Showing Spotlights 201 - 208 of 315 in category All (newest first):
 The use of spontaneous self-assembly as a lithography- and external field-free means to construct well-ordered, often intriguing structures has received much attention for its ease of organizing materials on the nanoscale into ordered structures and producing complex, large-scale structures with small feature sizes. These self-organized structures promise new opportunities for developing miniaturized optical, electronic, optoelectronic, and magnetic devices. An extremely simple route to intriguing structures is the evaporation-induced self-assembly of polymers and nanoparticles from a droplet on a solid substrate. However, flow instabilities within the evaporating droplet often result in non-equilibrium and irregular dissipative structures, e.g., randomly organized convection patterns, stochastically distributed multi-rings, etc. Therefore, fully utilizing evaporation as a simple tool for creating well-ordered structures with numerous technological applications requires precise control over several factors, including evaporative flux, solution concentration, and the interfacial interaction between solute and substrate.
The use of spontaneous self-assembly as a lithography- and external field-free means to construct well-ordered, often intriguing structures has received much attention for its ease of organizing materials on the nanoscale into ordered structures and producing complex, large-scale structures with small feature sizes. These self-organized structures promise new opportunities for developing miniaturized optical, electronic, optoelectronic, and magnetic devices. An extremely simple route to intriguing structures is the evaporation-induced self-assembly of polymers and nanoparticles from a droplet on a solid substrate. However, flow instabilities within the evaporating droplet often result in non-equilibrium and irregular dissipative structures, e.g., randomly organized convection patterns, stochastically distributed multi-rings, etc. Therefore, fully utilizing evaporation as a simple tool for creating well-ordered structures with numerous technological applications requires precise control over several factors, including evaporative flux, solution concentration, and the interfacial interaction between solute and substrate.
Jul 16th, 2008
 Electrometers are instruments that measure electric charge or electrical potential difference by means of electrostatic force. While early electrometers such as developed by Lord Kelvin in the 19th century were crude instruments, modern electrometers based on solid state technology are high-precision electronic devices that, in extreme cases, are so sensitive they can count individual electrons as they pass through a circuit. As the dimensions of electronic devices shrink further, the probes required to measure the voltage inside a miniature conductor have to be miniaturized, too. An alligator clip cannot be scaled down indefinitely to perform such tasks. Furthermore, as devices reach the nanoscale, the perturbation of the measurement on the device itself cannot be neglected and must be assessed. A few techniques, many of which are based on scanning a small object such as an atomic force microscope (AFM) tip, have been developed in the past to address this challenge. Each technique has its pros and cons.
Electrometers are instruments that measure electric charge or electrical potential difference by means of electrostatic force. While early electrometers such as developed by Lord Kelvin in the 19th century were crude instruments, modern electrometers based on solid state technology are high-precision electronic devices that, in extreme cases, are so sensitive they can count individual electrons as they pass through a circuit. As the dimensions of electronic devices shrink further, the probes required to measure the voltage inside a miniature conductor have to be miniaturized, too. An alligator clip cannot be scaled down indefinitely to perform such tasks. Furthermore, as devices reach the nanoscale, the perturbation of the measurement on the device itself cannot be neglected and must be assessed. A few techniques, many of which are based on scanning a small object such as an atomic force microscope (AFM) tip, have been developed in the past to address this challenge. Each technique has its pros and cons.
Jul 14th, 2008
 Various techniques are being developed to enhance the already impressive properties of carbon nanotubes (CNTs) further by combining them with other materials. We have covered plenty of examples in our Spotlights. For instance, encapsulating carbon nanofibers with CNTs transforms cheap commercial carbon nanotubes into highly efficient carbon for electrochemical energy storage applications. Another study demonstrated that the redox properties of iron and iron oxide particles are tunable via encapsulation within CNTs, suggesting that a host-guest interaction between the confined metal particles and CNTs, which is different from that on the outside of the nanotubes. Researchers are still busy trying to understand some of the CNT basics, for instance something as fundamental as 'how do nanotubes grow'? How can their various properties - electronic, transport, or mechanical - be modified? Or how can you make use of CNT's structure and properties to build novel nanotools. A new model demonstrates that sufficiently small liquid metal droplets can be drawn inside a CNT via capillary action.
