Showing Spotlights 1521 - 1528 of 2779 in category All (newest first):
 Metal-organic frameworks (MOFs) are well-ordered, lattice-like crystals. The nodes of the lattices are metals which are connected by organic molecules. Their controlled nanometer-sized pores provide MOFs with the potential to be used in next generation gas storage, gas separation and sensors. With their special structure and large surface area, MOFs open up new opportunities for alternative systems for gas and energy storage (e.g. carbon dioxide and hydrogen storage), in catalysis, chemical sensing, as nanoreactors, and in drug delivery, making them hugely interesting for both university research and industry. Researchers have now achieved the first microfluidic method for patterning MOF crystals. As such, high-throughput single crystal patterning is achieved with an unprecedented degree of flexibility.
Metal-organic frameworks (MOFs) are well-ordered, lattice-like crystals. The nodes of the lattices are metals which are connected by organic molecules. Their controlled nanometer-sized pores provide MOFs with the potential to be used in next generation gas storage, gas separation and sensors. With their special structure and large surface area, MOFs open up new opportunities for alternative systems for gas and energy storage (e.g. carbon dioxide and hydrogen storage), in catalysis, chemical sensing, as nanoreactors, and in drug delivery, making them hugely interesting for both university research and industry. Researchers have now achieved the first microfluidic method for patterning MOF crystals. As such, high-throughput single crystal patterning is achieved with an unprecedented degree of flexibility.
Feb 16th, 2012
 Following up on our recent Nanowerk Spotlight on nanofoods, new research shows that consumers could be exposed to nanoparticles in food by a much larger degree than has been expected so far. For a modern consumer it is hard to avoid titanium dioxide (TiO2) - a widely used additive in food, personal care and other household products. Approximately 7 million tons of bulk TiO2 are produced annually and used as white pigment in order to provide whiteness and opacity to foods and other products. Many applications of titanium dioxide would benefit from smaller primary particle sizes, and we can expect the percentage of TiO2 that is produced in or near the nano range to increase.
Following up on our recent Nanowerk Spotlight on nanofoods, new research shows that consumers could be exposed to nanoparticles in food by a much larger degree than has been expected so far. For a modern consumer it is hard to avoid titanium dioxide (TiO2) - a widely used additive in food, personal care and other household products. Approximately 7 million tons of bulk TiO2 are produced annually and used as white pigment in order to provide whiteness and opacity to foods and other products. Many applications of titanium dioxide would benefit from smaller primary particle sizes, and we can expect the percentage of TiO2 that is produced in or near the nano range to increase.
Feb 15th, 2012
 At the core of tissue engineering is the construction of three-dimensional scaffolds out of biomaterials to provide mechanical support and guide cell growth into new tissues or organs. In another advance for the field, researchers have now demonstrated a strategy to fabricate tubular structures with multiple types of cells as different layers of the tube walls. This method may be widely used in simulation of many tubular tissues and enriches the toolbox for 3D micro/nanofabrication by initially patterning in 2D and transforming it into 3D. Tubular tissues such as the trachea, blood vessels, lymph vessels, and intestines, have two distinguishing features: They have specific 3D shapes;and they have different types of cells at specific locations, i.e. different parts of the tube wall are made up of different cells. Mimicking both of these features is a prerequisite for fabricating functional tubular tissues in vitro, and the realization of structural-tissue mimicry may have wide applications.
At the core of tissue engineering is the construction of three-dimensional scaffolds out of biomaterials to provide mechanical support and guide cell growth into new tissues or organs. In another advance for the field, researchers have now demonstrated a strategy to fabricate tubular structures with multiple types of cells as different layers of the tube walls. This method may be widely used in simulation of many tubular tissues and enriches the toolbox for 3D micro/nanofabrication by initially patterning in 2D and transforming it into 3D. Tubular tissues such as the trachea, blood vessels, lymph vessels, and intestines, have two distinguishing features: They have specific 3D shapes;and they have different types of cells at specific locations, i.e. different parts of the tube wall are made up of different cells. Mimicking both of these features is a prerequisite for fabricating functional tubular tissues in vitro, and the realization of structural-tissue mimicry may have wide applications.
