| Posted: Sep 17, 2008 | |
Functionalization of carbon nanotubes is key to electrochemical nanotechnology devices |
|
| (Nanowerk Spotlight) Pristine carbon nanotubes (CNTs) are insoluble in many liquids such as water, polymer resins, and most solvents. This means they are difficult to evenly disperse in a liquid matrix such as epoxies and other polymers, complicating efforts to utilize CNTs' outstanding physical properties in the manufacture of nanocomposite materials, as well as in other practical nanotechnology applications which require preparation of uniform mixtures of CNTs with many different organic, inorganic, and polymeric materials. | |
| To make nanotubes more easily dispersible in liquids, it is necessary to physically or chemically attach certain molecules, or functional groups, to their sidewalls without significantly changing the nanotubes' desirable properties. This process is called functionalization. Numerous nanotechnology research projects around the world are dedicated to developing various kinds of CNT-based electrochemical devices through the rational functionalization of CNTs. A recent review report provides an overview of the latest developments and the current state of this research ("Rational Functionalization of Carbon Nanotubes Leading to Electrochemical Devices with Striking Applications"). | |
| The review focuses on electrochemical sensors and biosensors and applications in biofuel cells and neuro-electroanalysis based on work conducted at the Beijing National Laboratory for Molecular Sciences. | |
| Single-walled CNTs arguably are the ultimate biosensor among nanoscale semiconducting materials not only for their size – which is directly comparable to the size of single biomolecules – but also because their low charge-carrier density is directly comparable to the surface charge density of proteins. Furthermore, a single-walled CNT (SWCNT) consists solely of surface such that every single carbon atom is in direct contact with the environment, allowing optimal interaction with nearby biomolecules (see: Biosensing mechanism with carbon nanotubes explained). | |
| In their review, the authors, led by Prof. Lanqun Mao, write that the unique electronic and structural properties of pristine CNTs have endowed CNT-based electrochemical sensors with excellent analytical properties even without any surface functionalization of CNTs. But they also point out that the widespread development of novel electrochemical nanotube devices will require rational surface functionalization of CNTs because the CNTs themselves do not bear many (bio)electrochemically functional moieties that could be employed as electrocatalysts for electrochemical sensing or as bio-recognition elements for electrochemical biosensing and biofuel cells. | |
| Already in 2005, Mao and his group have been demonstrating a novel electrochemical method for sensitive determination of biological thiols based on rational functionalization of SWCNTs ("Rational Attachment of Synthetic Triptycene Orthoquinone onto Carbon Nanotubes for Electrocatalysis and Sensitive Detection of Thiols"). Unlike previous strategies used for the functionalization of the carbon nanotubes to fabricate new kind of electrochemically functional nanostructures, the method they demonstrated then is essentially based on understanding of the redox properties inherent in the SWCNTs themselves. | |
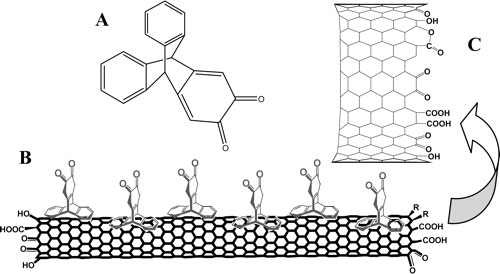 |
|
| Structure of Triptycene Orthoquinone (TOQ, A), Schematic Illustration of Attachment of TOQ onto Carbon Nanotubes (B), and Enlarged Tube Ends with Oxygen-Containing Moieties (C) (Reprinted with permission from American Chemical Society) | |
| "Another example of the functionalization of CNTs with organic compounds concerns polynuclear aromatic electroactive dyes" says Mao. "Such compounds have received much attention because they possess good electrochemically redox properties and have been widely used for electrocatalysis and electrochemical sensing. By taking advantage of the hydrophobic and/or charge-transfer interaction(s) between methylene blue (MB), a polynuclear aromatic electroactive dye, and CNTs, we have prepared a new type of electrochemically functional MB-CNT adsorptive nanostructure with good electrochemical properties that are distinct from those of MB adsorbed onto a glassy carbon electrode." | |
| The excellent electroactivity and the high stability of the layered nanocomposite gives them great potential for the development of electrochemical devices, such as biosensors and photovoltaic cells. | |
| Another strategy for the development of electrochemical devices is the functionalization of CNTs with metallic nanoparticles. | |
| "Motivated by the excellent electrochemical properties and the striking applications of the electrochemical devices based on the nanocomposites of transition metal hydroxides/oxides and CNTs, we have recently been interested in the functionalization of CNTs with metal hydroxide/oxide nanostructures" says Mao. | |
| In a recent study, the group essentially offers a facile but effective and relatively general electrochemical approach to the synthesis of the nanocomposites consisting of metal hydroxides/oxides and multi-walled CNTs ("A general electrochemical approach to deposition of metal hydroxide/oxide nanostructures onto carbon nanotubes"). | |
| Generally, the strategies involving smart functionalization of CNTs with chemically, biochemically, or electrochemically functional materials could provide a straightforward approach to the development of nanostructured electrochemical devices with promising properties and striking applications. The Chinese scientists caution, though, that such a strategy may yet be limited in because the strategies for the functionalization of the CNTs have strongly relied on the interactions inherent to the CNTs and the materials, which are mainly dependent on the structural and chemical properties of the materials. | |
| "As a consequence, the strategies demonstrated thus far might not be applicable for all kinds of target materials used for the smart functionalization of CNTs to form functional electrochemical devices" says Mao. "For example, some kinds of small molecular electrocatalysts might not strongly interact with CNTs and, as a result, may not stably functionalize the CNTs. To circumvent this problem, our recent strategy for the rational functionalization of CNTs and ionic liquid bucky gel might provide a platform for the development of CNT-based electrochemical devices through the functionalization of CNTs." | |
| This strategy is based on the formation of a bucky gel of CNTs in an ionic liquid through the p?cation interactions inherent to both components and on the capability of ionic liquids to dissolve a wide range of compounds ("Rational Functionalization of Carbon Nanotube/Ionic Liquid Bucky Gel with Dual Tailor-Made Electrocatalysts for Four-Electron Reduction of Oxygen"). | |
| In the final part of the review, Mao outlines his group's interest in the application of CNT-based electrochemical sensors and biosensors to probe brain chemistry. | |
| "The complexity of cerebral systems has made the effective analysis of brain chemistry a long-standing challenge" says Mao. "By combining electrochemistry, materials science, and neurosciences, we have recently been working on effective electrochemical methods for probing brain chemistry, with emphasis on in vivo and online monitoring of physiologically important species, such as ascorbic acid, neurotransmitters, free radicals, and glucose/lactate in the rat brain. Our protocols have been primarily based on the exploitation of CNTs to fulfill the requirements of the electrochemical methods employed for the above purpose with respect to selectivity, sensitivity, stability, and reproducibility." | |
| The examples showcased in this review demonstrate that the use of CNTs could provide an effective approach to the development of various kinds of nanostructured electrochemical devices, electrochemical sensors, biosensors, and biofuel cells, in particular, owing to the unique electronic and structural properties and rational functionalizability of CNTs. | |
| Moa and his colleagues point out, though, that challenges along the way include establishing effective methods both for the preparation of CNT-based electrochemical devices with tunable properties and for the smart functionalization of CNTs to endow the developed devices with the desired properties. | |
| The ultimate goal, of course, will be to extend CNT-based electrochemical devices into real-world applications by combining several research fields, including materials science, electrochemistry, energy conversion and storage, and life sciences. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