Various techniques are being developed to enhance the already impressive properties of carbon nanotubes (CNTs) further by combining them with other materials. We have covered plenty of examples in our Spotlights. For instance, encapsulating carbon nanofibers with CNTs transforms cheap commercial carbon nanotubes into highly efficient carbon for electrochemical energy storage applications. Another study demonstrated that the redox properties of iron and iron oxide particles are tunable via encapsulation within CNTs, suggesting that a host-guest interaction between the confined metal particles and CNTs, which is different from that on the outside of the nanotubes. Researchers are still busy trying to understand some of the CNT basics, for instance something as fundamental as 'how do nanotubes grow'? How can their various properties - electronic, transport, or mechanical - be modified? Or how can you make use of CNT's structure and properties to build novel nanotools. A new model demonstrates that sufficiently small liquid metal droplets can be drawn inside a CNT via capillary action.
Jul 11th, 2008
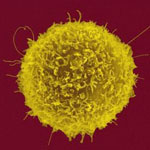 Scientists consider the exploration of carbon nanotubes (CNTs) for biomedical applications as a field with significant potential, leading to applications where CNTs could act as magnetic nano-heaters, drug-carrier systems and sensors which allow a diagnostic and therapeutic usage on a cellular level. The EU, for instance, has funded a four-year nanotechnology research program that studies the chemical and physical properties of CNTs in order to find mechanisms which can be applied for a biomedical purpose in appropriate medical devices. Studies of their interaction with biological environments (immune response, toxicity, interaction with the single cell) will provide the basis for applying the CNT for imaging (nanoparticles-based contrast agents), sensoring (nanoparticles-based diagnostics) and cancer treatment (hyperthermia, nanotechnology-based targeted drug delivery). A new study that exploits the unique properties of single-walled CNTs (SWCNT) for biomedical purposes shows the use of SWCNTs as an efficient platform for immunotherapeutic applications. Scientists demonstrate the surface area tunability of SWCNT bundles by chemical treatment and its effect on antibody adsorption and subsequent T cell activation. T cells are central players in initiating and maintaining immune responses. An important goal of successful immunotherapy is the stimulation of T cell immune responses against targets of interest such as tumors.
Scientists consider the exploration of carbon nanotubes (CNTs) for biomedical applications as a field with significant potential, leading to applications where CNTs could act as magnetic nano-heaters, drug-carrier systems and sensors which allow a diagnostic and therapeutic usage on a cellular level. The EU, for instance, has funded a four-year nanotechnology research program that studies the chemical and physical properties of CNTs in order to find mechanisms which can be applied for a biomedical purpose in appropriate medical devices. Studies of their interaction with biological environments (immune response, toxicity, interaction with the single cell) will provide the basis for applying the CNT for imaging (nanoparticles-based contrast agents), sensoring (nanoparticles-based diagnostics) and cancer treatment (hyperthermia, nanotechnology-based targeted drug delivery). A new study that exploits the unique properties of single-walled CNTs (SWCNT) for biomedical purposes shows the use of SWCNTs as an efficient platform for immunotherapeutic applications. Scientists demonstrate the surface area tunability of SWCNT bundles by chemical treatment and its effect on antibody adsorption and subsequent T cell activation. T cells are central players in initiating and maintaining immune responses. An important goal of successful immunotherapy is the stimulation of T cell immune responses against targets of interest such as tumors.
Jul 10th, 2008
 Cells are the basic building blocks of life. The ability to sense and modify intracellular processes is important for, among other things, bettering our understanding of biological processes, developing drugs and evaluating their effectiveness, and modifying cell function. Due to the cell's small size and fragility, probing the cell's interior with high precision is not a simple task. To address this challenge, researchers have developed nanoscale, carbon-based cellular probes ('carbon nanopipettes' or CNP). The CNP consists of a glass capillary lined with a carbon film along its inner surface and terminating with an exposed carbon nanopipe. The probes are fabricated through a process that does not require any assembly and that facilitates quantity fabrication. Depending on controllable process conditions, the carbon tip's diameter may vary from tens to hundreds of nanometers and its length can range from zero to a few micrometers.
