Showing Spotlights 49 - 56 of 203 in category All (newest first):
 Over the past ten years, paper has emerged as a focus area for researchers developing innovative techniques for printed basic electronics components. The goal of this research is to replace plastic substrates with low-cost, versatile and sustainable materials. The main advantages of investing in paper for electronics and energy storage devices are the low cost of the technology; the potentiality to recover device components and recycle the substrate as well as the active materials; and the production of environmentally harmless and biocompatible devices.
Over the past ten years, paper has emerged as a focus area for researchers developing innovative techniques for printed basic electronics components. The goal of this research is to replace plastic substrates with low-cost, versatile and sustainable materials. The main advantages of investing in paper for electronics and energy storage devices are the low cost of the technology; the potentiality to recover device components and recycle the substrate as well as the active materials; and the production of environmentally harmless and biocompatible devices.
Feb 7th, 2019
 Potassium-ion batteries (PIBs) have been considered as promising alternatives to lithium-ion batteries due to the rich natural abundance of potassium and similar redox potential with Li+/Li. Recently, researchers have designed and fabricated a novel antimony carbon composite PIB anode via a facile and scalable electrospray-assisted strategy and found that this anode delivered super high specific capacities as well as cycling stability in a highly concentrated electrolyte. These encouraging results will significantly promote the deep understanding of the fundamental electrochemistry in Potassium-ion batteries as well as rational development of efficient electrolyte systems for next generation high-performance Potassium-ion batteries.
Potassium-ion batteries (PIBs) have been considered as promising alternatives to lithium-ion batteries due to the rich natural abundance of potassium and similar redox potential with Li+/Li. Recently, researchers have designed and fabricated a novel antimony carbon composite PIB anode via a facile and scalable electrospray-assisted strategy and found that this anode delivered super high specific capacities as well as cycling stability in a highly concentrated electrolyte. These encouraging results will significantly promote the deep understanding of the fundamental electrochemistry in Potassium-ion batteries as well as rational development of efficient electrolyte systems for next generation high-performance Potassium-ion batteries.
Jan 21st, 2019
 By re-designing interconnected nanowire networks, researchers fabricated novel nanostructured current collectors, which are the first to combine very high surface area, high porosity, substantially large pores and mechanical flexibility. Thanks to its ordered microstructure, the material can uniquely combine high porosity of cellular metal foams with high surface area of state-of-the-art nanoporous dealloyed metals, which allowed it to surpass the electrochemical performance of up to 300-thicker commercial electrodes.
By re-designing interconnected nanowire networks, researchers fabricated novel nanostructured current collectors, which are the first to combine very high surface area, high porosity, substantially large pores and mechanical flexibility. Thanks to its ordered microstructure, the material can uniquely combine high porosity of cellular metal foams with high surface area of state-of-the-art nanoporous dealloyed metals, which allowed it to surpass the electrochemical performance of up to 300-thicker commercial electrodes.
Jan 18th, 2019
 One of the persistent challenges associated with electrode degradation and battery failure is the stress induced by ion intercalation, companied by large volume fluctuation of electrode materials. Although researchers have developed numerous in situ microscopy and spectrometry technologies that provided valuable insights into the impacts of the stress buildup and release, the strength of the localized stress at the micro- and nanoscale remains poorly understood. In new work, researchers have developed an in situ solution to quantitatively measure the localized stress on electrodes.
One of the persistent challenges associated with electrode degradation and battery failure is the stress induced by ion intercalation, companied by large volume fluctuation of electrode materials. Although researchers have developed numerous in situ microscopy and spectrometry technologies that provided valuable insights into the impacts of the stress buildup and release, the strength of the localized stress at the micro- and nanoscale remains poorly understood. In new work, researchers have developed an in situ solution to quantitatively measure the localized stress on electrodes.
Jan 10th, 2019
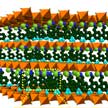 Due to the unique physical and chemical characteristics, such as good electrical conductivity, low weight, large surface areas, high electrochemical activity, and facile doping of heteroatoms or inorganic species, there are increasing demands of fabricating two-dimensional (2D) porous carbon for lithium-ion batteries, supercapacitors, electrochemical catalysts and many other emerged applications in energy storage and conversion. Researchers now have developed a fast, scalable, and eco-friendly active-salt-templating strategy to engineer different types of 2D porous carbons.
