Showing Spotlights 65 - 72 of 240 in category All (newest first):
 A tattoo-like sensor system that sticks to the skin and can indirectly tell the body's hydration level could help prevent heat strokes. The research team behind this invention demonstrated a completely new, versatile strategy to integrate pH sensing elements with high quality, physically transferable PDMS freestanding nanosheets. These sensor stickers can adhere to the skin without any glue. The simple structure of this sensor combined with a thin film pH sensor offers a unique advantage in testing in situ heatstroke detection.
A tattoo-like sensor system that sticks to the skin and can indirectly tell the body's hydration level could help prevent heat strokes. The research team behind this invention demonstrated a completely new, versatile strategy to integrate pH sensing elements with high quality, physically transferable PDMS freestanding nanosheets. These sensor stickers can adhere to the skin without any glue. The simple structure of this sensor combined with a thin film pH sensor offers a unique advantage in testing in situ heatstroke detection.
Mar 19th, 2020
 Researchers have demonstrated the first large-scale flexible thermal flow sensor array on a thin-film. This flow sensor - based on a calorimetric sensing mechanism - can not only monitor flow intensity but also flow direction. More importantly, these sensors can be attached onto curved surfaces for real-time flow monitoring. Flow separation is a common phenomenon affecting aircraft, wind turbine blades, micro aerials, and underwater vehicles. Owing to its unsteady flow profile, such separation is usually undesirable because either the airfoil efficiency decreases or large pressure fluctuations emerge. Consequently, precise monitoring is crucial to reduce or even prevent the flow separation effect.
Researchers have demonstrated the first large-scale flexible thermal flow sensor array on a thin-film. This flow sensor - based on a calorimetric sensing mechanism - can not only monitor flow intensity but also flow direction. More importantly, these sensors can be attached onto curved surfaces for real-time flow monitoring. Flow separation is a common phenomenon affecting aircraft, wind turbine blades, micro aerials, and underwater vehicles. Owing to its unsteady flow profile, such separation is usually undesirable because either the airfoil efficiency decreases or large pressure fluctuations emerge. Consequently, precise monitoring is crucial to reduce or even prevent the flow separation effect.
Nov 26th, 2019
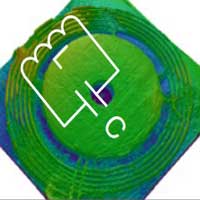 Radio-frequency wireless sensors are essential components of smart objects and internet-of-things components. However, these passive microsensors suffer from poor quality of data and sensitivity due to the environments they operate in and the need for sensors with extremely small footprints. Researchers have recently shown that a wireless system locked to an exceptional point (EP) can enhance the sensitivity of passive wireless sensors in practical applications. New work has theoretically introduced and experimentally demonstrated a new class of parity-time-symmetric RF electronic and telemetric systems, which combine EPs with divergent points.
Radio-frequency wireless sensors are essential components of smart objects and internet-of-things components. However, these passive microsensors suffer from poor quality of data and sensitivity due to the environments they operate in and the need for sensors with extremely small footprints. Researchers have recently shown that a wireless system locked to an exceptional point (EP) can enhance the sensitivity of passive wireless sensors in practical applications. New work has theoretically introduced and experimentally demonstrated a new class of parity-time-symmetric RF electronic and telemetric systems, which combine EPs with divergent points.
Nov 12th, 2019
 Researchers have developed a method for direct, point-of-use detection of mycotoxins in food. The method is based on the modification of electrolyte operated graphene filed effect transistors that have been specifically functionalized with aptamers by covalent binding. Demonstrating the performance of this method on Ochratoxin A, these sensors show a response time of within 5 minutes with a sensitivity down to 4 pg/ml - that is about three orders of magnitude less than accepted tolerance levels of Ochratoxin A.
Researchers have developed a method for direct, point-of-use detection of mycotoxins in food. The method is based on the modification of electrolyte operated graphene filed effect transistors that have been specifically functionalized with aptamers by covalent binding. Demonstrating the performance of this method on Ochratoxin A, these sensors show a response time of within 5 minutes with a sensitivity down to 4 pg/ml - that is about three orders of magnitude less than accepted tolerance levels of Ochratoxin A.
Oct 4th, 2019
 Researchers demonstrate the feasibility of an environmental moisture-induced, self-powered wearable multifunctional sensing system with the ability to detect both humidity and pressure. The self-powered wearable sensing system is fabricated by integrating a flexible micropatterned pressure-sensitive film on PET as the pressure sensor and attached it onto the back of a PDA power generator. Test results prove that this moisture-powered sensor can stably and sensitively realize real-time human physiological signal detection.
