Showing Spotlights 33 - 40 of 200 in category All (newest first):
 The concept of nanostructuring of battery electrodes is not new; it has been frequently employed to improve the charging speed and, in some cases, stability of Li-ion electrodes. Researchers now demonstrated 3D-interconnected Ni nanowire meshes as current collectors and structural scaffolds for building nanostructured Li-ion electrodes. This material exhibits a semi-ordered structure and is characterized by some of the highest combined porosity and surface-to-volume ratio among macroporous metals.
The concept of nanostructuring of battery electrodes is not new; it has been frequently employed to improve the charging speed and, in some cases, stability of Li-ion electrodes. Researchers now demonstrated 3D-interconnected Ni nanowire meshes as current collectors and structural scaffolds for building nanostructured Li-ion electrodes. This material exhibits a semi-ordered structure and is characterized by some of the highest combined porosity and surface-to-volume ratio among macroporous metals.
Jul 6th, 2020
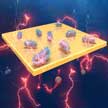 Researchers have identified the potential of using nanoscale carbon dots on bacteria for augmented bioelectricity production. They used this to demonstrate a novel method for significantly increasing the bacterial bioelectricity generation and applied it to set up microbial fuel cells. The results show that carbon dots' highly conductive carbon core not only improved overall systemic electrical conductivity but could also increase the packing density of the electron transfer path. This significantly improves the bioelectricity production of microbial fuel cells.
Researchers have identified the potential of using nanoscale carbon dots on bacteria for augmented bioelectricity production. They used this to demonstrate a novel method for significantly increasing the bacterial bioelectricity generation and applied it to set up microbial fuel cells. The results show that carbon dots' highly conductive carbon core not only improved overall systemic electrical conductivity but could also increase the packing density of the electron transfer path. This significantly improves the bioelectricity production of microbial fuel cells.
Mar 19th, 2020
 Oxygen reduction reaction (ORR), oxygen evolution reaction (OER), and hydrogen evolution reaction (HER) are among the core electrochemical processes in clean energy conversion and storage devices, such as metal-air batteries, water electrolyzers, and fuel cells. Single-atom catalysts have emerged as a new frontier of heterogeneous catalysts for these reactions due to their highly increased coverage of active sites, enhanced catalytic performance, and maximal metal utilization. By using machine learning algorithms, researchers provide a new paradigm for directly predicting the catalytic performance from physical properties of catalyst candidates.
Oxygen reduction reaction (ORR), oxygen evolution reaction (OER), and hydrogen evolution reaction (HER) are among the core electrochemical processes in clean energy conversion and storage devices, such as metal-air batteries, water electrolyzers, and fuel cells. Single-atom catalysts have emerged as a new frontier of heterogeneous catalysts for these reactions due to their highly increased coverage of active sites, enhanced catalytic performance, and maximal metal utilization. By using machine learning algorithms, researchers provide a new paradigm for directly predicting the catalytic performance from physical properties of catalyst candidates.
Feb 13th, 2020
 As an advanced fabrication technique, 3D printing has been increasingly utilized to fabricate complex 3D objects via digitally controlled deposition of phase change and reactive materials and solvent-based inks. When it comes to batteries, 3D printing has several significant advantages compared with conventional battery fabrication technologies and it opens new avenues for the rapid fabrication of 3D-structured batteries with complex architectures and high performance. In next generation futuristic 3D printed energy architectures batteries and supercapacitors could be printed in virtually any shape.
As an advanced fabrication technique, 3D printing has been increasingly utilized to fabricate complex 3D objects via digitally controlled deposition of phase change and reactive materials and solvent-based inks. When it comes to batteries, 3D printing has several significant advantages compared with conventional battery fabrication technologies and it opens new avenues for the rapid fabrication of 3D-structured batteries with complex architectures and high performance. In next generation futuristic 3D printed energy architectures batteries and supercapacitors could be printed in virtually any shape.
