Showing Spotlights 33 - 40 of 65 in category All (newest first):
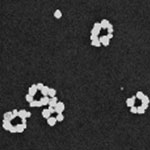 DNA origami is a design technique that is used by nanotechnology researchers to fold DNA strands into something resembling a programmable pegboard on which different nanocomponents can be attached. These DNA assemblies allow the bottom-up fabrication of complex nanostructures with arbitrary shapes and patterns on a 100 nm scale. For instance, DNA origami have been heralded as a potential breakthrough for the creation of nanoscale circuits and devices. DNA can also be metallized with different metals, resulting in conducting nanowires. Researchers have now have developed a method to assemble metallic nanocircuits with arbitrary shapes, by attaching metallic nanoparticles to select locations of the DNA origami and then fusing them to form wires, rings, or any other complex shape. These pre-designed structures are programmed by fully utilizing the self-assembling and recognition properties of DNA.
DNA origami is a design technique that is used by nanotechnology researchers to fold DNA strands into something resembling a programmable pegboard on which different nanocomponents can be attached. These DNA assemblies allow the bottom-up fabrication of complex nanostructures with arbitrary shapes and patterns on a 100 nm scale. For instance, DNA origami have been heralded as a potential breakthrough for the creation of nanoscale circuits and devices. DNA can also be metallized with different metals, resulting in conducting nanowires. Researchers have now have developed a method to assemble metallic nanocircuits with arbitrary shapes, by attaching metallic nanoparticles to select locations of the DNA origami and then fusing them to form wires, rings, or any other complex shape. These pre-designed structures are programmed by fully utilizing the self-assembling and recognition properties of DNA.
Aug 11th, 2011
 To build microprocessors with more than one billion transistors, manufacturers still use the same technique - photolithography, the high-tech, nanoscale version of printing technology - that they have been using for the past 50 years. State-of-the-art photolithography processes use 193 nm light to produce diffraction-limited features as small as 32 nm. Going beyond 32 nm, the cost and complexity rises significantly, posing massive technological and economic challenges for chip manufacturers. This provides plenty of incentives for researchers to explore alternative manufacturing technologies for chipmakers. One novel approach is based on the use of DNA nanostructures to pattern a silicon wafer.
To build microprocessors with more than one billion transistors, manufacturers still use the same technique - photolithography, the high-tech, nanoscale version of printing technology - that they have been using for the past 50 years. State-of-the-art photolithography processes use 193 nm light to produce diffraction-limited features as small as 32 nm. Going beyond 32 nm, the cost and complexity rises significantly, posing massive technological and economic challenges for chip manufacturers. This provides plenty of incentives for researchers to explore alternative manufacturing technologies for chipmakers. One novel approach is based on the use of DNA nanostructures to pattern a silicon wafer.
Jul 26th, 2011
 MicroRNAs (miRNAs) are short ribonucleic acid molecules, consisting of 21-25 nucleotide bases, that negatively regulate gene expression, also termed as gene silencing. Each miRNA is thought to regulate multiple genes, and since the human genome encodes hundreds of miRNAs, the potential regulatory circuitry afforded by miRNA is enormous. Recent discoveries suggest the association of specific miRNA sequences with a spectrum of diseases including cardiovascular and autoimmune diseases, as well as with a variety of cancers. It is therefore imperative, for diagnostics and prognostics, to accurately measure the expression levels of target miRNA molecules in patients' tissue samples or body fluids. To that end, researchers have developed an alternative way for the direct analysis of miRNAs in an array format, demonstrating fast and ultrasensitive detection of specific miRNAs.
MicroRNAs (miRNAs) are short ribonucleic acid molecules, consisting of 21-25 nucleotide bases, that negatively regulate gene expression, also termed as gene silencing. Each miRNA is thought to regulate multiple genes, and since the human genome encodes hundreds of miRNAs, the potential regulatory circuitry afforded by miRNA is enormous. Recent discoveries suggest the association of specific miRNA sequences with a spectrum of diseases including cardiovascular and autoimmune diseases, as well as with a variety of cancers. It is therefore imperative, for diagnostics and prognostics, to accurately measure the expression levels of target miRNA molecules in patients' tissue samples or body fluids. To that end, researchers have developed an alternative way for the direct analysis of miRNAs in an array format, demonstrating fast and ultrasensitive detection of specific miRNAs.
