researchers working on nanoparticle drug delivery - the majority of intravenously administered therapeutic nanoparticles are also reaching normal tissues, resulting in considerable adverse side effects. Another challenge of nanoparticle drug delivery includes the limited penetration depth of particles into the tumors. While extensive efforts have been devoted for designing therapeutic nanoparticles, a new study - echoing the journey through the human body in Fantastic Voyage - represents the first example of coupling such drug nanocarriers with self-propelled nanoshuttles. The ability of synthetic nanomotors to carry 'cargo' has already been demonstrated; but not in connection to common drug-loaded particles. In a new study, researchers demonstrate that catalytic nanoshuttles can readily pickup common biocompatible and biodegradable drug-loaded particles and liposomes and transport them over predefined routes towards predetermined destination.
Dec 10th, 2010
 The use of minute particles as drug carriers for targeted therapy has been studied and discussed for more than 20 years. A selective accumulation of active substances in target tissues has been demonstrated for certain so-called nanocarrier systems that are administered bound to pharmaceutical drugs. Great expectations are placed on nanocarrier systems that can overcome natural barriers such as the blood-brain barrier (BBB) and transport the medication directly to the desired tissue and thus heal neurological diseases that were formerly incurable. The BBB represents the border between the circulating blood and the fluid in the central nervous system. It functions to protect the sensitive nerve cells from foreign substances and infections from the blood. Whether nanoparticles enter the central nervous system unintentionally and induce health problems is also being debated.
The use of minute particles as drug carriers for targeted therapy has been studied and discussed for more than 20 years. A selective accumulation of active substances in target tissues has been demonstrated for certain so-called nanocarrier systems that are administered bound to pharmaceutical drugs. Great expectations are placed on nanocarrier systems that can overcome natural barriers such as the blood-brain barrier (BBB) and transport the medication directly to the desired tissue and thus heal neurological diseases that were formerly incurable. The BBB represents the border between the circulating blood and the fluid in the central nervous system. It functions to protect the sensitive nerve cells from foreign substances and infections from the blood. Whether nanoparticles enter the central nervous system unintentionally and induce health problems is also being debated.
Dec 8th, 2010
 Nanotechnology researchers working on self-powered nanodevices - nanoscale systems that scavenge energy from their surrounding environment - have been experimenting with various power sources ranging from piezoelectric systems to sound. However, the most abundant energy available in biosystems is chemical and biochemical energy, such as glucose. Researchers in China have now reported a nanowire-based biofuel cell based on a single proton conductive polymer nanowire for converting chemical energy from biofluids into electricity, using glucose oxidase and laccase as catalyst. The output of this biofuel cell is sufficient to drive pH, glucose or photon sensors. The high output power, low cost and easy fabrication process, large-scale manufacturability, high 'on-chip' integrability and stability demonstrates its great potential for in vivo biosensing.
Nanotechnology researchers working on self-powered nanodevices - nanoscale systems that scavenge energy from their surrounding environment - have been experimenting with various power sources ranging from piezoelectric systems to sound. However, the most abundant energy available in biosystems is chemical and biochemical energy, such as glucose. Researchers in China have now reported a nanowire-based biofuel cell based on a single proton conductive polymer nanowire for converting chemical energy from biofluids into electricity, using glucose oxidase and laccase as catalyst. The output of this biofuel cell is sufficient to drive pH, glucose or photon sensors. The high output power, low cost and easy fabrication process, large-scale manufacturability, high 'on-chip' integrability and stability demonstrates its great potential for in vivo biosensing.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





