| Posted: Jan 14, 2009 | |
A new class of radiation detection materials |
|
| (Nanowerk Spotlight) Scintillation – a flash of light, lasting only nanoseconds to microseconds, produced in a particular material when it absorbs ionizing radiation – is one of the main methods used for the detection of ionizing radiation. Scintillators are very versatile and can be used for detecting almost all types of radiation in a wide range of energy from several electron volts (eV) to tens of billion of electron volts (GeV). | |
| Scintillation is useful for sensing alpha, beta and gamma radiation as well as other nuclear particles and in some cases determining the particle type. For example, organic scintillators can sense fast neutrons emitted spontaneously from fissile isotopes of uranium and plutonium, enabling passive detection of nuclear weapons. The detection and identification of subatomic particles is not only important for nuclear nonproliferation efforts but is a wide-ranging scientific problem with implications for medical devices, radiography, biochemical analysis, particle physics, and even astrophysics. | |
| Existing scintillators, which are capable of neutron discrimination, are either organic crystals or liquids. Anisotropy of organic crystals limits their utility, while liquids pose transportation and safety hazards, and show poor performance when scaled to large volumes (liters). An ideal material for scintillation applications would be an isotropic solid scalable to many liters to increase sensitivity. | |
| Scientists from the Sandia National Laboratories in California have now proposed that metal-organic frameworks (MOFs) could potentially offer the desired level of structural control, leading to an entirely new class of radiation detection materials. | |
| "MOFs are crystalline materials consisting of metal clusters linked by coordinating organic groups," explains Mark Allendorf, who leads efforts at Sandia to develop both the fundamental science and applications of MOFs. "In our recent work, we have found that MOFs can be made to scintillate and that the materials tested are exceptionally robust against radiation damage. Our results demonstrate that control over both the electronic structure and local environment of a chromophore are possible in MOFs." | |
| "This effectively creates a nanolaboratory for the systematic study of a broad range of photophysical phenomena, including scintillation, fluorescence, and energy transfer," adds Patrick Doty, who collaborated with Allendorf in the development of MOF scintillators and is first author of a recent paper in Advanced Materials that reports the team's findings ("Scintillating Metal-Organic Frameworks: A New Class of Radiation Detection Materials"). | |
| In this paper, the Sandia researchers describe spectroscopic measurements using single crystals of MOF-S1 and MOF-S2. They demonstrate that these two MOFs respond to ionizing radiation by emitting light, creating the first completely new class of scintillation materials since the advent of plastic scintillators in 1950. | |
| "This opens a new route to the development of high-performance scintillators with properties tailored to the detection of specific particle types and applications," says Allendorf. "These MOFs have scintillation quantum yields comparable to commercial organic scintillators." | |
| He points out that, unlike either organic scintillators in solution or the pure materials themselves in their crystalline state, MOFs have a highly tailorable chemical structure that allows their electronic structure to be tuned to achieve specific desirable properties in a scintillator. Furthermore, some MOFs are nanoporous and can accommodate species (atoms or molecules) in their pores that can improve scintillator performance. | |
 |
|
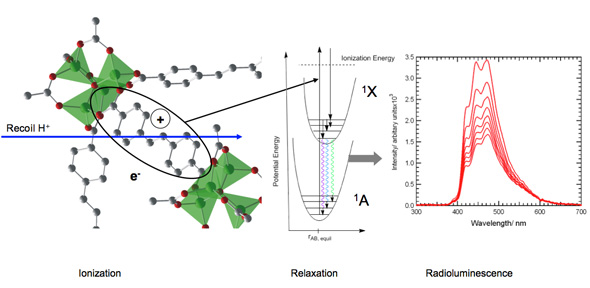
| Schematic of the fast neutron detection process (using a portion of the MOF-S1 structure for illustration), showing (left) the interaction of a high-energy recoil proton created by neutron elastic scattering the material. The resulting excitation decays (center) through radiationless pathways (solid arrows) to the lowest-lying excited state of the fluorescent SDC groups, which then decay by emission of photons (wavy arrows) to various ground-state vibrational levels, producing the prompt luminescence (right) displaying vibronic structure. Luminescence degrades very slowly with integrated dose dose up 10 MGy (1Gy = 1J/kg). (Image: Patrick Doty, Sandia National Laboratories) | |
| "We found that the luminosity of both MOFs is comparable to commercial organic scintillators" says Doty. "We did this by exposing both MOFs to high-energy (3 MeV) protons from an ion microprobe and a source of alpha particles. The proton beam simulates the production of recoil protons by elastic scattering of fast neutrons within an organic scintillator, leading to radioluminescence. To probe the interaction with protons, we used ion-beam-induced luminescence spectroscopy, a technique in which the luminescence is collected by an optical fiber, dispersed in a monochromator, and detected using a charge-coupled device." | |
| Compared with known scintillation materials, MOFs present an exceptional opportunity to perform nanoscale 'crystal engineering', creating the potential for rational design of new scintillation materials: MOF structures can be predicted, unlike other extended crystalline materials such as zeolites and molecular solids. Furthermore, variation of the organic component allows for structural modifications that can be used to tailor MOF properties. The relatively rigid structure of some MOFs can create permanent nanoporosity, which enable them to serve as hosts for atoms or molecules designed to improve the detection efficiency and specificity. | |
| "From a practical point of view, we envision the development of new MOF-based detector materials with luminosity, timing, and specificity tailored for detector applications, such as thermal and fast-neutron detection," Allendorf describes the team's ambitions. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