Feb 14th, 2012
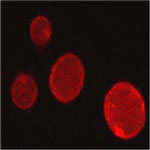 With the advent of nanomedicine, the concept of a "magic bullet" to fight cancer is getting closer to reality. Previously an idea straight out of science fiction, researchers around the world are working on perfecting nano- and microscale drug carriers that get injected into the body, transport themselves to the correct target, such as a tumor, and deliver the required dose of a medication or other substance to effectively destroy or repair this target. The controlled drug release required by these systems, however, has proven to be quite a challenging issue. To avoid the side effects of prematurely released toxic cancer drugs on healthy tissues, researchers have designed and fabricated an "active defense" system which could effectively keep the drug entrapped in its carrier in the blood and normal tissues whereas it would allow the explosive drug release under the right physiopathological stimuli once the drug carrier reaches the cancerous tissues.
With the advent of nanomedicine, the concept of a "magic bullet" to fight cancer is getting closer to reality. Previously an idea straight out of science fiction, researchers around the world are working on perfecting nano- and microscale drug carriers that get injected into the body, transport themselves to the correct target, such as a tumor, and deliver the required dose of a medication or other substance to effectively destroy or repair this target. The controlled drug release required by these systems, however, has proven to be quite a challenging issue. To avoid the side effects of prematurely released toxic cancer drugs on healthy tissues, researchers have designed and fabricated an "active defense" system which could effectively keep the drug entrapped in its carrier in the blood and normal tissues whereas it would allow the explosive drug release under the right physiopathological stimuli once the drug carrier reaches the cancerous tissues.
Feb 13th, 2012
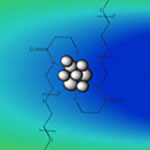 Bridging the world of atoms and nanoparticles is a class of ultrasmall nanoclusters that contain less than 100 atoms and measure just one or two nanometers. Exploiting the unique properties - optical, magnetic, catalytic - of these nanoclusters in fields such as sensors, microelectronics, biotechnology, energy, and especially catalysis, requires atomically precise fabrication techniques. Making the synthesis of ultrasmall metal nanoclusters much easier than before, researchers have now demonstrated the development of a millifluidic chip as a novel approach for reproducible, high-throughput, and controlled synthesis. Preliminary findings demonstrate that a simple, easy to fabricate millifluidic reactors has the potential for controlled synthesis of nanomaterials.
Bridging the world of atoms and nanoparticles is a class of ultrasmall nanoclusters that contain less than 100 atoms and measure just one or two nanometers. Exploiting the unique properties - optical, magnetic, catalytic - of these nanoclusters in fields such as sensors, microelectronics, biotechnology, energy, and especially catalysis, requires atomically precise fabrication techniques. Making the synthesis of ultrasmall metal nanoclusters much easier than before, researchers have now demonstrated the development of a millifluidic chip as a novel approach for reproducible, high-throughput, and controlled synthesis. Preliminary findings demonstrate that a simple, easy to fabricate millifluidic reactors has the potential for controlled synthesis of nanomaterials.
Feb 10th, 2012
 For years it has been known that scaling bulk silicon transistors would be extremely challenging, if not impossible, when lengths close in on 15 nm. Already, attention has turned to 3D transistor design and silicon-on-insulator (SOI) devices to improve the scalability of silicon technology. Carbon nanotube (CNT) transistors have been touted as a possible replacement for silicon devices but the crucial question so far has been if CNT transistors can offer performance advantages over silicon at sub-10 nm lengths? New experimental results from IBM Research are indicating that the answer is 'yes'. The findings by the research team defied the theoretical projections and exhibited encouraging performance for a device with a 9 nm channel length.
For years it has been known that scaling bulk silicon transistors would be extremely challenging, if not impossible, when lengths close in on 15 nm. Already, attention has turned to 3D transistor design and silicon-on-insulator (SOI) devices to improve the scalability of silicon technology. Carbon nanotube (CNT) transistors have been touted as a possible replacement for silicon devices but the crucial question so far has been if CNT transistors can offer performance advantages over silicon at sub-10 nm lengths? New experimental results from IBM Research are indicating that the answer is 'yes'. The findings by the research team defied the theoretical projections and exhibited encouraging performance for a device with a 9 nm channel length.