Due to the unique physical and chemical characteristics, such as good electrical conductivity, low weight, large surface areas, high electrochemical activity, and facile doping of heteroatoms or inorganic species, there are increasing demands of fabricating two-dimensional (2D) porous carbon for lithium-ion batteries, supercapacitors, electrochemical catalysts and many other emerged applications in energy storage and conversion. Researchers now have developed a fast, scalable, and eco-friendly active-salt-templating strategy to engineer different types of 2D porous carbons.
Dec 21st, 2018
 Recently, researchers have engineered a next-generation battery technology - known as metal-air batteries - which can be easily fabricated into flexible and wristband-like cells. Although metal-air batteries powered devices still are not ready yet for commercial applications, the current studies have established solid evidence that these devices have provided enormous opportunities to develop the next generation of flexible, wearable and bio-adaptable power sources. Researchers now report a solution to overcoming the current limitation on sluggish reaction kinetics of air cathodes in Mg-air batteries.
Recently, researchers have engineered a next-generation battery technology - known as metal-air batteries - which can be easily fabricated into flexible and wristband-like cells. Although metal-air batteries powered devices still are not ready yet for commercial applications, the current studies have established solid evidence that these devices have provided enormous opportunities to develop the next generation of flexible, wearable and bio-adaptable power sources. Researchers now report a solution to overcoming the current limitation on sluggish reaction kinetics of air cathodes in Mg-air batteries.
Nov 20th, 2018
 For the most part, advances in energy storage will rely on the continuing development of materials science, pushing the limits of performance of existing battery materials and developing entirely new battery structures and compositions. The battery industry is already working to reduce the cost of lithium-ion batteries, including by removing expensive cobalt from their positive electrodes. Researchers also are looking at the possibilities of using two-dimensional materials, essentially extremely thin sheets of substances with useful electronic properties.
For the most part, advances in energy storage will rely on the continuing development of materials science, pushing the limits of performance of existing battery materials and developing entirely new battery structures and compositions. The battery industry is already working to reduce the cost of lithium-ion batteries, including by removing expensive cobalt from their positive electrodes. Researchers also are looking at the possibilities of using two-dimensional materials, essentially extremely thin sheets of substances with useful electronic properties.
Oct 4th, 2018
 By combining advanced high-precision electron microscopy and computational methods, researchers have shed new light on the role of strain in nanoparticle catalysis. Catalytic metal nanoparticles supported on oxide substrates are used on a large scale in technological applications such as production of chemicals, abatement of air pollution and sustainable energy systems. The overall performance and durability of these systems is influenced by a range of factors, and every improvement could result in noticeable environmental and economic benefits.
By combining advanced high-precision electron microscopy and computational methods, researchers have shed new light on the role of strain in nanoparticle catalysis. Catalytic metal nanoparticles supported on oxide substrates are used on a large scale in technological applications such as production of chemicals, abatement of air pollution and sustainable energy systems. The overall performance and durability of these systems is influenced by a range of factors, and every improvement could result in noticeable environmental and economic benefits.
Jul 31st, 2018
 Over the past ten years, paper has emerged as a focus area for researchers developing innovative techniques for printed basic electronics components. The goal of this research is to replace plastic substrates with low-cost, versatile and sustainable materials. The main advantages of investing in paper for electronics and energy storage devices are the low cost of the technology; the potentiality to recover device components and recycle the substrate as well as the active materials; and the production of environmentally harmless and biocompatible devices.
Over the past ten years, paper has emerged as a focus area for researchers developing innovative techniques for printed basic electronics components. The goal of this research is to replace plastic substrates with low-cost, versatile and sustainable materials. The main advantages of investing in paper for electronics and energy storage devices are the low cost of the technology; the potentiality to recover device components and recycle the substrate as well as the active materials; and the production of environmentally harmless and biocompatible devices.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