Jun 6th, 2011
 A Single Nucleotide Polymorphism (SNP) is a single nucleotide replacement in a DNA sequence - occurring when a single nucleotide (A, T, C, or G) in the genome differs - which can result in different reaction by people to pathogens and medicines. Detection of these SNPs is becoming increasingly important with the move towards more personalized healthcare. Researchers are therefore working hard in developing biomedical lab-on-chip sensors that allow the fast detection of SNPs in DNA using only very small samples of a patient's blood. Already, nanoscale detection techniques such as synthetic nanochannels are being used for DNA detection by specific DNA hybridization with molecular probes immobilized on the nanochannel walls. However, the preparation of these sensors is not easy and specific functionalization at the wall surface remains a critical issues. Researchers have now introduced a new concept of DNA-based molecular recognition agents which allows detecting SNPs with very high precision and efficiency.
A Single Nucleotide Polymorphism (SNP) is a single nucleotide replacement in a DNA sequence - occurring when a single nucleotide (A, T, C, or G) in the genome differs - which can result in different reaction by people to pathogens and medicines. Detection of these SNPs is becoming increasingly important with the move towards more personalized healthcare. Researchers are therefore working hard in developing biomedical lab-on-chip sensors that allow the fast detection of SNPs in DNA using only very small samples of a patient's blood. Already, nanoscale detection techniques such as synthetic nanochannels are being used for DNA detection by specific DNA hybridization with molecular probes immobilized on the nanochannel walls. However, the preparation of these sensors is not easy and specific functionalization at the wall surface remains a critical issues. Researchers have now introduced a new concept of DNA-based molecular recognition agents which allows detecting SNPs with very high precision and efficiency.
Feb 25th, 2011
 Researchers worldwide are working on fast and low-cost strategies to sequence DNA, that is, to read off the content of our genome. Particularly promising for future genome sequencing are devices that measure single molecules. In this respect, the creation of nanochannels or nanopores in thin membranes has attracted much interest due to the potential to isolate and sense single DNA molecules while they translocate through the highly confined channels. Particularly interesting are techniques that can offer fast and low cost readout of long DNA molecules without the need of DNA labelling or amplification. In very interesting work performed at Imperial College London, researchers have now successfully developed a protocol for the fabrication of a solid state nanopore aligned to a tunneling junction.
Researchers worldwide are working on fast and low-cost strategies to sequence DNA, that is, to read off the content of our genome. Particularly promising for future genome sequencing are devices that measure single molecules. In this respect, the creation of nanochannels or nanopores in thin membranes has attracted much interest due to the potential to isolate and sense single DNA molecules while they translocate through the highly confined channels. Particularly interesting are techniques that can offer fast and low cost readout of long DNA molecules without the need of DNA labelling or amplification. In very interesting work performed at Imperial College London, researchers have now successfully developed a protocol for the fabrication of a solid state nanopore aligned to a tunneling junction.
Dec 13th, 2010
 A very promising field of nanomotor research are DNA nanomachines. These are synthetic DNA assemblies that switch between defined molecular shapes upon stimulation by external triggers. They can be controlled by a variety of methods that include pH changes and the addition of other molecular components, such as small molecule effectors, proteins and DNA strands. Researchers have now designed and built a simple DNA machine that is capable of continuous rotation with controlled speed and direction - a function that might be very useful for example for molecular transport. This machine is driven by an externally controlled electric field. When this field is oscillated between four directions, it continuously reorients a rotor DNA that is asymmetrically attached to a DNA axle.
A very promising field of nanomotor research are DNA nanomachines. These are synthetic DNA assemblies that switch between defined molecular shapes upon stimulation by external triggers. They can be controlled by a variety of methods that include pH changes and the addition of other molecular components, such as small molecule effectors, proteins and DNA strands. Researchers have now designed and built a simple DNA machine that is capable of continuous rotation with controlled speed and direction - a function that might be very useful for example for molecular transport. This machine is driven by an externally controlled electric field. When this field is oscillated between four directions, it continuously reorients a rotor DNA that is asymmetrically attached to a DNA axle.
