| Posted: Mar 15, 2018 | |
Cell sex impacts the biological uptake of nanoparticles |
|
| (Nanowerk Spotlight) One of the key issues in nanomedicine is the question of how to effectively transport therapeutical nanoparticles and associated drugs to and into cells. | |
| Researchers have been studying the underlying mechanisms that so far have prevented successful clinical translation of a majority of medical nanotechnology applications. It turns out that when nanoparticles enter a biological environment such as blood, they immediately get exposed to a range of factors that can impact their effectiveness and efficiency. It has become quite clear over time that the cellular response to nanomaterials depends on the physiological environment. | |
| For instance, biological responses to nanoparticles are temperature-dependant. Researchers also have discovered the crucial role of biomolecular coronas for nanoparticle-cell interactions: the biological responses to nanoparticles are strongly dependent on the type and amount of associated proteins in the composition of the biomolecular corona. | |
| Scientists now have identified yet another, so far overlooked factor that impacts nanoparticle uptake. | |
| In a new study accepted in ACS Nano ("Effect of Cell Sex on Uptake of Nanoparticles: The Overlooked Factor at the Nanobio Interface"), an international team of researchers discovered that cell sex is an important overlooked factor at the nanobio interfaces. | |
| More specifically, depending on their sex, cells respond differently to the exact same type of nanoparticles. | |
 |
|
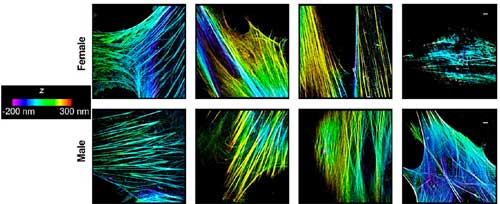
| Super-resolution images (color corresponds to position in z) demonstrating differences between organization, distribution, and morphology of actin filaments/bundles in the cytoplasm of amniotic mesenchymal stem cells extracted from the male versus female fetuses. Scale bar is 1 µm. (Reprinted with permission by American Chemical Society) (click on image to enlarge) | |
| "Our findings have a capacity to optimize clinical translation of nanoparticles and also to help researchers to better design and produce safe and efficient therapeutic sex-specific nanoparticles," Morteza Mahmoudi, an Instructor at Harvard Medical School and the former Director of and Principal Investigator at the NanoBio Interactions Laboratory at Tehran University of Medical Sciences, tells Nanowerk. | |
| It has been know that the differences between female and male embryos originate at a very early developmental stage prior to the initiation of hormonal changes; therefore genetically and structurally driven sexual dimorphisms are expected at this early stage. | |
| For their study, the team selected human amniotic cells (hAMSCs), which represent one of the earliest sources of somatic stem cells, to uncover the potential effect of sex differences in nanoparticle uptake. As these cells are extracted from the amniotic layer of the placenta, they are in their early passages, which can provide more accurate nanoparticle update outcomes compared to other cell types. | |
| They also tested whether cell sex affects nanoparticles' uptake in different cell types. For that they repeated nanoparticle uptake experiments with somatic cells derived from the salivary gland of adult male and female patients. Here as well the results show different outcomes in terms of QD uptake. | |
| As nanoparticle model system they selected monodispersed commercially available quantum dots (QD). | |
| "In this work, our team tested the hypothesis that the observed significant difference in the QD uptake between male and female cells could be because of, but not limited to, 1) variations in secreted biomolecules (i.e., paracrine factors) and/or 2) sex-based variation of cell functions and structures (e.g., various membrane composition and intracellular pathways and, also, cell stiffness, i.e., structural differences in their cytoskeleton)," explains Mahmoudi. "We were able to demonstrate the importance of cell sex in uptake of nanoparticles. Consequently, we suggest that cell sex is an overlooked factor in research relevant to the nanobio interface." | |
| "Considering that our study revealed differences between male and female cells based on analysis of a limited number of factors and also that the effect of cell sex varied between cell types (fibroblasts and hAMSCs), it is likely that there are other undiscovered differences that could influence nanoparticle uptake," he concludes. "Further research will be required on a greater variety of cell types." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
