Showing Spotlights 41 - 48 of 203 in category All (newest first):
 Researchers have developed a comprehensive approach involving simple and facile steps to fabricate a wearable energy storage device based on carbon nanotube coated cotton yarn. All device components are flexible. According to the team, this is the first device that has been proven to be stable under rigorous washing conditions in the presence of hot water, detergents and high torque (spinning action of washing machine). This provides the device with comprehensive mechanical stability.
Researchers have developed a comprehensive approach involving simple and facile steps to fabricate a wearable energy storage device based on carbon nanotube coated cotton yarn. All device components are flexible. According to the team, this is the first device that has been proven to be stable under rigorous washing conditions in the presence of hot water, detergents and high torque (spinning action of washing machine). This provides the device with comprehensive mechanical stability.
May 15th, 2019
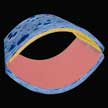 Researchers introduced an exclusive self-charging and self-healing flexible asymmetric supercapacitor power cell proficient to sustainably power-up portable electronics by simply applying mechanical bio-mechanical stresses. This self-charging power cell/package comprises an asymmetric supercapacitor unit sandwiched in-between two parallel connected triboelectric nanogenerators and reveals exceptional output performances. Additionally, this nanogenerator cell exhibits remarkable self-healing behavior.
Researchers introduced an exclusive self-charging and self-healing flexible asymmetric supercapacitor power cell proficient to sustainably power-up portable electronics by simply applying mechanical bio-mechanical stresses. This self-charging power cell/package comprises an asymmetric supercapacitor unit sandwiched in-between two parallel connected triboelectric nanogenerators and reveals exceptional output performances. Additionally, this nanogenerator cell exhibits remarkable self-healing behavior.
Apr 30th, 2019
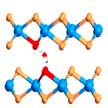 Aqueous rechargeable batteries based on earth abundant materials are promising alternatives to lithium-ion batteries, which are plagued by safety and environmental concerns. These batteries utilize water-based electrolytes, which are safe, low-cost, and environment-friendly, but also possess a much higher ionic conductivity than that of the organic electrolytes. In new work, researchers report an effective and general strategy to transform inactive intercalation hosts into efficient zinc ion storage materials through intercalation energy tuning.
Aqueous rechargeable batteries based on earth abundant materials are promising alternatives to lithium-ion batteries, which are plagued by safety and environmental concerns. These batteries utilize water-based electrolytes, which are safe, low-cost, and environment-friendly, but also possess a much higher ionic conductivity than that of the organic electrolytes. In new work, researchers report an effective and general strategy to transform inactive intercalation hosts into efficient zinc ion storage materials through intercalation energy tuning.
Apr 26th, 2019
 Dozens of nanotechnology research groups worldwide are working on the development of triboelectric nanogenerators (TENGs) for harvesting energy from mechanical vibrations. The huge interest in TENGs stems from their ability to convert ambient mechanical energy into electricity for powering wearable electronics, energy generation using sound, sensor networks, removing air pollution with nanogenerator-enhanced air filters, implantable medical devices, and other small systems. A recent article comprehensively reviews the recent advances in TENG-based health monitoring.
Dozens of nanotechnology research groups worldwide are working on the development of triboelectric nanogenerators (TENGs) for harvesting energy from mechanical vibrations. The huge interest in TENGs stems from their ability to convert ambient mechanical energy into electricity for powering wearable electronics, energy generation using sound, sensor networks, removing air pollution with nanogenerator-enhanced air filters, implantable medical devices, and other small systems. A recent article comprehensively reviews the recent advances in TENG-based health monitoring.
Apr 12th, 2019
 Researchers propose a highly integrated system of an 'air charging' zinc-ion capacitor/battery. By scavenging energy from pervasive air, a zinc-ion capacitor/battery can be conveniently and easily charged without applying an additional power source. The system possesses a simply structure that consists of a flexible bifunctional U-shaped electrode (with the dual function of energy harvesting and storage), a zinc-metal electrode in middle, and two different polyelectrolytes (PAM and PANa) sandwiched between the metal zinc and the electrode.
