| Posted: May 20, 2014 | |
Nanotechnology in food quality control: Preventing food from biting back (page 4 of 4) |
|
| Modified NPs as sensors can be used to create e-nose and tongues (based on MOSFET-metal oxide semiconductor field effect transistor technology) in artificial systems that mimic the natural olfactory sense and provide an aromatic response to detect odours and tastes in food. | |
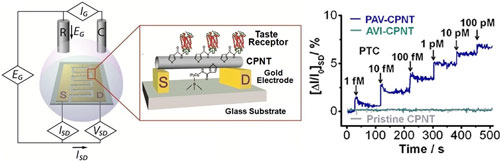
| Tai Hyun Park and his team at Seoul National University, South Korea have developed a nano-bioelectronic tongue using a human bitter taste receptor protein as a sensing element and conducting polypyrrole nanotube FET sensor platform with detection limit of 1 fM that has high selectivity and human tongue-like properties. This device proves to have a prominent potential in food and beverages industry for flavour testing (Fig. 5)28. | |
 |
|
| Fig. 5: Fabrication of nano-bioelectronic tongue. Left: FET sensor with human bitter taste receptor and conducting polypyrrole nanotube (CPNT). Right: Bitterness perception of taster type (PAV) and non-taster type (AVI) receptors in FET sensor. (Reprinted with permission from American Chemical Society, H.S. Song, et al., Nano Lett., 13(1) (2013) 172-178). (click image to enlarge) | |
| An olfactory-nanovesicle-fused CNT-transistor biosensor has been developed by a team of scientists at Seoul National University, South Korea that mimics a canine nose for detecting an indicator of the food oxidation; hexanal. This electronic nose can detect spoiled milk at 1 fM sensitivity in real-time without any pretreatment processes29. | |
| Nosang Myung, a professor at University of California, Riverside has developed CNTs-based ‘electronic nose’ nanosensors to detect specific volatile organic compounds in food supply chain. This research innovation is in the process of being commercialized in collaboration with US-based company; ieCrowd (web: http://iecrowd.com) and will further be customized for the food safety and potency measurements30. | |
| Chemists at Johannes Gutenberg University, Germany have made test strips bearing aptamer functionalized Au nanorods as sensor elements with nanomolar sensitivity to detect numerous proteins/toxins in the food simultaneously, particularly in milk or baby food. When a protein docks with one of the aptamers, the corresponding nanorod changes its color, which can be detected by a spectrometer and sensor recycling is also possible31. In addition to these specialized devices, CNT-array-based sensor chips fabricated via NEMS (nano-electro mechanical system) technique are also used to miniaturize the ELISA test in various food analysis systems with enhanced molecular recognition and signal transduction. | |
| Outlook | |
| The development of novel analytical methods for food industry is among the key application fields for nanotechnology, which is not only beneficial for the consumers to purchase products at the peak of freshness and flavour but also for industry stakeholders to keep market leadership and help food regulatory authorities to reduce the frequency of food-borne illness. | |
| Nano-enabled analytical methods are expected to meet less consumer resistance than other nanotechnology applications (e.g. packaging materials, food ingredients, etc.) in the food industry due to their limited interaction with food. | |
| Many technological challenges such as stability under variable conditions, improved signal conduction and processing remain to be addressed before nano-enabled sensing platforms are commercialized and compete with existing technologies effectively. | |
| However, regardless of how nano-enabled sensing products in the food sector are ultimately governed, marketed and perceived by the public, it seems clear that nanotechnology will continue to play an important role in making food supply safer, healthier and more prolific. | |
| References | |
| 1. Nanomaterials for Biosensors, Challa S. S. R. Kumar, John Wiley & Sons, (2007) | |
| 2. T.V. Duncan, J. Colloid Interface Sci., (2011) doi:10.1016/j.jcis.2011.07.017 | |
| 3. Suman, V. Gaur, et al., Journal of Experimental Nanoscience, 7(6) (2011) 608-615 | |
| 4. N.A. Luechinger, S. Loher, E.K. Athanassiou, et al., Langmuir, 23 (2007) 3473-3477 | |
| 5. H.Tao, M.A. Brenckle, M. Yang, et al., Adv. Mater., (24) (2012) 1067-1072 | |
| 6. B. Esser, J.M. Schnorr, and T.M. Swager, Angew. Chem. Int. Ed., (51) (2012) 5752 -5756 | |
| 7. Photonic gels are colorful sensors | |
| 8. Thinfilm and PST Sensors to Jointly Develop Printed Temperature Tags to Monitor Food and Other Perishable Goods | |
| 9. Thinfilm Demonstrates First Integrated Printed Electronic System with Rewritable Memory | |
| 10. Biopesticides: The Global Market | |
| 11. Detecting Pesticides using Nano-oxide particles | |
| 12. B. Liu, P. Zhou, X. Liu, et al., Food and Bioprocess Technology, 6(3) (2013) 710-718 | |
| 13. A. Kim and Z. Li, HP Laboratories, HPL-2011-72, http://www.hpl.hp.com/techreports/2011/HPL-2011-72.html | |
| 14. H. Ping, M. Zhang, H. Li, et al., Food Control, 23 (2012) 191-197 | |
| 15. D. Zhai, B. Liu, Y. Shi, et al., ACS Nano, 7(4) (2013) 3540-3546 | |
| 16. M.E. Ali, S. Mustafa, U. Hashim, et al., Journal of Nanomaterials, (2012), Article ID 832387 | |
| 17. P. Chu, C. Lin, W. Chen, et al., J. Agric. Food Chem., 60 (2012) 6483-6492 | |
| 18. Y. Zhang, X. Zhang, X. Lu, et al., Food Chemistry, 122 (2010) 909-913 | |
| 19. M. Chen, X. Ma, X. Li, Turkish Journal of Chemistry, 37 (2013) 959-965 | |
| 20. L.S. Selvakumar, K.V. Ragavan, K.S. Abhijith, et al., Anal. Methods, 5 (2013) 1806-1810 | |
| 21. Nanosensor Based Determination of Total Cholesterol in Food Samples | |
| 22. Pathogen and Toxin Detection | |
| 23. X. Wang, R.J. Meier, and O.S. Wolfbeis, Angew. Chem. Int. Ed., 52(1) (2013) 406 -409 | |
| 24. Y. Yang, F. Xu, H. Xu, et al., Food Microbiol., 34(2) (2013) 418-424 | |
| 25. H. Kim, S. Kim, J. Lee, et al., J Vet Sci., 13(4) (2012) 363-369 | |
| 26. J. Xu, J.W. Turner, M. Idso, et al., Anal. Chem., 85(5) (2013) 2630-2637 | |
| 27. H. Zhu, U. Sikora and A. Ozcan, Analyst, 137 (2012) 2541-2544 | |
| 28. H.S. Song, O.S. Kwon, S.H. Lee, et al., Nano Lett., 13(1) (2013) 172-178 | |
| 29. J. Park, J.H. Lim, H.J. Jin, et al., Analyst, 137 (2012) 3249-3254 | |
| 30. New 'electronic nose' nanotechnology sensor being developed for food safety, health | |
| 31. C. Rosman, J. Prasad, A. Neiser, et al., Nano Lett., 13(7) (2013) 3243-3247 | |
|
By Abhilasha Verma , (Email: [email protected]),Centre for Knowledge Management of Nanoscience and Technology (CKMNT). The full article has appeared in the January 2014 issue of "Nanotech Insights" and the above article is an abridged and revised version of the same.
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
