Showing Spotlights 1505 - 1512 of 2785 in category All (newest first):
 Of all the methods that have been developed to produce carbon nanotubes (CNTs), including arc discharge, laser ablation, and chemical vapor deposition (CVD), CVD is the most technically important and the most widely used in industry. Using the CVD process, manufacturers can combine a metal catalyst such as iron or nickel with reaction gases such as hydrocarbon to form carbon nanotubes inside a high-temperature furnace. Semiconductor nanoparticles such as silicon carbide, germanium and silicon have also been used for single CNT catalysis. However, these catalyst materials are usually expensive and need to be of high purity in order to be useful for the growth of carbon nanotubes. Researchers have now found that cheap and plentiful calcium silicate can absorb carbon species and grow multi-walled carbon nanotubes.
Of all the methods that have been developed to produce carbon nanotubes (CNTs), including arc discharge, laser ablation, and chemical vapor deposition (CVD), CVD is the most technically important and the most widely used in industry. Using the CVD process, manufacturers can combine a metal catalyst such as iron or nickel with reaction gases such as hydrocarbon to form carbon nanotubes inside a high-temperature furnace. Semiconductor nanoparticles such as silicon carbide, germanium and silicon have also been used for single CNT catalysis. However, these catalyst materials are usually expensive and need to be of high purity in order to be useful for the growth of carbon nanotubes. Researchers have now found that cheap and plentiful calcium silicate can absorb carbon species and grow multi-walled carbon nanotubes.
Apr 2nd, 2012
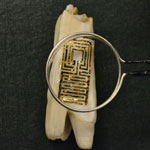 Early detection of pathogenic bacteria is critical to prevent disease outbreaks and preserve public health. This has led to urgent demands to develop highly efficient strategies for isolating and detecting this microorganism in connection to food safety, medical diagnostics, water quality, and counter-terrorism. A team of scientists has now developed a novel approach to interfacing passive, wireless graphene nanosensors onto biomaterials via silk bioresorption. The nanoscale nature of graphene allows for high adhesive conformality after biotransfer and highly sensitive detection. The team demonstrates their nanosensor by attaching it to a tooth for battery-free, remote monitoring of respiration and bacteria detection in saliva.
Early detection of pathogenic bacteria is critical to prevent disease outbreaks and preserve public health. This has led to urgent demands to develop highly efficient strategies for isolating and detecting this microorganism in connection to food safety, medical diagnostics, water quality, and counter-terrorism. A team of scientists has now developed a novel approach to interfacing passive, wireless graphene nanosensors onto biomaterials via silk bioresorption. The nanoscale nature of graphene allows for high adhesive conformality after biotransfer and highly sensitive detection. The team demonstrates their nanosensor by attaching it to a tooth for battery-free, remote monitoring of respiration and bacteria detection in saliva.
Mar 29th, 2012
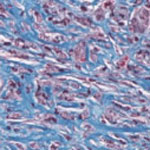 Wound healing is an exceedingly complex process, involving a multitude of signaling pathways, effector molecules, response phases, as well as a moderated balance between all these components. Nitric oxide (NO) plays a critical role in the wound-healing process via antimicrobial properties, modulation of platelet/cytokine function, vasodilatory effects, and promotion of angiogenesis and matrix deposition. While attempts to administer NO to wound areas have shown some promise, the current modalities all suffer from varying drawbacks, such as administration site irritation or the burden of large, expensive equipment. Researchers have now introduced a nanoparticle platform comprised of silane based sol-gel and sugar-derived glasses that can generate, store, and deliver NO in a controlled and sustained manner is utilized to enhance wound healing in immunodeficient mice.
Wound healing is an exceedingly complex process, involving a multitude of signaling pathways, effector molecules, response phases, as well as a moderated balance between all these components. Nitric oxide (NO) plays a critical role in the wound-healing process via antimicrobial properties, modulation of platelet/cytokine function, vasodilatory effects, and promotion of angiogenesis and matrix deposition. While attempts to administer NO to wound areas have shown some promise, the current modalities all suffer from varying drawbacks, such as administration site irritation or the burden of large, expensive equipment. Researchers have now introduced a nanoparticle platform comprised of silane based sol-gel and sugar-derived glasses that can generate, store, and deliver NO in a controlled and sustained manner is utilized to enhance wound healing in immunodeficient mice.
