Showing Spotlights 969 - 976 of 2786 in category All (newest first):
 Metal nanoparticles, when excited at optical frequencies, may experience localized surface plasmon resonances, which determine enhanced local electric fields, increased scattering cross sections, and high sensitivity to the environment refractive index. Thanks to these unique properties, they are widely utilized especially in biomedical sciences and engineering. Researchers have now conceived and demonstrated a new method to fully automate the design of metal nanoparticles.
Metal nanoparticles, when excited at optical frequencies, may experience localized surface plasmon resonances, which determine enhanced local electric fields, increased scattering cross sections, and high sensitivity to the environment refractive index. Thanks to these unique properties, they are widely utilized especially in biomedical sciences and engineering. Researchers have now conceived and demonstrated a new method to fully automate the design of metal nanoparticles.
Jan 7th, 2016
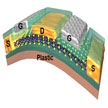 In the past, the performance of synthesized MoS2 had been poor, especially when integrated on flexible substrates. A new study have now yielded the highest performance for CVD-grown monolayer MoS2 device properties on flexible substrates to date. MoS2 exhibits unique physical, optical and electrical properties correlated with its single-layer atomic layer structure. Important for electronics applications, and in contrast to graphene, MoS2 has a bandgap.
In the past, the performance of synthesized MoS2 had been poor, especially when integrated on flexible substrates. A new study have now yielded the highest performance for CVD-grown monolayer MoS2 device properties on flexible substrates to date. MoS2 exhibits unique physical, optical and electrical properties correlated with its single-layer atomic layer structure. Important for electronics applications, and in contrast to graphene, MoS2 has a bandgap.
Jan 6th, 2016
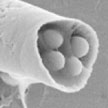 The goal of a vast amount of nanomedicine research is the perfect drug carrier: it is injected into the body and transports itself to the correct target, such as a tumor, and delivers the required therapeutic drug dose at this target. This idealized concept was first proposed at the beginning of the 20th century and was nicknamed the 'magic bullet' concept. Taking this 'bullet' concept literally, researchers have developed acoustically triggered microcannons, capable of versatile loading and effective firing of nanobullets, as novel tools toward advancing microscale tissue penetration of therapeutic payloads.
The goal of a vast amount of nanomedicine research is the perfect drug carrier: it is injected into the body and transports itself to the correct target, such as a tumor, and delivers the required therapeutic drug dose at this target. This idealized concept was first proposed at the beginning of the 20th century and was nicknamed the 'magic bullet' concept. Taking this 'bullet' concept literally, researchers have developed acoustically triggered microcannons, capable of versatile loading and effective firing of nanobullets, as novel tools toward advancing microscale tissue penetration of therapeutic payloads.
Jan 5th, 2016
 Engineered nanoparticles are being used in a wide range of product areas, including composite materials, coatings, electronics, food, agriculture, cosmetics, healthcare, and biotechnology. As a consequence, human exposure to nanoparticles has become a prominent environmental concern; especially since these potential pollutants are not visible to the human eye or detectable by smell. However, there is no current technology that provides rapid, sensitive and highly portable detection and identification of nanoparticles. Now though, researchers have developed a simple colorimetric sensor array approach capable of detection and unambiguous differentiation of a wide range of nanoparticles in aqueous solutions.
Engineered nanoparticles are being used in a wide range of product areas, including composite materials, coatings, electronics, food, agriculture, cosmetics, healthcare, and biotechnology. As a consequence, human exposure to nanoparticles has become a prominent environmental concern; especially since these potential pollutants are not visible to the human eye or detectable by smell. However, there is no current technology that provides rapid, sensitive and highly portable detection and identification of nanoparticles. Now though, researchers have developed a simple colorimetric sensor array approach capable of detection and unambiguous differentiation of a wide range of nanoparticles in aqueous solutions.
Jan 4th, 2016
 Here are the 10 most popular Nanowerk Nanotechnology Spotlight articles of 2015. This year, the list includes a quick and simple blood test to detect early-stage cancer; self-powered smart suits; nanomaterials for camouflage and stealth applications; nanotechnology energy applications; 3D-printing with graphene; fuzzy and Boolean logic gates based on DNA nanotechnology; a path towards self-powered electronic papers; a look at whether nanomedicine lhas ived up to its promise; smart materials that become 'alive' with living bacteria in supramolecular assemblies; and repair nanobots on damage patrol.