Cells are the basic building blocks of life. The ability to sense and modify intracellular processes is important for, among other things, bettering our understanding of biological processes, developing drugs and evaluating their effectiveness, and modifying cell function. Due to the cell's small size and fragility, probing the cell's interior with high precision is not a simple task. To address this challenge, researchers have developed nanoscale, carbon-based cellular probes ('carbon nanopipettes' or CNP). The CNP consists of a glass capillary lined with a carbon film along its inner surface and terminating with an exposed carbon nanopipe. The probes are fabricated through a process that does not require any assembly and that facilitates quantity fabrication. Depending on controllable process conditions, the carbon tip's diameter may vary from tens to hundreds of nanometers and its length can range from zero to a few micrometers.
Jul 8th, 2008
 In recent years, great progress has been made in the synthesis and application studies of hybrid nanomaterial systems involving carbon nanotubes (CNTs). Efforts involve the alteration of physical properties of CNTs via the use of organic, inorganic, and biological species to produce functionalized CNTs for further applications. In one such hybrid system, aligned CNT templates serve as a natural 3D scaffold ('CNT forests'). Preferential assembly of nanoparticles onto targeted locations in this 3D scaffold creates novel hybrid nanomaterial systems with a unique architecture comprised of different functional components. For example, these CNT forests could serve as a template for controlled assembly of various semiconducting nanoparticles such as quantum dots. The resulting hybrid nanomaterial has the effect of changing both optical and electronic properties of the CNTs.
In recent years, great progress has been made in the synthesis and application studies of hybrid nanomaterial systems involving carbon nanotubes (CNTs). Efforts involve the alteration of physical properties of CNTs via the use of organic, inorganic, and biological species to produce functionalized CNTs for further applications. In one such hybrid system, aligned CNT templates serve as a natural 3D scaffold ('CNT forests'). Preferential assembly of nanoparticles onto targeted locations in this 3D scaffold creates novel hybrid nanomaterial systems with a unique architecture comprised of different functional components. For example, these CNT forests could serve as a template for controlled assembly of various semiconducting nanoparticles such as quantum dots. The resulting hybrid nanomaterial has the effect of changing both optical and electronic properties of the CNTs.
Jun 27th, 2008
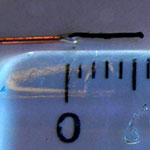 Carbon nanotubes (CNTs) have been widely used as electrodes for chemical and biological sensing applications and many other electrochemical studies. With their unique one-dimensional molecular geometry of a large surface area coupled with their excellent electrical properties, CNTs have become important materials for the molecular engineering of electrode surfaces where the development of electrochemical devices with region-specific electron-transfer capabilities is of paramount importance. It has been demonstrated that carbon nanotubes enhance the electrochemical activity of biomolecules and promote the electron-transfer reactions of redox proteins, such as myoglobin, cyctochrome c, and microperoxidase MP-11. The enhanced electrochemical activity and electron transfer rate at CNT electrodes have been widely believed to arise from the nanotube tips. However, no convincing experimental evidence has been obtained to prove that the CNT tip is more electrochemically active than its sidewall. Contradicting this common belief, researchers have now found that, surprisingly, the electrochemistry at carbon nanotube electrodes is not always facilitated by the nanotube tip. In fact, the relative electrochemical sensitivity of the nanotube tip and sidewall varies for different electrochemical probes proceeding with different reaction mechanisms.
Carbon nanotubes (CNTs) have been widely used as electrodes for chemical and biological sensing applications and many other electrochemical studies. With their unique one-dimensional molecular geometry of a large surface area coupled with their excellent electrical properties, CNTs have become important materials for the molecular engineering of electrode surfaces where the development of electrochemical devices with region-specific electron-transfer capabilities is of paramount importance. It has been demonstrated that carbon nanotubes enhance the electrochemical activity of biomolecules and promote the electron-transfer reactions of redox proteins, such as myoglobin, cyctochrome c, and microperoxidase MP-11. The enhanced electrochemical activity and electron transfer rate at CNT electrodes have been widely believed to arise from the nanotube tips. However, no convincing experimental evidence has been obtained to prove that the CNT tip is more electrochemically active than its sidewall. Contradicting this common belief, researchers have now found that, surprisingly, the electrochemistry at carbon nanotube electrodes is not always facilitated by the nanotube tip. In fact, the relative electrochemical sensitivity of the nanotube tip and sidewall varies for different electrochemical probes proceeding with different reaction mechanisms.