Mar 28th, 2012
 Optical fibers have revolutionized telecommunications by providing higher performance, more reliable telecommunication links with ever decreasing bandwidth cost. In parallel with these developments, fiber-optic sensor technology has been a major user of technologies associated with the optoelectronic and fiber optic communications industry. Today, with the rapid advance of communications and especially sensing applications, there is an ever increasing need for advanced performance and additional functionalities. This, however, is difficult to achieve without addressing fundamental fabrication issues related to the integration onto optical fibers of advanced functional materials at the micro- and nanoscale. Solving these technical problems will open up the possibility of developing multifunctional labs integrated onto a single optical fiber, exchanging information and combining sensorial data. This could result in auto diagnostic features as well as new photonic and electro-optic functionalities useful in many strategic sectors such as optical processing, environment, life science, safety and security.
Optical fibers have revolutionized telecommunications by providing higher performance, more reliable telecommunication links with ever decreasing bandwidth cost. In parallel with these developments, fiber-optic sensor technology has been a major user of technologies associated with the optoelectronic and fiber optic communications industry. Today, with the rapid advance of communications and especially sensing applications, there is an ever increasing need for advanced performance and additional functionalities. This, however, is difficult to achieve without addressing fundamental fabrication issues related to the integration onto optical fibers of advanced functional materials at the micro- and nanoscale. Solving these technical problems will open up the possibility of developing multifunctional labs integrated onto a single optical fiber, exchanging information and combining sensorial data. This could result in auto diagnostic features as well as new photonic and electro-optic functionalities useful in many strategic sectors such as optical processing, environment, life science, safety and security.
Mar 27th, 2012
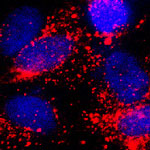 Nanotechnology offers new strategies to enable minimally invasive and localized approaches for diagnosing and treating cancer, thereby avoiding the serious side effects and shortcomings of chemotherapy. For instance, it has been shown that often less than 1% of the administered drug molecules during chemotherapy enter tumor cells and bind to the nuclear DNA. Another complication is drug resistance of cancer cells. This actually is one of the main causes of failure in the treatment of cancer. Cancer researchers are looking to nanoparticles as a drug carrier capable of localizing and directly releasing drugs into the cell nucleus, leading to a high therapeutic efficacy. Although increased therapeutic efficacy has been realized, there have been no reports on visualizing at nanoscale dimensions how nanoparticles interact with specific organelles. In a new breakthrough for nanomedicine cancer research, scientists have now reported the direct visualization of interactions between drug-loaded nanoparticles and the nucleus of a cancer cell.
Nanotechnology offers new strategies to enable minimally invasive and localized approaches for diagnosing and treating cancer, thereby avoiding the serious side effects and shortcomings of chemotherapy. For instance, it has been shown that often less than 1% of the administered drug molecules during chemotherapy enter tumor cells and bind to the nuclear DNA. Another complication is drug resistance of cancer cells. This actually is one of the main causes of failure in the treatment of cancer. Cancer researchers are looking to nanoparticles as a drug carrier capable of localizing and directly releasing drugs into the cell nucleus, leading to a high therapeutic efficacy. Although increased therapeutic efficacy has been realized, there have been no reports on visualizing at nanoscale dimensions how nanoparticles interact with specific organelles. In a new breakthrough for nanomedicine cancer research, scientists have now reported the direct visualization of interactions between drug-loaded nanoparticles and the nucleus of a cancer cell.
Mar 26th, 2012
 The fascination with two-dimensional (2D) materials that has started with graphene has spurred researchers to look for other 2D structures like for instance metal carbides and nitrides. One particularly interesting analogue to graphene would be 2D silicon - silicene - because it could be synthesized and processed using mature semiconductor techniques, and more easily integrated into existing electronics than graphene is currently. However, silicene does not seem to exist in nature nor is there any solid phase of silicon similar to graphite. Nevertheless, silicene has been predicted by theory as early as 1994. Researchers have now presented the first clear evidence for the synthesis and thus the existence of silicene - a two-dimensional material, with a honeycomb-like arrangement of silicon atoms.
The fascination with two-dimensional (2D) materials that has started with graphene has spurred researchers to look for other 2D structures like for instance metal carbides and nitrides. One particularly interesting analogue to graphene would be 2D silicon - silicene - because it could be synthesized and processed using mature semiconductor techniques, and more easily integrated into existing electronics than graphene is currently. However, silicene does not seem to exist in nature nor is there any solid phase of silicon similar to graphite. Nevertheless, silicene has been predicted by theory as early as 1994. Researchers have now presented the first clear evidence for the synthesis and thus the existence of silicene - a two-dimensional material, with a honeycomb-like arrangement of silicon atoms.
