| Posted: Jan 03, 2007 | |
Nanowire producing bacteria form an electrically integrated community |
|
| (Nanowerk News) Bacteria are ubiquitous in the earth’s surface, subsurface, fresh water, and oceanic environment. Bacteria are remarkable in that they are capable of respiring aerobically and anaerobically using a variety of compounds, including metals, as terminal electron acceptors. | |
| Metal reducing bacteria can significantly affect the geochemistry of aquatic sediments, submerged soils, and the terrestrial subsurface. Microbial dissimilatory reduction of metals is a globally important biogeochemical process driving the cycling of iron and manganese, associated trace metals, and organic matte. Microbial metal reduction is of significant interest among scientists who are researching remediation of environmental contaminants. | |
| However, little is known about the biochemical or molecular mechanisms underlying bacterial metal reduction. Conducting research with toxic metal reducing bacteria, researchers discovered that bacteria produce electrically conductive nanowires in response to electron-acceptor limitation. These findings could be used to bioengineer electrical devices such as microbial fuel cells. | |
| The Shewanella Federation is a cross-institution consortium consisting of teams of scientists from academia, national laboratories, and private industry working collaboratively to achieve a systems-level understanding of how Shewanella oneidensis MR-1 (S. oneidensis) senses and responds to its environment. | |
| S. oneidensis is a versatile bacterium that can use a variety of organic compounds and metals as electron acceptors for respiration. Many of the compounds and metals that S. oneidensis can use as electron acceptors, such as uranium, chromate, technetium, and nitrates, can be toxic to humans and other organisms and are environmental contaminants. | |
| Microbes such as S. oneidensis have a natural ability to pluck electrons off their food or other things in their environment and transfer those electrons to nearby metal ions or oxygen molecules, thereby significantly reducing their toxicity. The intriguing aspect is that this electron parade generates a small electrical current and a tiny source of renewable energy. | |
| "Until recently, scientists thought the bacteria had to have direct contact with the metal ions or oxygen molecules in order to complete the electron transfer", says Yuri Gorby of Pacific Northwest National Laboratories. | |
 |
|
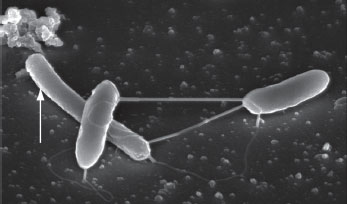
| SEM image of P. thermopropionicum and M. thermoautotrophicus (arrow) in methanogenic cocultures showing nanowires connecting the two genera. | |
| However, in a series of experiments, Gorby and colleagues induced nanowires in S. oneidensis and demonstrated that they were electrically conductive. The bacterial nanowires were as small as 10 nanometers in diameter and formed bundles as wide as 150 nanometers. They grew to be tens of microns to hundreds of microns long. | |
| It now turns out that not only are the wires and their ability to alter metal connected but that many other bacteria, including species involved in fermentation and photosynthesis, can also form wires under a variety of environmental conditions. | |
| "Earth appears to be hard-wired" says Gorby, "although we have yet to conclusively identify the electrically conductive components of Shewanella nanowires." | |
| The common thread involved depriving a microbe of something it needed to shed excess energy in the form of electrons. For example, S. oneidensis requires oxygen or other electron acceptors for respiration, whereas Synechocystis, a cyanobacterium, combines electrons with carbon dioxide during photosynthesis. Bereft of these "electron acceptors," bacterial nanowires "will literally reach out and connect cells from one to another to form an electrically integrated community," says Gorby. | |
| Gorby and his colleagues hypothesize that the nanowires may facilitate electron transfer from bacteria to metal oxides without the need for direct cell-surface-to-mineral contact or the use of dissolved electron shuttles. Nanowires significantly increase the reactive surface area involved in metal reduction well beyond that of the cell surface area. | |
| "The physiological and ecological implications for these interactions are not currently known," he said, "but the effect is suggestive of a highly organized form of energy distribution among members of the oldest and most sustainable life forms on the planet." | |
| The current hypothesis is that microorganisms may participate in interspecies electron transfer as an means to augment more familiar energy exchange processes, such as interspecies hydrogen transfer. The possibility that metabolically and taxonomically diverse microbial groups have adopted a common strategy for efficient energy transduction and distribution via electrically conductive nanowires is scientifically intriguing. | |
| When energy is efficiently distributed in a ecosystem, Gorby says, everybody wins: "One of the problems we have in the world is the inequity of energy and wealth. So I think this cooperation among different groups of bacteria leads to sustainability. And since they've been around about 3.5 billion years, I'd say it's a pretty effective strategy." | |
| To measure currents as precisely as possible, Gorby and colleagues from the University of Southern California have built a microbial fuel cell laboratory at PNNL. The small bacteria-powered batteries, cultured under electron-acceptor limitations and fueled by lactate or light, produce very little power, as measured by a voltmeter hooked to a laptop computer. | |
| But co-author and PNNL scientist Jeff Mclean, who works towards developing and operating the Microbial Cell Dynamics Laboratory at PNNL, said that small changes in fuel cell design and culture conditions have already shown large improvements in the efficiency of the fuel cells. For example, so-called biofilms – a highly interconnected bacterial community – put out much more energy than other configurations. | |
| The researchers are now trying to figure out how to make the bacteria grow additional nanowires, which could help making the fuel cells more conductive and therefore more efficient. | |
| The researchers findings were published in Proceedings of the National Academy of Sciences ("Electrically conductive bacterial nanowires produced by Shewanella oneidensis strain MR-1 and other microorganisms"). | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
