| Posted: Apr 04, 2008 | |
A role for nanotechnology in capturing and storing greenhouse gases |
|
| (Nanowerk Spotlight) The greenhouse effect is primarily a function of the concentration of water vapor, carbon dioxide, and other trace gases in the Earth's atmosphere that absorb the terrestrial radiation leaving the surface of the Earth. Changes in the atmospheric concentrations of these greenhouse gases can alter the balance of energy transfers between the atmosphere, space, land, and the oceans. The capture and storage of greenhouse gases could play a significant role in reducing the release of greenhouse gases into the atmosphere is the most important greenhouse gas and captures the limelight in most reports on global warming. While other greenhouse gases make up less of the atmosphere, they account for about 40 percent of the greenhouse gas radiation sent back to Earth. They can also be much more efficient at absorbing and re-emitting radiation than carbon dioxide, so they are small but important elements in the equation. In fact, molecule-for-molecule some gases containing lots of fluorine are 10,000 times stronger at absorbing radiation than carbon dioxide. A new systematic computational study shows an interesting approach of how nanotechnology, in this case the use of carbon nanotubes and other nanomaterials, could lead to effective filters for the capture and storage of greenhouse gases. | |
| Tetrafluoromethane (CF4), an extremely stable molecule whose lifetime in the atmosphere is 50 000 years (compared to an average CO2 lifetime of 50-200 years), is such a particularly powerful greenhouse gas – also called super greenhouse gas – that, when present in the troposphere, has a particular ability to absorb the outgoing infrared radiation, thus causing a temperature increase of the atmosphere. | |
| The global warming potential (GWP) index is intended as a quantified measure of the globally averaged relative radiative forcing impacts of a particular greenhouse gas. It is defined as the cumulative radiative forcing – both direct and indirect effects – integrated over a period of time from the emission of a unit mass of gas relative to CO2 as a reference gas. The GWP for carbon dioxide is 1 per hundred years while that for tetrafluoromethane is 6500 per hundred years. | |
| Total worldwide emissions of CF4, a perfluorocarbon (PFC) gas, are small in comparison to carbon dioxide but it is a much more efficient absorber of infrared radiation than CO2 and therefore its global warming potential is vastly higher. PFCs have been extensively used in the microelectronic and semiconductor industry in plasma cleaning of chemical vapor deposition chambers. Although the semiconductor industry is moving away from the use of PFCs toward other less problematic gases, other sources of PFCs are still significant: as an unintended byproduct during aluminum production; as drop-in replacement of chlorofluorocarbon refrigerants; as potential solvents and cosolvents for supercritical fluid extraction processes; and in the petrochemical industry. New uses for PFCs are being explored, e.g. for therapeutic purposes (e.g., NMR imaging), eye surgery, and modifiers for inhaled anesthetics. As a consequence, it is estimated that global emissions of PFCs will rise 150% in the next 50 years. That means that super greenhouse gases even with small emissions have the potential to influence climate far into the future and could become a serious environmental problem. | |
| "We have conducted simulations that show that single-walled carbon nanotubes (SWCNTs) can serve as efficient nanoscale vessels for encapsulation of tetrafluoromethane at room temperature" Dr. Robert Holyst tells Nanowerk. "A good filter should keep the CF4 molecules inside and stay cheap. Carbon seems promising, but in order to efficiently store CF4 we need well defined pores inside the carbon material. Carbon nanotubes are a carbon material that offers well defined pore sizes for storage applications and they are also most efficient in terms of storage capacity." | |
 |
|
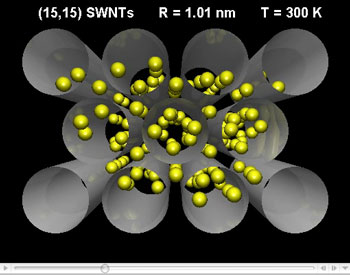
| This movie shows a Monte Carlo simulations of CF4 adsorption in carbon nanotubes with pore sizes of 1.01 nm) | |
| Holyst, a professor in the Institute of Physical Chemistry at the Polish Academy of Sciences, together with Dr. Piotr Kowalczyk, a Postdoctoral Research Fellow in the Department of Applied Physics at RMIT University in Australia, has published his findings in the March 7, 2008 online edition of Environmental Science & Technology (Efficient Adsorption of Super Greenhouse Gas (Tetrafluoromethane) in Carbon Nanotubes). The two scientists have found that the amount of the encapsulated CF4 under the ambient external conditions (1 bar, 300 K) is maximized for well defined pore sizes of SWCNTs. These pore sizes change as we change the external pressure. They also demonstrate that the high enthalpy of adsorption cannot be used as an only measure of storage efficiency. | |
| "We found that the optimal balance between the binding energy (i.e., enthalpy of adsorption) and space available for the accommodation of molecules (i.e., presence of inaccessible pore volume) is a key for encapsulation of van der Waals molecules (a stable cluster consisting of two or more molecules held together by van der Waals forces or by hydrogen bonds)" says Holyst. | |
| He explains that carbon nanotubes can have a very narrow distribution of pore sizes, which is particularly important in view of the results predicting a maximum adsorption at some pore sizes. "In carbon nanotubes we can highly compress the gas reaching the density of a solid phase. We also point out that optimal structure of carbon nanotubes for volumetric storage capacity is different from the structure for the optimal mass storage capacity, thus it is important whether we consider optimal adsorbent for mass or for volumetric storage." | |
| In their models the two researchers show that in the search for optimal adsorbents one has to take into account two elements: heat of adsorption and pore sizes, since it is not true as is commonly believed that high adsorption enthalpy is the sole condition for high adsorption capacity. | |
| "The key for optimizing the amount of CF4 trapped in the nanotubes upon assumed operating external conditions is the size of the internal cylindrical pores and interstitials channels of an idealized bundle of SWCNTs" explains Kowalczyk. "Due to a large molecular size of CF4, the internal pores play predominant role in the process of encapsulation of CF4 via the physical adsorption mechanism." | |
| This work shows that an optimized structure of SWCNTs bundles seem to be very promising for the encapsulation of CF4 and superior in comparison to the currently used activated carbons and zeolites. The efficiency of encapsulation in nanotubes can be explained by their intermediate properties in comparison to these other materials. | |
| Holyst raises the point that, in practice, CF4 exists as a gas mixture (for example, a mixture with nitrogen that can mimic the air mixture). "So the question arises about the transferability of our simulation results to the selective adsorption of CF4 from a gas mixture" he says. "Previous studies suggest that our current simulation results of CF4 adsorption in carbon nanotubes are transferable for the problem of CF4-N2 mixture adsorption. Our results as well as those previous results show that optimal adsorption is achieved only when the distribution of pore sizes is sharp." | |
| Based on these model simulations, what is now needed are experimental investigations of the capture and storage of CF4 in a real bundle of SWCNTs in order to develop this concept as a practical solution that works on an industrial scale. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
