Showing Spotlights 105 - 112 of 203 in category All (newest first):
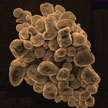 Although developed only recently, inorganic halide perovskite quantum dot systems have exhibited comparable and even better performances than traditional quantum dots in many fields. They are expected to be applied in display and lighting technologies. Researchers now have reported an interesting cyclable surface dissolution and recrystallization phenomenon of inorganic perovskite crystals. This allows them to freely change size between nanometer and micrometer scales, and can be used to healing the defects inside perovskite films and hence improve the performances of optoelectronic devices.
Although developed only recently, inorganic halide perovskite quantum dot systems have exhibited comparable and even better performances than traditional quantum dots in many fields. They are expected to be applied in display and lighting technologies. Researchers now have reported an interesting cyclable surface dissolution and recrystallization phenomenon of inorganic perovskite crystals. This allows them to freely change size between nanometer and micrometer scales, and can be used to healing the defects inside perovskite films and hence improve the performances of optoelectronic devices.
Jul 7th, 2016
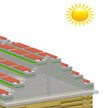 Self-powered nanotechnology based on these nanogenerators aims at powering nanodevices and nanosystems using the energy harvested from the environment in which these systems are suppose to operate. An interesting approach comes from a group of Chinese scientists who propose to scavenge the large amounts of wasted wind energy in cities: they propose hybridized nanogenerator that consists of a solar cell and a triboelectric nanogenerator, which can be utilized to individually or simultaneously scavenge solar and wind energies.
Self-powered nanotechnology based on these nanogenerators aims at powering nanodevices and nanosystems using the energy harvested from the environment in which these systems are suppose to operate. An interesting approach comes from a group of Chinese scientists who propose to scavenge the large amounts of wasted wind energy in cities: they propose hybridized nanogenerator that consists of a solar cell and a triboelectric nanogenerator, which can be utilized to individually or simultaneously scavenge solar and wind energies.
May 23rd, 2016
 Today, the best performing battery in terms of specific energy and specific power is the secondary lithium-metal (Li-metal). However, uncontrolled dendrite growth during Li depositing/stripping in rechargeable Li metal based batteries has prevented their practical applications over the past 40 years. To address this issue, researchers have now proposed a novel method of modulating the lithium ion adsorption to suppress lithium dendrite growth by employing glass fiber as solid electrolytes with plenty of polar functional groups as the interlayer between Li metal anode and routine polymer separator.
Today, the best performing battery in terms of specific energy and specific power is the secondary lithium-metal (Li-metal). However, uncontrolled dendrite growth during Li depositing/stripping in rechargeable Li metal based batteries has prevented their practical applications over the past 40 years. To address this issue, researchers have now proposed a novel method of modulating the lithium ion adsorption to suppress lithium dendrite growth by employing glass fiber as solid electrolytes with plenty of polar functional groups as the interlayer between Li metal anode and routine polymer separator.
Apr 19th, 2016
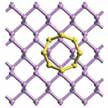 Lithium-sulfur (Li-S) batteries, which employ sulfur as cathode and metallic lithium as anode materials, have been extensively studied as promising alternatives to the widely used lithium-ion batteries because - theoretically - they can render 3-6 times higher energy density. In practice, though, it has proven challenging to approach that theoretical value. Specifically, the rapid capacity fading, low Coulombic efficiency, and irreversible loss of active materials have impeded large-scale commercial use of Li-S batteries. Researchers now have shown that trapping lithium polysulfide species on (nanoscale) host materials is an effective way to overcome these challenges.
Lithium-sulfur (Li-S) batteries, which employ sulfur as cathode and metallic lithium as anode materials, have been extensively studied as promising alternatives to the widely used lithium-ion batteries because - theoretically - they can render 3-6 times higher energy density. In practice, though, it has proven challenging to approach that theoretical value. Specifically, the rapid capacity fading, low Coulombic efficiency, and irreversible loss of active materials have impeded large-scale commercial use of Li-S batteries. Researchers now have shown that trapping lithium polysulfide species on (nanoscale) host materials is an effective way to overcome these challenges.
Apr 14th, 2016
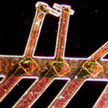 To explore the intrinsic mechanisms of the electrochemical reactions of porous graphene oxide in situ, the single-nanowire electrochemical probe is an effective tool. Although graphene is usually used as an additive in active materials to improve the electrochemical performance, how graphene influences the electrochemical performance and reaction mechanisms of electrode materials is under dispute. To address these issues, researchers have explored single-nanowire electrochemical devices to investigate the capacitance, ion diffusion coefficient, and charge storage mechanisms of graphene.
