Showing Spotlights 57 - 64 of 544 in category All (newest first):
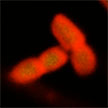 The antibiotic resistance crisis has been ascribed to the overuse and misuse of these medications, as well as a lack of motivation to develop new drugs. In the field of nanotechnology, a variety of innovative materials are being studied to evaluate their potential applications as antimicrobial agents. Recently, researchers have shown that boron nitride nanosheets as a nano-antibacterial agent displays antibiotic-like activities and BN nanosheets were found to show potent antibacterial efficiency in five multidrug resistant bacteria strains.
The antibiotic resistance crisis has been ascribed to the overuse and misuse of these medications, as well as a lack of motivation to develop new drugs. In the field of nanotechnology, a variety of innovative materials are being studied to evaluate their potential applications as antimicrobial agents. Recently, researchers have shown that boron nitride nanosheets as a nano-antibacterial agent displays antibiotic-like activities and BN nanosheets were found to show potent antibacterial efficiency in five multidrug resistant bacteria strains.
May 17th, 2022
 The two-dimensional carbon allotrope graphdiyne is capable of inhibiting broad-spectrum bacterial growth while exerting moderate cytotoxicity on mammalian cells. Researchers now demonstrate a high-performance bactericid with graphdiyne functionalized by silver nanoparticles. The material killed the bacteria through membrane destruction and reactive oxygen species production. These findings present an avenue to harness 2D materials to stabilize metal nanoparticles as a promising option for combating evolving bacteria.
The two-dimensional carbon allotrope graphdiyne is capable of inhibiting broad-spectrum bacterial growth while exerting moderate cytotoxicity on mammalian cells. Researchers now demonstrate a high-performance bactericid with graphdiyne functionalized by silver nanoparticles. The material killed the bacteria through membrane destruction and reactive oxygen species production. These findings present an avenue to harness 2D materials to stabilize metal nanoparticles as a promising option for combating evolving bacteria.
May 11th, 2022
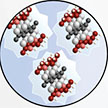 Researchers have have reported the synthesis process and biomedical applications of a novel nanomaterial for in vivo treatment of vasculopathy - a general term used to describe any disease affecting blood vessels. Through their innovative approach using rational design and synthesis strategies, the researchers developed intrinsically immunomodulatory and anti-inflammatory tantalum carbide MXene quantum dots. The team is confident that this new nanomaterial could reduce or even avoid the need for anti-rejection drugs for heart transplant patients.
Researchers have have reported the synthesis process and biomedical applications of a novel nanomaterial for in vivo treatment of vasculopathy - a general term used to describe any disease affecting blood vessels. Through their innovative approach using rational design and synthesis strategies, the researchers developed intrinsically immunomodulatory and anti-inflammatory tantalum carbide MXene quantum dots. The team is confident that this new nanomaterial could reduce or even avoid the need for anti-rejection drugs for heart transplant patients.
May 9th, 2022
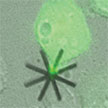 The mechanical properties of cells and their intracellular mechanical loads are as relevant for a cell's accurate functioning as the biochemical portion of the cell. This offers a novel perspective that addresses the study of cell mechanics and even the alteration of the mechanical equilibrium of the cells by using silicon chips as an intracellular intervention mechanism. These 'echanical drugs' open the possibility to determine and measure intracellular forces and internalization routes. They can also produce intracellular mechanical perturbations by altering the cell cycle and even, if they are designed accordingly, destroy the cells.
The mechanical properties of cells and their intracellular mechanical loads are as relevant for a cell's accurate functioning as the biochemical portion of the cell. This offers a novel perspective that addresses the study of cell mechanics and even the alteration of the mechanical equilibrium of the cells by using silicon chips as an intracellular intervention mechanism. These 'echanical drugs' open the possibility to determine and measure intracellular forces and internalization routes. They can also produce intracellular mechanical perturbations by altering the cell cycle and even, if they are designed accordingly, destroy the cells.
May 2nd, 2022
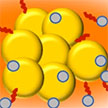 Researchers have developed a point-of-care, portable, rapid, and cost-effective test for clinical diagnosis of sodium, which uses a paper printed colorimetric sensor that is based on Curcumin-functionalized copper (CuC) nanoparticles. They demonstrate that the proposed CuC paper strip can be used for detecting Na+ concentration within the whole physiological range in both blood serum and urine. Sodium is a vitally important electrolyte present in all body fluids and it plays a crucial role in maintaining normal body function, including nerve and muscle function.