Here are the 10 most popular Nanowerk Nanotechnology Spotlight articles of 2015. This year, the list includes a quick and simple blood test to detect early-stage cancer; self-powered smart suits; nanomaterials for camouflage and stealth applications; nanotechnology energy applications; 3D-printing with graphene; fuzzy and Boolean logic gates based on DNA nanotechnology; a path towards self-powered electronic papers; a look at whether nanomedicine lhas ived up to its promise; smart materials that become 'alive' with living bacteria in supramolecular assemblies; and repair nanobots on damage patrol.
Jan 1st, 2016
 Researchers have developed a suspended planar-array chip whose in situ capabilities with a spatial molecular-probe arrangement combine the advantages of both suspended arrays and planar arrays. This opens the way towards the multiplexed detection of intracellular biological parameters using a single device in dramatically reduced volumes, such as inside a living HeLa cell. The chip's volume represents only about 0.35% of the total volume of a typical HeLa cell.
Researchers have developed a suspended planar-array chip whose in situ capabilities with a spatial molecular-probe arrangement combine the advantages of both suspended arrays and planar arrays. This opens the way towards the multiplexed detection of intracellular biological parameters using a single device in dramatically reduced volumes, such as inside a living HeLa cell. The chip's volume represents only about 0.35% of the total volume of a typical HeLa cell.
Dec 29th, 2015
 Current research on tactile sensors is mostly focused on the improvement of sensitivity and multi-functionality to emulate the function of natural skin. However, natural skin can sense external pressure and help form haptic memory, while current flexible tactile sensors for electronic skin can only perform sensing functions. This functionality gap between state-of-the-art tactile sensing devices and natural skin inspired a team of researchers to develop haptic memory devices that integrate sensor and memory functions.
Current research on tactile sensors is mostly focused on the improvement of sensitivity and multi-functionality to emulate the function of natural skin. However, natural skin can sense external pressure and help form haptic memory, while current flexible tactile sensors for electronic skin can only perform sensing functions. This functionality gap between state-of-the-art tactile sensing devices and natural skin inspired a team of researchers to develop haptic memory devices that integrate sensor and memory functions.
Dec 23rd, 2015
 Bubble-pen lithography (BPL) is a novel optically controlled nanofabrication technique that can be widely applied to pattern colloidal and biological particles on substrates in order to build functional optic, electronic, and magnetic devices. In BPL, an optically controlled microbubble is generated to capture and immobilize colloidal particles on the plasmonic substrates. With this new lithographic technique, the researchers can generate bubbles down to 1 micron in diameter. The smaller bubbles provide an enhanced patterning resolution.
Bubble-pen lithography (BPL) is a novel optically controlled nanofabrication technique that can be widely applied to pattern colloidal and biological particles on substrates in order to build functional optic, electronic, and magnetic devices. In BPL, an optically controlled microbubble is generated to capture and immobilize colloidal particles on the plasmonic substrates. With this new lithographic technique, the researchers can generate bubbles down to 1 micron in diameter. The smaller bubbles provide an enhanced patterning resolution.
Dec 22nd, 2015
 Metal nanoparticles, when excited at optical frequencies, may experience localized surface plasmon resonances, which determine enhanced local electric fields, increased scattering cross sections, and high sensitivity to the environment refractive index. Thanks to these unique properties, they are widely utilized especially in biomedical sciences and engineering. Researchers have now conceived and demonstrated a new method to fully automate the design of metal nanoparticles.
Metal nanoparticles, when excited at optical frequencies, may experience localized surface plasmon resonances, which determine enhanced local electric fields, increased scattering cross sections, and high sensitivity to the environment refractive index. Thanks to these unique properties, they are widely utilized especially in biomedical sciences and engineering. Researchers have now conceived and demonstrated a new method to fully automate the design of metal nanoparticles.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