Jun 26th, 2008
 You might have seen our news item from a few days ago about BMW's shape shifting concept car. NASA has worked on something much more revolutionary, called the 'Morphing' program, for a few years already. The idea is that aircraft of the future will not be built of traditional, multiple, mechanically connected parts and systems. Instead, aircraft wing construction will employ fully-integrated, nanotechnology enabled embedded 'smart' materials and actuators that will enable aircraft wings with unprecedented levels of aerodynamic efficiencies and aircraft control. Able to respond to the constantly varying conditions of flight, sensors will act like the nerves in a bird's wing and will measure the pressure over the entire surface of the wing. The response to these measurements will direct actuators, which will function like the bird's wing muscles. Just as a bird instinctively uses different feathers on its wings to control its flight, the actuators will change the shape of the aircraft's wings to continually optimize flying conditions. Active flow control effectors will help mitigate adverse aircraft motions when turbulent air conditions are encountered.
You might have seen our news item from a few days ago about BMW's shape shifting concept car. NASA has worked on something much more revolutionary, called the 'Morphing' program, for a few years already. The idea is that aircraft of the future will not be built of traditional, multiple, mechanically connected parts and systems. Instead, aircraft wing construction will employ fully-integrated, nanotechnology enabled embedded 'smart' materials and actuators that will enable aircraft wings with unprecedented levels of aerodynamic efficiencies and aircraft control. Able to respond to the constantly varying conditions of flight, sensors will act like the nerves in a bird's wing and will measure the pressure over the entire surface of the wing. The response to these measurements will direct actuators, which will function like the bird's wing muscles. Just as a bird instinctively uses different feathers on its wings to control its flight, the actuators will change the shape of the aircraft's wings to continually optimize flying conditions. Active flow control effectors will help mitigate adverse aircraft motions when turbulent air conditions are encountered.
Jun 16th, 2008
 The use of spontaneous self-assembly as a lithography- and external field-free means to construct well-ordered, often intriguing structures has received much attention for its ease of organizing materials on the nanoscale into ordered structures and producing complex, large-scale structures with small feature sizes. These self-organized structures promise new opportunities for developing miniaturized optical, electronic, optoelectronic, and magnetic devices. An extremely simple route to intriguing structures is the evaporation-induced self-assembly of polymers and nanoparticles from a droplet on a solid substrate. However, flow instabilities within the evaporating droplet often result in non-equilibrium and irregular dissipative structures, e.g., randomly organized convection patterns, stochastically distributed multi-rings, etc. Therefore, fully utilizing evaporation as a simple tool for creating well-ordered structures with numerous technological applications requires precise control over several factors, including evaporative flux, solution concentration, and the interfacial interaction between solute and substrate.
The use of spontaneous self-assembly as a lithography- and external field-free means to construct well-ordered, often intriguing structures has received much attention for its ease of organizing materials on the nanoscale into ordered structures and producing complex, large-scale structures with small feature sizes. These self-organized structures promise new opportunities for developing miniaturized optical, electronic, optoelectronic, and magnetic devices. An extremely simple route to intriguing structures is the evaporation-induced self-assembly of polymers and nanoparticles from a droplet on a solid substrate. However, flow instabilities within the evaporating droplet often result in non-equilibrium and irregular dissipative structures, e.g., randomly organized convection patterns, stochastically distributed multi-rings, etc. Therefore, fully utilizing evaporation as a simple tool for creating well-ordered structures with numerous technological applications requires precise control over several factors, including evaporative flux, solution concentration, and the interfacial interaction between solute and substrate. 
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