Feb 9th, 2012
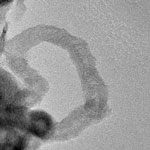 Noble metal nanoparticles such as gold, silver or platinum are widely used by scientists to develop novel applications in sensing, energy, spectroscopy, and catalysis. For instance, the combination of metal nanoparticles and carbon nanomaterials - graphene and nanotubes - has met with great interest in the area of biosensor applications as well as composite fabrication for light-energy conversion. In these applications, researchers make use of the formation of organic/inorganic hybrid nanosystems by incorporating metal nanoparticles in or onto the graphitic structures of carbon nanotubes or graphene. Researchers have now discovered a novel phenomenon whereby graphene can be catalytically transformed into carbon nanotubes by gold nanoparticles at relatively low temperatures.
Noble metal nanoparticles such as gold, silver or platinum are widely used by scientists to develop novel applications in sensing, energy, spectroscopy, and catalysis. For instance, the combination of metal nanoparticles and carbon nanomaterials - graphene and nanotubes - has met with great interest in the area of biosensor applications as well as composite fabrication for light-energy conversion. In these applications, researchers make use of the formation of organic/inorganic hybrid nanosystems by incorporating metal nanoparticles in or onto the graphitic structures of carbon nanotubes or graphene. Researchers have now discovered a novel phenomenon whereby graphene can be catalytically transformed into carbon nanotubes by gold nanoparticles at relatively low temperatures.
Feb 8th, 2012
 Technological advances have made desalination and demineralization of seawater feasible, albeit expensive, solutions for increasing the world's supply of freshwater. Among various desalination technologies, reverse osmosis membranes have been widely used for water reclamation. However, external energy required and high operational pressure used (above 75 bar for reverse osmosis desalination and 25 bar for reverse osmosis water recovery from wastewater) make reverse osmosis membrane water reclamation processes energy intensive - not exactly an advantage given the rising cost of energy and the negative climate impact of fossil fuels. Researchers have now demonstrate the novel concept of a "desalination battery", which operates by performing cycles in reverse on our previously reported mixing entropy battery.
Technological advances have made desalination and demineralization of seawater feasible, albeit expensive, solutions for increasing the world's supply of freshwater. Among various desalination technologies, reverse osmosis membranes have been widely used for water reclamation. However, external energy required and high operational pressure used (above 75 bar for reverse osmosis desalination and 25 bar for reverse osmosis water recovery from wastewater) make reverse osmosis membrane water reclamation processes energy intensive - not exactly an advantage given the rising cost of energy and the negative climate impact of fossil fuels. Researchers have now demonstrate the novel concept of a "desalination battery", which operates by performing cycles in reverse on our previously reported mixing entropy battery.
Feb 7th, 2012
 Metal-organic frameworks (MOFs) are well-ordered, lattice-like crystals. The nodes of the lattices are metals which are connected by organic molecules. Their controlled nanometer-sized pores provide MOFs with the potential to be used in next generation gas storage, gas separation and sensors. With their special structure and large surface area, MOFs open up new opportunities for alternative systems for gas and energy storage (e.g. carbon dioxide and hydrogen storage), in catalysis, chemical sensing, as nanoreactors, and in drug delivery, making them hugely interesting for both university research and industry. Researchers have now achieved the first microfluidic method for patterning MOF crystals. As such, high-throughput single crystal patterning is achieved with an unprecedented degree of flexibility.
Metal-organic frameworks (MOFs) are well-ordered, lattice-like crystals. The nodes of the lattices are metals which are connected by organic molecules. Their controlled nanometer-sized pores provide MOFs with the potential to be used in next generation gas storage, gas separation and sensors. With their special structure and large surface area, MOFs open up new opportunities for alternative systems for gas and energy storage (e.g. carbon dioxide and hydrogen storage), in catalysis, chemical sensing, as nanoreactors, and in drug delivery, making them hugely interesting for both university research and industry. Researchers have now achieved the first microfluidic method for patterning MOF crystals. As such, high-throughput single crystal patterning is achieved with an unprecedented degree of flexibility. 
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





