| Posted: Nov 25, 2015 | |
Just shake it! A simple way to remove nanomaterial pollutants from water |
|
| (Nanowerk Spotlight) As the production and use of nanotechnology-based products in our daily life is rapidly growing, the risks of environmental pollution due to nanomaterials are increasing as well. | |
| The spreading of nanomaterials in manufacturing, product application, and waste management processes will eventually lead to some degree of contamination of water. While 'conventional' water contaminants can be cleaned up by state-of-the-art technologies with filtration and condensation processes, this is not the case for nanoscale pollutants. | |
| The size of nanomaterials commonly is below 100 nm – thousands of times smaller than regular water contaminants and much smaller than the micron-sized or larger pores used in conventional water filters. Simply reducing the filter pore size is not a solution either since that will cause clogging of the filtration line. | |
| "In our new work, we have demonstrated that water contaminated with nanomaterials can be cleaned up by a 'hand shaking' approach that can be performed even in a kitchen." Dr. Yoke Khin Yap, a professor in the Department of Physics at Michigan Technological University, tells Nanowerk. "Our approach is simple and universal, and can be used for many one-dimensional (1D) and two-dimensional (2D) nanomaterials including nanotubes, nanowires, graphene, and nanosheets. Therefore, our approach would support continued development of nanotechnology by reducing the risk of water contamination." | |
| Yap and his team recently have found that microscopic fluid interface dynamics could be utilized to extract and remove nanomaterials from the contaminated water. They describe the details of this novel extraction technique in ACS Applied Materials & Interfaces ("A Simple and Universal Technique To Extract One- and Two-Dimensional Nanomaterials from Contaminated Water"). | |
 |
|
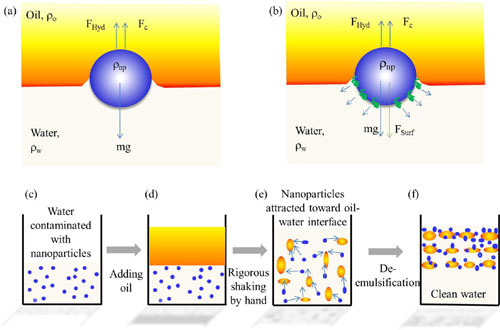
| Force diagram of an (a) unfunctionalized and a (b) functionalized spherical nanoparticle. The schematic of the extraction mechanism are shown in panels c, d, e, and f. (Reprinted with permission by American Chemical Society) (click on image to enlarge) | |
| This novel technique is applicable to the extraction of 1D and 2D nanomaterials from water with an efficiency of almost 100%. The approach involves emulsification of the contaminated water with oil – or other organic solvents – by hand shaking. These nanomaterials will then be captured in the oil phase. They can be removed after oil and water are separated, i.e. the water is condensed. | |
| The extraction of nanomaterials demonstrated here is based on the capillary force generated at the interface of oil (or organic solvent) droplets in water during the emulsification process. | |
| According to the researchers, the extraction of functionalized nanomaterials is dependent on the concentration of surfactants used. If the concentration is sufficiently low, then the extraction is possible with high efficiency. | |
| "Our attempts to extract spherical particles confirmed that the shape of the particles has an effect on the extraction mechanism," Yap points out. | |
| He also notes that, in principle, the nanomaterials can be reused once the organic solvent/oil has been evaporated. | |
| While in general, this simple approach will be applicable to extract many other particulates or solids in water, the extraction of zero dimensional (0D) materials such as quantum dots is still low in efficiency. The team hopes to overcome this issue in its next study. | |
| The idea for this technique goes back several years to when the team devised a nanofilter to separate water and oil and published a well-cited paper on the results (Carbon, "The performance of superhydrophobic and superoleophilic carbon nanotube meshes in water?oil filtration"). | |
| "We thought that the superoleophilicity of nanotubes will allow us to extract nanotubes from contaminated water by the technique reported in our present paper," says Yap. "Indeed, it works fine but we also found that the same technique works as well for nanowires, which are not superoleophilic. That prompted us to find the explanation for our data and the capillary force was the answer." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