To explore the intrinsic mechanisms of the electrochemical reactions of porous graphene oxide in situ, the single-nanowire electrochemical probe is an effective tool. Although graphene is usually used as an additive in active materials to improve the electrochemical performance, how graphene influences the electrochemical performance and reaction mechanisms of electrode materials is under dispute. To address these issues, researchers have explored single-nanowire electrochemical devices to investigate the capacitance, ion diffusion coefficient, and charge storage mechanisms of graphene.
Mar 29th, 2016
 Lithium (Li) metal-based batteries such as Li-sulfur and Li-air batteries have received considerable attention because Li metal can store 10 times more energy than graphite. However, there are safety and performance concerns for these types of batteries that arise from the formation of dendrites on the electrodes. To address this issue, researchers have proposed an unstacked graphene nanostructured lithium metal anode for lithium metal batteries to inhibit lithium dendrite growth and bring superior electrochemistry performance.
Lithium (Li) metal-based batteries such as Li-sulfur and Li-air batteries have received considerable attention because Li metal can store 10 times more energy than graphite. However, there are safety and performance concerns for these types of batteries that arise from the formation of dendrites on the electrodes. To address this issue, researchers have proposed an unstacked graphene nanostructured lithium metal anode for lithium metal batteries to inhibit lithium dendrite growth and bring superior electrochemistry performance.
Mar 15th, 2016
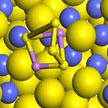 Capacity decay caused by polysulfides' detachment from the cathode framework has been a major issue preventing the broad application of lithium-sulfur (Li-S) batteries. Researchers discovered that it is the incompatibility between polar lithium polysulfide molecules and commonly used nanocarbon cathode scaffolds that restraines the redox reactivity. By incorporating sulfiphilic cobalt disulfide into carbon/sulfur cathodes, They managed to introduce strong interaction between lithium polysulfides and CoS2 under working conditions.
Capacity decay caused by polysulfides' detachment from the cathode framework has been a major issue preventing the broad application of lithium-sulfur (Li-S) batteries. Researchers discovered that it is the incompatibility between polar lithium polysulfide molecules and commonly used nanocarbon cathode scaffolds that restraines the redox reactivity. By incorporating sulfiphilic cobalt disulfide into carbon/sulfur cathodes, They managed to introduce strong interaction between lithium polysulfides and CoS2 under working conditions.
Jan 13th, 2016
 The two major problems with lithium-sulfur batteries stem from the intrinsic inert reaction kinetics of sulfur redox and the unique 'shuttle' mechanism described as that soluble intermediates - polysulfides consisting of lithium-terminated sulfur-chains - diffuse between the cathode and anode, thus being consumed within the battery instead of being utilized. To solve these issues and to improve battery performance relies on not only the electrode materials but also other cell components such as the separator.
The two major problems with lithium-sulfur batteries stem from the intrinsic inert reaction kinetics of sulfur redox and the unique 'shuttle' mechanism described as that soluble intermediates - polysulfides consisting of lithium-terminated sulfur-chains - diffuse between the cathode and anode, thus being consumed within the battery instead of being utilized. To solve these issues and to improve battery performance relies on not only the electrode materials but also other cell components such as the separator.
Nov 23rd, 2015
 Although developed only recently, inorganic halide perovskite quantum dot systems have exhibited comparable and even better performances than traditional quantum dots in many fields. They are expected to be applied in display and lighting technologies. Researchers now have reported an interesting cyclable surface dissolution and recrystallization phenomenon of inorganic perovskite crystals. This allows them to freely change size between nanometer and micrometer scales, and can be used to healing the defects inside perovskite films and hence improve the performances of optoelectronic devices.
Although developed only recently, inorganic halide perovskite quantum dot systems have exhibited comparable and even better performances than traditional quantum dots in many fields. They are expected to be applied in display and lighting technologies. Researchers now have reported an interesting cyclable surface dissolution and recrystallization phenomenon of inorganic perovskite crystals. This allows them to freely change size between nanometer and micrometer scales, and can be used to healing the defects inside perovskite films and hence improve the performances of optoelectronic devices.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





