Showing Spotlights 65 - 72 of 203 in category All (newest first):
 So far, most of the developed self-powered piezoelectric devices are rigid or have limited lateral stretchability and could not be used to harvest energy from lateral strain, which greatly limits their applications on large strain deformation. In new work, researchers have successfully fabricated a piezoelectric nanocomposite device with good transparency, high stretchability, and self-powered sensing characteristics. Attached to the human body, it can harvest biomechanical energy and monitor physiological signals.
So far, most of the developed self-powered piezoelectric devices are rigid or have limited lateral stretchability and could not be used to harvest energy from lateral strain, which greatly limits their applications on large strain deformation. In new work, researchers have successfully fabricated a piezoelectric nanocomposite device with good transparency, high stretchability, and self-powered sensing characteristics. Attached to the human body, it can harvest biomechanical energy and monitor physiological signals.
Dec 4th, 2017
 The wearable power sources required for wearable and implantable electronic devices are limited by the size of the gadgets they power. Microsupercapacitors are newly emerging miniaturized high-power microelectrochemical energy-storage devices that can deliver high power density, fast charge and discharge, and a superior cycling lifetime. A new study shows that electrode fractal design is a viable strategy for improving the performance of integrated microsupercapacitors that use thin-film electrodes at no extra processing or fabrication cost.
The wearable power sources required for wearable and implantable electronic devices are limited by the size of the gadgets they power. Microsupercapacitors are newly emerging miniaturized high-power microelectrochemical energy-storage devices that can deliver high power density, fast charge and discharge, and a superior cycling lifetime. A new study shows that electrode fractal design is a viable strategy for improving the performance of integrated microsupercapacitors that use thin-film electrodes at no extra processing or fabrication cost.
Nov 20th, 2017
 Self-powered nanotechnology based on one type of nanogenerators - piezoelectric nanogenerators - aims at powering nanodevices and nanosystems using the energy harvested from the environment in which these systems are suppose to operate. This offers a completely new approach for harvesting mechanical energy using organic and inorganic materials. Researchers have now reported a novel bio-piezoelectric nanogenerator using naturally abundant, self-aligned cellulose fibrous untreated onion skin as efficient piezoelectric material.
Self-powered nanotechnology based on one type of nanogenerators - piezoelectric nanogenerators - aims at powering nanodevices and nanosystems using the energy harvested from the environment in which these systems are suppose to operate. This offers a completely new approach for harvesting mechanical energy using organic and inorganic materials. Researchers have now reported a novel bio-piezoelectric nanogenerator using naturally abundant, self-aligned cellulose fibrous untreated onion skin as efficient piezoelectric material.
Nov 15th, 2017
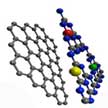 Efficient electrocatalysts lie at the heart of a series of significant energy conversion and storage technologies, and atomically precise understanding of the influences of component dopants is crucial for looking into the reaction mechanism and controlled synthesis of the desired electrocatalysts. Graphitic carbon nitride is a promising electrocatalytic material owing to its intrinsically high N content and abundant edge sites. This material has been researched towards some of the most significant electrocatalytic reactions including oxygen reduction/evolution reaction and hydro evolution reaction. New work has comprehensively explored the influences of component elements within graphitic carbon nitride motiety for electrocatalytic reactions.
Efficient electrocatalysts lie at the heart of a series of significant energy conversion and storage technologies, and atomically precise understanding of the influences of component dopants is crucial for looking into the reaction mechanism and controlled synthesis of the desired electrocatalysts. Graphitic carbon nitride is a promising electrocatalytic material owing to its intrinsically high N content and abundant edge sites. This material has been researched towards some of the most significant electrocatalytic reactions including oxygen reduction/evolution reaction and hydro evolution reaction. New work has comprehensively explored the influences of component elements within graphitic carbon nitride motiety for electrocatalytic reactions.
Nov 13th, 2017
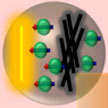 Chemical engineering researchers have reported the usage of activated carbon prepared from tea leaves, improving the mass transport phenomenon (33 % performance improvement) in an operating direct methanol fuel cell, owing to its pore structure characteristics. The cell performance underwent drastic changes in the mass transport region of the fuel cell polarization curve, comparable to the standard membrane electrode assembly. This is attributed to the pore structure of this framework aiding in enhanced water removal, as a result more air molecules react with the platinum catalyst sites finally improving the fuel cell performance.