Researchers demonstrate the feasibility of an environmental moisture-induced, self-powered wearable multifunctional sensing system with the ability to detect both humidity and pressure. The self-powered wearable sensing system is fabricated by integrating a flexible micropatterned pressure-sensitive film on PET as the pressure sensor and attached it onto the back of a PDA power generator. Test results prove that this moisture-powered sensor can stably and sensitively realize real-time human physiological signal detection.
Aug 12th, 2019
 Due to their fascinating optoelectronic properties, halide perovskites have attracted tremendous research interest as promising materials for photovoltaics, photodetectors, LEDs, and lasers. Researchers report report a strategy to synthesize perovskite nanocrystals using diblock copolymer micelles as a growth template. This work constitutes a new approach for synthesizing perovskite nanocrystals of controllable size and composition with vastly improved resistance to halide ion migration and environmentally induced degradation.
Due to their fascinating optoelectronic properties, halide perovskites have attracted tremendous research interest as promising materials for photovoltaics, photodetectors, LEDs, and lasers. Researchers report report a strategy to synthesize perovskite nanocrystals using diblock copolymer micelles as a growth template. This work constitutes a new approach for synthesizing perovskite nanocrystals of controllable size and composition with vastly improved resistance to halide ion migration and environmentally induced degradation.
Aug 7th, 2019
 Electronic skin (e-skin) technology will give prosthetics and soft robotics a finer sense of touch, of what's hard and soft, hot and cold, smooth and rough. Researchers have now demonstrated a soft robotic hand integrated with four tactile force sensors and one temperature sensor to give it similar sensitivity to a human hand. By integrating an e-skin, the tactile pressure to grab an object and the friction movement of an object from the hand can be monitored. Thanks to the arrangement of multiple tactile sensors, e-skin-integrated soft robotic hand also can monitor sliding of an object by detecting the time delay of the tactile force.
Electronic skin (e-skin) technology will give prosthetics and soft robotics a finer sense of touch, of what's hard and soft, hot and cold, smooth and rough. Researchers have now demonstrated a soft robotic hand integrated with four tactile force sensors and one temperature sensor to give it similar sensitivity to a human hand. By integrating an e-skin, the tactile pressure to grab an object and the friction movement of an object from the hand can be monitored. Thanks to the arrangement of multiple tactile sensors, e-skin-integrated soft robotic hand also can monitor sliding of an object by detecting the time delay of the tactile force.
Jun 20th, 2019
 Recently, a brand-new mechanism of pressure sensing, known as flexible iontronic sensing (FITS), has been introduced, which utilizes pressure-induced capacitive changes between electrodes and ionic surfaces. Researchers have used FITS to develop a single-sheet iontronic paper substrate with both ionic and conductive patterns as an all-in-one flexible sensing platform. This novel paper sensor extends the iontronic sensing principle to a more adaptive material system, with direct printability, custom cuttability, and 3D foldability at a low cost - just as regular paper.
Recently, a brand-new mechanism of pressure sensing, known as flexible iontronic sensing (FITS), has been introduced, which utilizes pressure-induced capacitive changes between electrodes and ionic surfaces. Researchers have used FITS to develop a single-sheet iontronic paper substrate with both ionic and conductive patterns as an all-in-one flexible sensing platform. This novel paper sensor extends the iontronic sensing principle to a more adaptive material system, with direct printability, custom cuttability, and 3D foldability at a low cost - just as regular paper.
Mar 27th, 2019
 A tattoo-like sensor system that sticks to the skin and can indirectly tell the body's hydration level could help prevent heat strokes. The research team behind this invention demonstrated a completely new, versatile strategy to integrate pH sensing elements with high quality, physically transferable PDMS freestanding nanosheets. These sensor stickers can adhere to the skin without any glue. The simple structure of this sensor combined with a thin film pH sensor offers a unique advantage in testing in situ heatstroke detection.
A tattoo-like sensor system that sticks to the skin and can indirectly tell the body's hydration level could help prevent heat strokes. The research team behind this invention demonstrated a completely new, versatile strategy to integrate pH sensing elements with high quality, physically transferable PDMS freestanding nanosheets. These sensor stickers can adhere to the skin without any glue. The simple structure of this sensor combined with a thin film pH sensor offers a unique advantage in testing in situ heatstroke detection.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





