| Jul 27, 2020 | |
A low-temperature bubble-generating system for enhanced surface capture of proteins |
|
| (Nanowerk Spotlight) A large variety of measurements in nanotechnology and the life sciences requires analytes in solution to react with receptors immobilized on a surface. The diffusional transport of biomolecules to a substrate is one of the factors that determines sensing performance of this kind of surface-based biosensing. | |
| In the quest to miniaturize sensors and analyte volumes – ultimately down to the nanoscale – as well as increase speed and sensitivity of the results, researchers have explored and developed a variety of platforms. One of them is optothermal microbubble assisted bio-sensing. | |
| Convective fluid flow generated by an optically controlled surface microbubble is a fascinating phenomenon for overcoming the diffusion limit in surface-based biosensing. The irradiation of a plasmonic substrate with a focused laser beam at the plasmon resonance wavelength generates microbubbles at the substrate-solution interface (read more: "Nanotechnology in a bubble"). These microbubbles can quickly accumulate solutes at the bubble-liquid-substrate interface due to high-velocity fluid flows. | |
| However, the high temperature of above 100 °C required for producing water bubbles has limited this technique's sensing applications to only certain target biomolecules. For instance, it is not suitable for sensing proteins that are subject to thermal denaturation. | |
| Motivated by the question of how to reduce and modulate the working temperature of bubble-generating systems from a material perspective, a team of researchers at The University of Texas at Austin have demonstrated a proof-of-concept study that reduces the working temperature of bubble generation simply by introducing a volatile, water-immiscible liquid into an aqueous medium. | |
| They published their findings in Nano Letters ("Enhancing Surface Capture and Sensing of Proteins with Low-Power Optothermal Bubbles in a Biphasic Liquid"). | |
| "Here, we demonstrate a biphasic system that generates microbubbles at a low optical power/temperature, thus enabling its application to protein sensing," Youngsun Kim, a member of Yuebing Zheng's research group and the paper's first author, tells Nanowerk. "Specifically, we describe the capability of a bubble, generated from a perfluoropentane (PFP)-in-water biphasic liquid, as an in situ concentrator in surface-based biosensing." | |
| By introducing a biphasic liquid system wherein a volatile, water-immiscible liquid (PFP) is formulated into an aqueous medium, the researchers were able to generate bubbles at a much lower temperature of around 30 °C – compared to over 100 °C for water bubbles – not only making this sensing platform compatible with proteins but also enhancing the surface capture of proteins. | |
 |
|
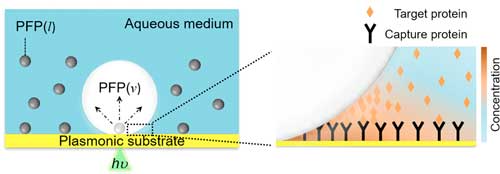
| Scheme of bubble-enhanced surface capture of proteins and description of biphasic fluid. Left: Schematic illustration of the bubble-generating perfluoropentane (PFP)-in-water system and, right: bubble-mediated concentration of target proteins near the bubble-liquid-substrate interface. Arrows in panel a indicate the expansion of the PFP droplet into the bubble. (Reprinted with permission by American Chemical Society) (click on image to enlarge) | |
| "We were able to reduce the threshold optical power for bubble generation to 33% of that in a pure aqueous medium," Kim points out. "The generated bubble was able to induce Marangoni flow due to a surface tension gradient, which was large enough to accumulate proteins from the bulk solution near the bubble-liquid-substrate interface. We observed one-order-of magnitude enhancement of surface capture within 1 minute in a single antigen-antibody model, compared to that from diffusion-limited static incubation for 30 minutes." | |
| One of the benefits of this concept is that the working temperature can be tuned simply by varying the components of the biphasic liquid, leading to its wide applicability in biosensing. | |
| "One of the merits of the bubble-based approach, composed of fluid formulation and an optothermal add-on, resides in its compatibility with conventional surface-based assay platforms," Kim notes. "Given the working mode of our concept, the same method can be sequentially applied to multiple steps of solution-to-surface conjugation as in sandwich-type ELISA, e.g., capture antibody/antigen, antigen/probe antibody, probe antibody/secondary antibody, and enzyme/substrate." | |
| Consequently, the team now plans to integrate the biphasic bubble-generating system into standard surface-based biosensing such as an enzyme-linked immunosorbent assay (ELISA). They expect a collective improvement of sensitivity and throughput performance resulting from enhancements in binding events at multiple steps of ELISA. | |
| Overall, the present study could find a wider range of scientific and clinical applications in biosensing when combined with rational designs of sensor configurations. It also suggests a way toward improving the performance of sensor and spectroscopy applications. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