Chemical engineering researchers have reported the usage of activated carbon prepared from tea leaves, improving the mass transport phenomenon (33 % performance improvement) in an operating direct methanol fuel cell, owing to its pore structure characteristics. The cell performance underwent drastic changes in the mass transport region of the fuel cell polarization curve, comparable to the standard membrane electrode assembly. This is attributed to the pore structure of this framework aiding in enhanced water removal, as a result more air molecules react with the platinum catalyst sites finally improving the fuel cell performance.
Oct 3rd, 2017
 Electrocatalysis offers important opportunities for clean fuel production, but uncovering the chemistry at the electrode surface remains a challenge. Here, this work exploits a single-nanosheet device to perform in-situ measurements of water oxidation electrocatalysis and reveal a crucial interaction with oxygen. The obtained in-depth understanding could provide valuable clues for catalysis system design and the in-situ measurement could be also useful to analyze other interfacial reaction processes.
Electrocatalysis offers important opportunities for clean fuel production, but uncovering the chemistry at the electrode surface remains a challenge. Here, this work exploits a single-nanosheet device to perform in-situ measurements of water oxidation electrocatalysis and reveal a crucial interaction with oxygen. The obtained in-depth understanding could provide valuable clues for catalysis system design and the in-situ measurement could be also useful to analyze other interfacial reaction processes.
Sep 29th, 2017
 Two-dimensional (2D) energy materials have outstanding physical and chemical properties in contrast to their bulk counterparts. This is particularly true for charge storage devices such as lithium-ion batteries and supercapacitors. Unfortunately, when directly applying these 2D nanostructured materials for energy storage, there is still a significant challenge as they may have serious self-restacking leading to decreased active surface areas and sluggish ion transport kinetics. Researchers have now developed an effective interlayer engineering strategy to improve sodium ion transport in 2D nanosheets via controlled organic intercalation.
Two-dimensional (2D) energy materials have outstanding physical and chemical properties in contrast to their bulk counterparts. This is particularly true for charge storage devices such as lithium-ion batteries and supercapacitors. Unfortunately, when directly applying these 2D nanostructured materials for energy storage, there is still a significant challenge as they may have serious self-restacking leading to decreased active surface areas and sluggish ion transport kinetics. Researchers have now developed an effective interlayer engineering strategy to improve sodium ion transport in 2D nanosheets via controlled organic intercalation.
Sep 13th, 2017
 Sodium-ion battery, as an emerging battery technology beyond lithium-ion battery, has attracted great research interests recent years. Sodium-ion batteries have a similar configuration and electrochemical reaction processes with lithium-ion batteries. But the Na resources are much more abundant and cost-effective than Li resources, which makes sodium-ion batteries highly promising as next-generation energy storage devices, especially for large-scale energy storage. However, the practical application of Na-ion batteries is still not currently realized.
Sodium-ion battery, as an emerging battery technology beyond lithium-ion battery, has attracted great research interests recent years. Sodium-ion batteries have a similar configuration and electrochemical reaction processes with lithium-ion batteries. But the Na resources are much more abundant and cost-effective than Li resources, which makes sodium-ion batteries highly promising as next-generation energy storage devices, especially for large-scale energy storage. However, the practical application of Na-ion batteries is still not currently realized.
Sep 12th, 2017
 So far, most of the developed self-powered piezoelectric devices are rigid or have limited lateral stretchability and could not be used to harvest energy from lateral strain, which greatly limits their applications on large strain deformation. In new work, researchers have successfully fabricated a piezoelectric nanocomposite device with good transparency, high stretchability, and self-powered sensing characteristics. Attached to the human body, it can harvest biomechanical energy and monitor physiological signals.
So far, most of the developed self-powered piezoelectric devices are rigid or have limited lateral stretchability and could not be used to harvest energy from lateral strain, which greatly limits their applications on large strain deformation. In new work, researchers have successfully fabricated a piezoelectric nanocomposite device with good transparency, high stretchability, and self-powered sensing characteristics. Attached to the human body, it can harvest biomechanical energy and monitor physiological signals.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