Jan 12th, 2010
 One challenge in designing nanomachines is being able to establish how well they work and optimize their performance. This is where single molecule techniques will play an important role. With advances in nanotechnologies, it is possible to construct simple nanomachines that can perform simple functions such as opening and closing of a DNA device (e.g. DNA tweezers or DNA switches), small rotational and translational motors and energy transfer cascades. Using single-molecule techniques researchers can watch individual nanomachines working and determine the functionality of their design. Researchers in Germany now have incorporated optical addressability to these nanomachines. Hence, they can optically detect and eventually control the state of the nanodevice.
One challenge in designing nanomachines is being able to establish how well they work and optimize their performance. This is where single molecule techniques will play an important role. With advances in nanotechnologies, it is possible to construct simple nanomachines that can perform simple functions such as opening and closing of a DNA device (e.g. DNA tweezers or DNA switches), small rotational and translational motors and energy transfer cascades. Using single-molecule techniques researchers can watch individual nanomachines working and determine the functionality of their design. Researchers in Germany now have incorporated optical addressability to these nanomachines. Hence, they can optically detect and eventually control the state of the nanodevice.
Dec 23rd, 2009
 Much of today's genetic research and diagnostics uses tools and technologies enabled by DNA's ability to bind to its complementary strand in a sequence specific manner. For biologists studying molecular mechanisms inside cells, this information helps to quantify the expression levels of genes. Detection of the binding - or hybridization - of DNA strands is at the heart of modern medicine. The technology for detecting DNA hybridization mainly relies on the use of fluorescent labels. The complementary strand coming from the sample bears a label, so detection of florescence signal indicates hybridization. While this may sound straightforward, it has major limitations. Researchers have now reported a new technique for genetic analysis using nanomechanical response of hybridized DNA/RNA molecules. This new technique is several orders of magnitude more sensitive than other approaches and it is a lot simpler to use.
Much of today's genetic research and diagnostics uses tools and technologies enabled by DNA's ability to bind to its complementary strand in a sequence specific manner. For biologists studying molecular mechanisms inside cells, this information helps to quantify the expression levels of genes. Detection of the binding - or hybridization - of DNA strands is at the heart of modern medicine. The technology for detecting DNA hybridization mainly relies on the use of fluorescent labels. The complementary strand coming from the sample bears a label, so detection of florescence signal indicates hybridization. While this may sound straightforward, it has major limitations. Researchers have now reported a new technique for genetic analysis using nanomechanical response of hybridized DNA/RNA molecules. This new technique is several orders of magnitude more sensitive than other approaches and it is a lot simpler to use.
Dec 21st, 2009
 DNA origami is a design technique that is used by nanotechnology researchers to fold DNA strands into something resembling a programmable pegboard on which different nanocomponents can be attached. These DNA assemblies allow the bottom-up fabrication of complex nanostructures with arbitrary shapes and patterns on a 100 nm scale. For instance, DNA origami have been heralded as a potential breakthrough for the creation of nanoscale circuits and devices. DNA can also be metallized with different metals, resulting in conducting nanowires. Researchers have now have developed a method to assemble metallic nanocircuits with arbitrary shapes, by attaching metallic nanoparticles to select locations of the DNA origami and then fusing them to form wires, rings, or any other complex shape. These pre-designed structures are programmed by fully utilizing the self-assembling and recognition properties of DNA.
DNA origami is a design technique that is used by nanotechnology researchers to fold DNA strands into something resembling a programmable pegboard on which different nanocomponents can be attached. These DNA assemblies allow the bottom-up fabrication of complex nanostructures with arbitrary shapes and patterns on a 100 nm scale. For instance, DNA origami have been heralded as a potential breakthrough for the creation of nanoscale circuits and devices. DNA can also be metallized with different metals, resulting in conducting nanowires. Researchers have now have developed a method to assemble metallic nanocircuits with arbitrary shapes, by attaching metallic nanoparticles to select locations of the DNA origami and then fusing them to form wires, rings, or any other complex shape. These pre-designed structures are programmed by fully utilizing the self-assembling and recognition properties of DNA.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