Mar 23rd, 2012
 One of the greatest challenges in harnessing the power of nanotechnology is achieving dynamic control of mechanical, electronic, optical and chemical properties of nanoscale structures and devices. Dynamic control can be achieved through the use of piezoelectric materials. These are materials where applying a mechanical strain results in an electrical voltage or conversely, application of an electric field induces mechanical deformation. While piezoelectricity has mainly been shown for 3D bulk materials, the piezoelectric effect has also been demonstrated and exploited at the nanoscale. Researchers have now demonstrated through density functional theory calculations that piezoelectricity can be engineered into non-piezoelectric graphene by selective surface adsorption of atoms on only one side, which breaks inversion symmetry.
One of the greatest challenges in harnessing the power of nanotechnology is achieving dynamic control of mechanical, electronic, optical and chemical properties of nanoscale structures and devices. Dynamic control can be achieved through the use of piezoelectric materials. These are materials where applying a mechanical strain results in an electrical voltage or conversely, application of an electric field induces mechanical deformation. While piezoelectricity has mainly been shown for 3D bulk materials, the piezoelectric effect has also been demonstrated and exploited at the nanoscale. Researchers have now demonstrated through density functional theory calculations that piezoelectricity can be engineered into non-piezoelectric graphene by selective surface adsorption of atoms on only one side, which breaks inversion symmetry.
Mar 22nd, 2012
 Various types of nanostructures are used in the development of nanosensors: nanoparticles, nanotubes, nanorods, two-dimensional materials like graphene, embedded nanostructures, porous silicon, and self-assembled materials. For instance, gas sensors often operate by detecting the subtle changes that deposited gas molecules make in the way electricity moves through a surface layer. Researchers have fabricated gas sensors based on carbon nanotube-based field effect transistors, which can detect electrical potential changes around them. While these and related sensing schemes can be all-electronic - i.e., not requiring optical readout - they all require sophisticated nanolithographic techniques to isolate, identify, and integrate electrical contact to the active nanosensor. Researchers have now presented a nanoscale 3D architecture that can afford highly sensitive, room temperature, rapid response, and all-electronic chemical detection.
Various types of nanostructures are used in the development of nanosensors: nanoparticles, nanotubes, nanorods, two-dimensional materials like graphene, embedded nanostructures, porous silicon, and self-assembled materials. For instance, gas sensors often operate by detecting the subtle changes that deposited gas molecules make in the way electricity moves through a surface layer. Researchers have fabricated gas sensors based on carbon nanotube-based field effect transistors, which can detect electrical potential changes around them. While these and related sensing schemes can be all-electronic - i.e., not requiring optical readout - they all require sophisticated nanolithographic techniques to isolate, identify, and integrate electrical contact to the active nanosensor. Researchers have now presented a nanoscale 3D architecture that can afford highly sensitive, room temperature, rapid response, and all-electronic chemical detection.
Mar 21st, 2012
 Of all the methods that have been developed to produce carbon nanotubes (CNTs), including arc discharge, laser ablation, and chemical vapor deposition (CVD), CVD is the most technically important and the most widely used in industry. Using the CVD process, manufacturers can combine a metal catalyst such as iron or nickel with reaction gases such as hydrocarbon to form carbon nanotubes inside a high-temperature furnace. Semiconductor nanoparticles such as silicon carbide, germanium and silicon have also been used for single CNT catalysis. However, these catalyst materials are usually expensive and need to be of high purity in order to be useful for the growth of carbon nanotubes. Researchers have now found that cheap and plentiful calcium silicate can absorb carbon species and grow multi-walled carbon nanotubes.
Of all the methods that have been developed to produce carbon nanotubes (CNTs), including arc discharge, laser ablation, and chemical vapor deposition (CVD), CVD is the most technically important and the most widely used in industry. Using the CVD process, manufacturers can combine a metal catalyst such as iron or nickel with reaction gases such as hydrocarbon to form carbon nanotubes inside a high-temperature furnace. Semiconductor nanoparticles such as silicon carbide, germanium and silicon have also been used for single CNT catalysis. However, these catalyst materials are usually expensive and need to be of high purity in order to be useful for the growth of carbon nanotubes. Researchers have now found that cheap and plentiful calcium silicate can absorb carbon species and grow multi-walled carbon nanotubes. 
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





