Showing Spotlights 25 - 32 of 203 in category All (newest first):
 EV manufacturing requires more energy and produces more emissions than manufacturing a conventional car because of batteries. There are many unanswered questions about the life-cycle greenhouse gas implications of electric vehicles, especially related to early estimates of battery production emissions. A new review addresses these questions and the advances in new battery chemistries, plus new insights into CO2 emissions from battery manufacturing.
EV manufacturing requires more energy and produces more emissions than manufacturing a conventional car because of batteries. There are many unanswered questions about the life-cycle greenhouse gas implications of electric vehicles, especially related to early estimates of battery production emissions. A new review addresses these questions and the advances in new battery chemistries, plus new insights into CO2 emissions from battery manufacturing.
Dec 1st, 2021
 Researchers demonstrated that two dissimilar materials - molecular energetic materials and ferroelectrics - can be combined to obtain a chemically driven electrical energy source with high-power density. They obtained chemically driven electrical energy with a high specific power of 1.8 kW/kg and achieve an estimated detonation velocity comparable to trinitrotoluene (TNT) and hexanitrostilbene (HNS). Such a power source could potentially be employed for on-demand energy sources, propulsion, or thermal batteries.
Researchers demonstrated that two dissimilar materials - molecular energetic materials and ferroelectrics - can be combined to obtain a chemically driven electrical energy source with high-power density. They obtained chemically driven electrical energy with a high specific power of 1.8 kW/kg and achieve an estimated detonation velocity comparable to trinitrotoluene (TNT) and hexanitrostilbene (HNS). Such a power source could potentially be employed for on-demand energy sources, propulsion, or thermal batteries.
Oct 14th, 2021
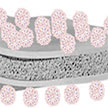 Lithium-sulfur (Li-S) batteries are a promising alternative to lithium-ion batteries because - at least theoretically - they can render 3-6 times higher energy density. In developing Li-S battery technology, researchers have borrowed many components from the mature lithium-ion battery, such as the separator. However, because the working mechanism of Li-S cells is fundamentally different, there is a need for a suitable separator specifically designed for Li-S. Re-engineering the separator can improve the energy density of Li-S batteries.
Lithium-sulfur (Li-S) batteries are a promising alternative to lithium-ion batteries because - at least theoretically - they can render 3-6 times higher energy density. In developing Li-S battery technology, researchers have borrowed many components from the mature lithium-ion battery, such as the separator. However, because the working mechanism of Li-S cells is fundamentally different, there is a need for a suitable separator specifically designed for Li-S. Re-engineering the separator can improve the energy density of Li-S batteries.
Aug 5th, 2021
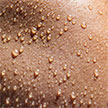 Self-charging biosupercapacitors (BSCs) that can store energy and be self-charged via chemical or solar energy conversion through bioreaction have recently attracted considerable attention. As human sweat also contains a high concentration of lactate biofuel, the harvesting and storage of the bioenergy in sweat holds the potential to provide the power for wearable electronics. A new wearable hybrid device functions as both a biofuel cell and a supercapacitor.
Self-charging biosupercapacitors (BSCs) that can store energy and be self-charged via chemical or solar energy conversion through bioreaction have recently attracted considerable attention. As human sweat also contains a high concentration of lactate biofuel, the harvesting and storage of the bioenergy in sweat holds the potential to provide the power for wearable electronics. A new wearable hybrid device functions as both a biofuel cell and a supercapacitor.
Jul 8th, 2021
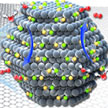 Scientists have used metal carbides as the carrier to support the transition metal Fe and Ni atoms to engineer single-atom oxygen evolution catalysts for the first time. Distinct from previous studies, this novel metal carbide support shows obvious advantages for supporting different single atoms, especially its non-strong bonding features, which resulted in high mobility of the supported atoms, which might be the key point for realizing excellent OER activities.
Scientists have used metal carbides as the carrier to support the transition metal Fe and Ni atoms to engineer single-atom oxygen evolution catalysts for the first time. Distinct from previous studies, this novel metal carbide support shows obvious advantages for supporting different single atoms, especially its non-strong bonding features, which resulted in high mobility of the supported atoms, which might be the key point for realizing excellent OER activities.
Jul 2nd, 2021
 Researchers report an in vitro and in vivo proof-of-concept for the capacity of triboelectric nanogenerator (TENG) technology to function as a simple, scalable, inexpensive, and self-powered device for tactile sensory restoration. This integrated tactile sensory restoration device powers itself and is suitable for implantation. It bypasses damaged nerves and activate sensory neurons at various levels of electrical potential, generated by different levels of tactile pressure on the implanted device.
Researchers report an in vitro and in vivo proof-of-concept for the capacity of triboelectric nanogenerator (TENG) technology to function as a simple, scalable, inexpensive, and self-powered device for tactile sensory restoration. This integrated tactile sensory restoration device powers itself and is suitable for implantation. It bypasses damaged nerves and activate sensory neurons at various levels of electrical potential, generated by different levels of tactile pressure on the implanted device.
Jul 1st, 2021
 In general, there are three types of driving force for ion transport: electric field; mechanical pressure; and concentration gradient. Recently, light has been propsoed as a fourth. Now, researchers report the incorporation of a transition metal dichalcogenides based van der Waals multilayer heterostructure into nanofluidic materials, and demonstrate a new photo-induced active ion transport phenomenon. This will inspire a broad range of fundamental research and practical application for light-controlled ionic circuits, artificial photosynthesis, biomimetic energy conversion, and so on.
In general, there are three types of driving force for ion transport: electric field; mechanical pressure; and concentration gradient. Recently, light has been propsoed as a fourth. Now, researchers report the incorporation of a transition metal dichalcogenides based van der Waals multilayer heterostructure into nanofluidic materials, and demonstrate a new photo-induced active ion transport phenomenon. This will inspire a broad range of fundamental research and practical application for light-controlled ionic circuits, artificial photosynthesis, biomimetic energy conversion, and so on.
Mar 4th, 2021
 A major challenge in the development of implantable and ingestible biomedical electronic devices is the limited lifetime of their power sources. The energy requirements of these devices are highly dependent on their application and the complexity of the required electrical systems. The power unit, which is composed of one or more energy sources - batteries, energy-harvesting, and energy transfer - as well as power management circuits, supplies electrical energy to the whole system.
A major challenge in the development of implantable and ingestible biomedical electronic devices is the limited lifetime of their power sources. The energy requirements of these devices are highly dependent on their application and the complexity of the required electrical systems. The power unit, which is composed of one or more energy sources - batteries, energy-harvesting, and energy transfer - as well as power management circuits, supplies electrical energy to the whole system.
Feb 16th, 2021
 EV manufacturing requires more energy and produces more emissions than manufacturing a conventional car because of batteries. There are many unanswered questions about the life-cycle greenhouse gas implications of electric vehicles, especially related to early estimates of battery production emissions. A new review addresses these questions and the advances in new battery chemistries, plus new insights into CO2 emissions from battery manufacturing.
EV manufacturing requires more energy and produces more emissions than manufacturing a conventional car because of batteries. There are many unanswered questions about the life-cycle greenhouse gas implications of electric vehicles, especially related to early estimates of battery production emissions. A new review addresses these questions and the advances in new battery chemistries, plus new insights into CO2 emissions from battery manufacturing. 
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





