| Posted: Oct 27, 2011 | |
Lipid monolayers herald a new class of ultrathin dielectric |
|
| (Nanowerk Spotlight) Lipids are the main component of the outermost membrane of cells. Their role is to seperate the inner and outer media of the cell and prevent any ionic current between these two media. Because of this last property, lipid layers can be thought of as good ultra-thin insulators that could be used in the development of electronic devices. So far though, because of their inherent instability in air, their use in advanced processes has been limited. This might change, though, since in 2010 researchers in France have shown the possibility to stabilize by polymerization a lipid monolayer with a thickness of 2.7 nm directly at the surface of H-terminated silicon surface therefore opening a whole new world of possibilities of the use of these layers ("Direct Stabilization of a Phospholipid Monolayer on H-Terminated Silicon"). | |
| Now, a team from the Centre Interdisciplinaire de Nanoscience de Marseille (CINaM) has reported, for the first time, the electrical performance of stabilized lipid monolayers on H-terminated silicon. Writing in the October 3, 2011 online edition of Langmuir, a team led by Dr. Anne Charrier, a CNRS researcher, reports the reliable and reproducible electrical performances of phospholipid layers as novel dielectric layer ("Autonomic Self-Healing Lipid Monolayer: A New Class of Ultrathin Dielectric"). | |
| These promising results suggest that lipid layers will constitute a new class of ultrathin dielectric to be used in electrical devices. | |
| "We have studied the electrical properties of these monolayers and showed that even though they are ultra-thin, they have very good insulating properties with densities of leakage current about a hundred times smaller than that of a silicon oxide layer of similar thickness and with a dielectric breakdown of typically 10 MV/cm as high as that of inorganic insulators," Charrier explains to Nanowerk. "These results are very interesting, since they show the impact of the polymerization on the electrical stability of these monolayers as breakdown values around 2 MV/cm were previously reported on non-polymerized lipid bilayers." | |
 |
|
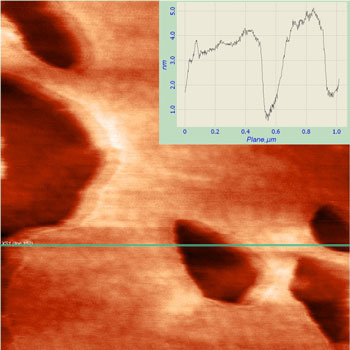
| AFM image of a lipid monolayer with holes to show the thickness (image size 1x1 µm). (Image: Dr. Charrier, CINaM) | |
| However, the major finding of Charrier and her team concerns the autonomic self-healing of the monolayer after electrical breakdown. After the breakdown, the monolayer is damaged and becomes conductive. Usually this kind of event is irreversible, meaning that your device will be dead if you consider an electronic device such as a field effect transistor for example. | |
| In some cases however, especially for polymers, the material can be fixed by the application of an external input such as heating the material or exposure to UV to allow for new polymerization, addition of material or of polymerizing agent. | |
| "In our case, the healing of the lipid layer is autonomic" says Charrier. "The device simply needs to rest at room temperature for a short amount of time – one hour or even less. Moreover, submitting the same layer to several successive breakdowns always led to the recovery of the layer. We are still investigating the process involved, but we believe this is due to the nature of the polymerized layer which is stabilized due to polymerization and still allowed to move and fluctuate on the surface as each molecule only has a maximum of two bonds with other molecules in the plane of the layer. This autonomic self-healing is a very unique feature of the lipids" | |
| One of the major interests in using such ultra-thin layer as a dielectric in electronic devices is to allow low operating voltages, therefore reducing the heating problem encountered in highly dense electronic devices as well as allowing working with biomolecules. Indeed these have bonds with ionization energies ranging from 3 eV to 10 eV which do not stand high energy electrons. | |
| Similarly, with the development of organic electronics that cannot handle the high temperatures required for making inorganic oxide dielectrics – typically to make a SiO2 layers, one needs to heat the device to 1000°C – low temperature processes need to be developed and the use of lipid monolayers could be an option. | |
| While this kind of lipid monolayer can already be used for the development of biosensors, one of the challenges for implementing this new class of ultrathin dielectric into microelectronic processes is to find a way to connect the lipid layer with a contact. | |
| "Once again, these layers are only 2.7 nm thick, the actual processes which usually involve metal evaporation are not suitable for this type of layers as they get damaged over the process," says Charrier. "If we could manage to connect these thin layers it will certainly be a breakthrough for this kind of technology." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
