| Posted: Mar 24, 2009 | |
Count your carbs with nanotechnology probes |
|
| (Nanowerk Spotlight) Over the past few years, scientists have taken advantage of the unique optical and other physical properties of metal nanoparticles to create a wide range of nanotechnology probes for electronic, optical, and microgravimetric transduction of different biomolecular recognition events. An interesting approach that was reported a couple of years ago deals with a technique that estimates the antioxidant power of certain food samples by measuring the generation and growth of gold nanoparticles (see: "Nanoparticle-Based Assays of Antioxidant Activity"). | |
| In a previous Spotlight, we have reported on how scientists are starting to explore the ability of plants to synthesize metal nanoparticles such as silver or gold ("Truly green nanotechnology – growing nanomaterials in plants"), and it has already been demonstrated that enzyme-mediated growth of metallic nanoparticles provides a general means to follow biocatalyzed transformations, and to develop optical sensors for different substrates such as glucose, alcohols, lactate or nerve gas analogs ("Growing Metal Nanoparticles by Enzymes"). | |
| For instance, researchers have shown that these colorimetric detection processes can be used to evaluate the antioxidant power of food-related phenolic acids – plant metabolites widely spread throughout the plant kingdom – based on the polyphenol-mediated growth of gold nanoparticles. | |
| "In previous work it was shown that he antioxidant power of food can be explored by its ability to form gold nanoparticles from a gold solution," Matteo Scampicchio tells Nanowerk. "We have built on these findings by developing a novel optical nanoprobe based on nylon nanofibrous membranes produced by electrospinning and impregnated with gold ions. The optical properties of the gold nanoparticles relate to the total reducing sugar content of the samples analyzed." | |
| This technique could lead to the development of inexpensive and disposable optical nanoprobes that could find applications in a host of industrial, biomedical and clinical fields. | |
| Scampicchio is a researcher in the Department of Food Science and Microbiology at the University of Milan in Italy and first author of a recent paper in Nanotechnology that describes these findings ("Optical nanoprobes based on gold nanoparticles for sugar sensing"). | |
| Natural antioxidants are of great interest due to several claims about their health feature and, hence, their potential contribution to the defense against oxidative stress and protection against cancers and cardiovascular diseases. Unfortunately, routinely methods to evaluate antioxidant power of foods require generally long time of analysis (up to two hours), or laborious procedures. | |
| "What we were able to demonstrate is that foods are inherently able to reduce gold (III) to metal particles to form a colored colloidal suspension," says Scampicchio. "This capability is directly related to the antioxidant power of the foods. The generation of gold nanoparticles in solution produce a purple color, whose intensity correlate well with the redox characteristics of the samples analyzed." | |
| The team took advantage of these findings by transforming this bionano-metallization concept into a novel analytical application for food analysis. | |
 |
|
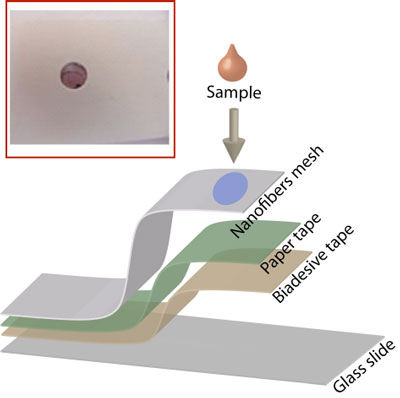
| Fabrication of the optical nanoprobe, made with electrospun nylon-6 nanofibrous membranes. Also shown as inset the colored spot obtained by spiking a food solution rich in reducing sugars into the nanofibrous membranes impregnated with gold in alkaline media. (Source: Dr. Matteo Scampicchio, University of Milan) | |
| "We started by analyzing the reaction between food rich in antioxidants – vitamin C and polyphenols – like green, black and oolong tea, wine and fruit juice and gold salt," explains Scampicchio. "The results indicated that green tea is the best sample to induce the growth of gold nanoparticles. This is related of course to its highest content in polyphenols like epigallocatechins." | |
| The researchers then extended this observation to the reducing sugars present in commercial foods and beverages, like Diet Coke, honey, milk and other low-carb foods. By changing the experimental conditions to alkaline, they observed that the sample solutions rich in reducing sugars turned purple, indicating the presence of carbohydrates. This reaction takes place within only minutes and without the use of any other chemicals except few nanomoles of gold salt and the alkaline media to dissolve the samples. | |
| "We were able to clearly see that the intensity of the coloration varies with the sugar, the strongest purple coloration is shown using galactose, followed by glucose, then fructose, whilst the non-reducing sugar sucrose shows no change," says Scampicchio. | |
| The nanoprobe membranes work as a disposable test strip. Once an drop of a sample is added to the transparent membrane, the carbohydrates generate gold nanoparticles from the gold salts, which turn the color of the membrane purple, indicating the presence of the reducing sugars. | |
| Optical nanoprobes like this one can find important applications in the control of sugars, for example in the rapid screening of diabetic foods and beverages. It would also be feasible to functionalize the gold nanoparticles and the nanofibrous membranes with reagents, antibodies and/or enzymes to detect a range of important foods analytes and contaminants. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
