| Posted: Sep 06, 2006 | |
Nanotechnology lego construction inspired by biominerals |
|
| (Nanowerk Spotlight) Nanocrystal engineering learned from biominerals holds promises for the development in biology, chemistry, and materials science. Biominerals have inspired novel bottom-up approaches to the development of functional materials for some time now. The morphology, crystallographic orientation, incorporated organic molecules, and emergent properties of carbonate-based biominerals already have been demonstrated. Typical examples of these biominerals are certain layers of seashells, corals, and eggshells. New research now clarifies that biominerals are oriented architectures of calcium carbonate nanocrystals 20–100 nm in size with incorporation of biopolymers. | |
| These calcium carbonate superstructures give rise to questions such as, Why do various curved morphologies emerge from the same combination of calcium carbonate and biopolymers? How do living organisms build up inorganic materials with characteristic macroscopic morphologies from their precursors? Why are many bioinorganic architectures colored with dye molecules? | |
| The nanoscopic structure of biominerals, especially in terms of the growth and orientation of the crystals, was not fully understood until recent years, while the macroscopic morphology, crystal structure, and the included organic molecules were well investigated. Recently, the oriented architectures of nanoparticles have featured by a research group at the Max Planck Institute of Colloids and Interfaces in Potsdam/Germany ("Oriented attachment and mesocrystals: Non-classical crystallization mechanisms based on nanoparticle assembly"). | |
| In earlier work, low-magnification images showed the smooth crystal surface of biominerals as a single crystal. Now, working with selected-area electron diffraction (SAED) and X-ray diffraction (XRD) researchers found that oriented architectures of nanocrystals formed in various biominerals, at least within a certain area that is several hundred nanometers in scale, regardless of the species and crystal phases. | |
| Dr. Yuya Oaki, first author, and Prof. Hiroaki Imai from the Department of Applied Chemistry at Keio University in Japan, published a recent paper on nanoscale architectures in biominerals and their biomimetics that will lead to the improved understanding of biomineralization and the further development of materials science. The paper, titled "Bridged Nanocrystals in Biominerals and Their Biomimetics: Classical Yet Modern Crystal Growth on the Nanoscale" appeared in the July 3, 2006 online edition of Advanced Functional Materials. | |
 |
|
| The oriented architecture consisting of bridged nanocrystals with incorporated biopolymers. a–c) The FETEM images of the mineral bridges in sea urchin spines (a) and eggshells (b,c). d) The normal unfiltered FETEM image acquired from the dye-incorporated sample of a sea urchin spine. e) The corresponding EF-TEM image for the mapping of bromine-substituent EY molecules, implying that the dye molecules were homogeneously included in the oriented architecture. f) The schematic model for the oriented architecture made by the bridged nanocrystals. g) The nanoscopic space for dye molecules in the architecture consisting of nanocrystals and organic polymers. (Reprinted with permission from Wiley) | |
| The authors explain the recent findings to Nanowerk: "We have proposed a new idea that the nanoscopic mineral bridges between the tiny grains play an important role for the generation of oriented architecture. We show that both biominerals and biomimetics form oriented architectures of bridged nanocrystals with incorporated organic polymers." | |
| The oriented architectures may be classified into a family of "mesocrystals" but further investigations are required to understand the detailed structures and formation mechanisms. | |
| The growth of nanocrystals with mineral bridges leads to oriented structures and subsequent macroscopic morphologies, says Oaki. "The bricklaying growth strategy using inorganic nanobricks of crystals and organic nanomortars of polymers can realize a versatile morphological design of materials through a bottom-up approach." | |
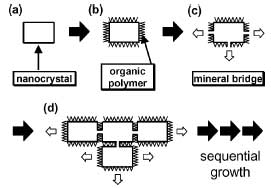 |
The schematic representation for the growth of bridged nanocrystals leading to the oriented architecture. a) The formation of a nanocrystal. b) The growth inhibition because of the adsorption of polymers. c) The growth resumption by the formation of a mineral bridge. d) The growth of the adjacent nanocrystals. This growth mode sequentially occurs on the nanoscopic scale and then leads to the oriented architecture having a specific macroscopic shape. (Reprinted with permission from Wiley) |
| This means that the design of a macroscopic morphology can be compared to a nanoscale Lego construction that has no restrictions. | |
| The growth of nanocrystals by association with polymers is a significant step for the understanding and developing of crystal growth. The strategy could be applied to various systems in nanoscale crystal growth leading to functional materials such as metal oxide. | |
| "We actually published ("Biomimetic Synthesis of Wurtzite ZnO Nanowires Possessing a Mosaic Structure")the preparation of zinc oxide nanowires based on our approach" says Oaki. "As you know, the fabrication of low-dimensional nanomaterials has been well demonstrated in recent studies. On the other hand, the integration of modular nanomaterials is an important challenge in the next stage of materials science. Our biomimetic approach would be a powerful tool for the synthesis and integration of nanomaterials through self-organization." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