Nov 4th, 2019
 Ocean wave energy is an abundant resource of clean energy. However, having been explored for decades, there is still lacking efficient and economical technologies to convert water wave energy into electricity for large-scale applications Researchers have developed a new type of wave energy harvester, hierarchically structured triboelectric nanogenerator (HS-TENG), based on the triboelectric effect, targeting to harvest the huge blue energy for large-scale deployments.
Ocean wave energy is an abundant resource of clean energy. However, having been explored for decades, there is still lacking efficient and economical technologies to convert water wave energy into electricity for large-scale applications Researchers have developed a new type of wave energy harvester, hierarchically structured triboelectric nanogenerator (HS-TENG), based on the triboelectric effect, targeting to harvest the huge blue energy for large-scale deployments.
Oct 7th, 2019
 Researchers have developed a comprehensive approach involving simple and facile steps to fabricate a wearable energy storage device based on carbon nanotube coated cotton yarn. All device components are flexible. According to the team, this is the first device that has been proven to be stable under rigorous washing conditions in the presence of hot water, detergents and high torque (spinning action of washing machine). This provides the device with comprehensive mechanical stability.
Researchers have developed a comprehensive approach involving simple and facile steps to fabricate a wearable energy storage device based on carbon nanotube coated cotton yarn. All device components are flexible. According to the team, this is the first device that has been proven to be stable under rigorous washing conditions in the presence of hot water, detergents and high torque (spinning action of washing machine). This provides the device with comprehensive mechanical stability.
May 15th, 2019
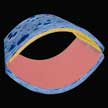 Researchers introduced an exclusive self-charging and self-healing flexible asymmetric supercapacitor power cell proficient to sustainably power-up portable electronics by simply applying mechanical bio-mechanical stresses. This self-charging power cell/package comprises an asymmetric supercapacitor unit sandwiched in-between two parallel connected triboelectric nanogenerators and reveals exceptional output performances. Additionally, this nanogenerator cell exhibits remarkable self-healing behavior.
Researchers introduced an exclusive self-charging and self-healing flexible asymmetric supercapacitor power cell proficient to sustainably power-up portable electronics by simply applying mechanical bio-mechanical stresses. This self-charging power cell/package comprises an asymmetric supercapacitor unit sandwiched in-between two parallel connected triboelectric nanogenerators and reveals exceptional output performances. Additionally, this nanogenerator cell exhibits remarkable self-healing behavior.
Apr 30th, 2019
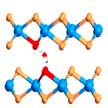 Aqueous rechargeable batteries based on earth abundant materials are promising alternatives to lithium-ion batteries, which are plagued by safety and environmental concerns. These batteries utilize water-based electrolytes, which are safe, low-cost, and environment-friendly, but also possess a much higher ionic conductivity than that of the organic electrolytes. In new work, researchers report an effective and general strategy to transform inactive intercalation hosts into efficient zinc ion storage materials through intercalation energy tuning.
Aqueous rechargeable batteries based on earth abundant materials are promising alternatives to lithium-ion batteries, which are plagued by safety and environmental concerns. These batteries utilize water-based electrolytes, which are safe, low-cost, and environment-friendly, but also possess a much higher ionic conductivity than that of the organic electrolytes. In new work, researchers report an effective and general strategy to transform inactive intercalation hosts into efficient zinc ion storage materials through intercalation energy tuning.
Apr 26th, 2019
 The concept of nanostructuring of battery electrodes is not new; it has been frequently employed to improve the charging speed and, in some cases, stability of Li-ion electrodes. Researchers now demonstrated 3D-interconnected Ni nanowire meshes as current collectors and structural scaffolds for building nanostructured Li-ion electrodes. This material exhibits a semi-ordered structure and is characterized by some of the highest combined porosity and surface-to-volume ratio among macroporous metals.
The concept of nanostructuring of battery electrodes is not new; it has been frequently employed to improve the charging speed and, in some cases, stability of Li-ion electrodes. Researchers now demonstrated 3D-interconnected Ni nanowire meshes as current collectors and structural scaffolds for building nanostructured Li-ion electrodes. This material exhibits a semi-ordered structure and is characterized by some of the highest combined porosity and surface-to-volume ratio among macroporous metals. 
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