Researchers have developed a point-of-care, portable, rapid, and cost-effective test for clinical diagnosis of sodium, which uses a paper printed colorimetric sensor that is based on Curcumin-functionalized copper (CuC) nanoparticles. They demonstrate that the proposed CuC paper strip can be used for detecting Na+ concentration within the whole physiological range in both blood serum and urine. Sodium is a vitally important electrolyte present in all body fluids and it plays a crucial role in maintaining normal body function, including nerve and muscle function.
Apr 26th, 2022
 A recent review article highlights how nanoscience and nanotechnology can solve three acute problems in the treatment, prevention, and diagnosis of viruses like SARS-CoV-2: 1) synthesis of novel drugs and vaccines based on nanocarriers with enhanced efficiency and reduced side-effects for the treatment of the current pandemic and the prevention of future virus attacks; 2) design of protective equipment based on nanoparticles, such as medical face masks and outfits and 3) development of nanobiosensors for early detection of infections in a maximally efficient way.
A recent review article highlights how nanoscience and nanotechnology can solve three acute problems in the treatment, prevention, and diagnosis of viruses like SARS-CoV-2: 1) synthesis of novel drugs and vaccines based on nanocarriers with enhanced efficiency and reduced side-effects for the treatment of the current pandemic and the prevention of future virus attacks; 2) design of protective equipment based on nanoparticles, such as medical face masks and outfits and 3) development of nanobiosensors for early detection of infections in a maximally efficient way.
Apr 6th, 2022
 Since the beginning of the COVID-19 pandemic in 2020, researchers have intensified their efforts to improve the performance of antiviral and antibacterial face masks by adding various functionalities such as metal nanoparticles and herbal extracts to inactivate pathogens, using graphene to make masks photothermal and superhydrophobic, and even using triboelectric nanogenerators to prolong mask lifetime. Based on these works, manufacturers have developed different mask styles and air filtration materials using various nanomaterials.
Since the beginning of the COVID-19 pandemic in 2020, researchers have intensified their efforts to improve the performance of antiviral and antibacterial face masks by adding various functionalities such as metal nanoparticles and herbal extracts to inactivate pathogens, using graphene to make masks photothermal and superhydrophobic, and even using triboelectric nanogenerators to prolong mask lifetime. Based on these works, manufacturers have developed different mask styles and air filtration materials using various nanomaterials.
Mar 14th, 2022
 The design of chemical communication networks at the micro/nanoscale is an emergent interdisciplinary topic with potential applications in diverse areas such as sensing, biomedicine, biotechnology, and information and communication technologies. Previously, researchers have demonstrated communication between synthetic microvesicles and one type of cells. Communicating cells from different kingdoms has not been achieved before. Now, scientists report a proof-of-concept study that appears to be the first realization of a programmed cross-kingdom communication involving two species of living cells enabled by nanoparticles.
The design of chemical communication networks at the micro/nanoscale is an emergent interdisciplinary topic with potential applications in diverse areas such as sensing, biomedicine, biotechnology, and information and communication technologies. Previously, researchers have demonstrated communication between synthetic microvesicles and one type of cells. Communicating cells from different kingdoms has not been achieved before. Now, scientists report a proof-of-concept study that appears to be the first realization of a programmed cross-kingdom communication involving two species of living cells enabled by nanoparticles.
Feb 25th, 2022
 The antibiotic resistance crisis has been ascribed to the overuse and misuse of these medications, as well as a lack of motivation to develop new drugs. In the field of nanotechnology, a variety of innovative materials are being studied to evaluate their potential applications as antimicrobial agents. Recently, researchers have shown that boron nitride nanosheets as a nano-antibacterial agent displays antibiotic-like activities and BN nanosheets were found to show potent antibacterial efficiency in five multidrug resistant bacteria strains.
The antibiotic resistance crisis has been ascribed to the overuse and misuse of these medications, as well as a lack of motivation to develop new drugs. In the field of nanotechnology, a variety of innovative materials are being studied to evaluate their potential applications as antimicrobial agents. Recently, researchers have shown that boron nitride nanosheets as a nano-antibacterial agent displays antibiotic-like activities and BN nanosheets were found to show potent antibacterial efficiency in five multidrug resistant bacteria strains.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