Researchers propose a highly integrated system of an 'air charging' zinc-ion capacitor/battery. By scavenging energy from pervasive air, a zinc-ion capacitor/battery can be conveniently and easily charged without applying an additional power source. The system possesses a simply structure that consists of a flexible bifunctional U-shaped electrode (with the dual function of energy harvesting and storage), a zinc-metal electrode in middle, and two different polyelectrolytes (PAM and PANa) sandwiched between the metal zinc and the electrode.
Apr 5th, 2019
 Researchers report a simple, innovative and inexpensive design of a vitamin-based hybrid biocompatible nanogenerator with energy harvesting ability from various energy sources in a single device. This is the first time that inexpensive and biocompatible vitamin B2 has been used as a novel and effective beta-phase stabilizer to enhance the piezoelectric performance of PVDF. The attractiveness of using vitamin B2 is that it is biodegradable and biocompatible, thermodynamically stable, low-cost, has delocalized pi-electrons, contains multiple numbers of hydroxyl groups, carbonyl groups and amino groups in its backbone.
Researchers report a simple, innovative and inexpensive design of a vitamin-based hybrid biocompatible nanogenerator with energy harvesting ability from various energy sources in a single device. This is the first time that inexpensive and biocompatible vitamin B2 has been used as a novel and effective beta-phase stabilizer to enhance the piezoelectric performance of PVDF. The attractiveness of using vitamin B2 is that it is biodegradable and biocompatible, thermodynamically stable, low-cost, has delocalized pi-electrons, contains multiple numbers of hydroxyl groups, carbonyl groups and amino groups in its backbone.
Feb 28th, 2019
 Directly carbonizing from organic precursors is the most frequently used method to prepare nanoporous carbons, widely used as electrode materials, due to its flexibility and simplicity. These materials present certain drawbacks, though, such as low surface areas, disordered structures, and non-uniform sizes, which will greatly limit their applications. However, researchers found that carbon materials derived from metal-organic frameworks (MOFs) could overcome these limitations. A review summarizes the applications of MOF-derived carbon materials.
Directly carbonizing from organic precursors is the most frequently used method to prepare nanoporous carbons, widely used as electrode materials, due to its flexibility and simplicity. These materials present certain drawbacks, though, such as low surface areas, disordered structures, and non-uniform sizes, which will greatly limit their applications. However, researchers found that carbon materials derived from metal-organic frameworks (MOFs) could overcome these limitations. A review summarizes the applications of MOF-derived carbon materials.
Feb 20th, 2019
 The formation of dendrites on the metal electrodes of lithium metal batteries causes safety and performance concerns. To avoid dendrites, researchers are experimenting with new battery electrolyte chemistries, new separator technologies, and new physical hosts for the lithium metal. Researchers now have discovered a key design rule for Li metal batteries: If you want to suppress dendrites, you have to use a defect-free host. More generally, carbon defects catalyze dendrite growth in metal anodes.
The formation of dendrites on the metal electrodes of lithium metal batteries causes safety and performance concerns. To avoid dendrites, researchers are experimenting with new battery electrolyte chemistries, new separator technologies, and new physical hosts for the lithium metal. Researchers now have discovered a key design rule for Li metal batteries: If you want to suppress dendrites, you have to use a defect-free host. More generally, carbon defects catalyze dendrite growth in metal anodes.
Feb 13th, 2019
 Researchers have developed a comprehensive approach involving simple and facile steps to fabricate a wearable energy storage device based on carbon nanotube coated cotton yarn. All device components are flexible. According to the team, this is the first device that has been proven to be stable under rigorous washing conditions in the presence of hot water, detergents and high torque (spinning action of washing machine). This provides the device with comprehensive mechanical stability.
Researchers have developed a comprehensive approach involving simple and facile steps to fabricate a wearable energy storage device based on carbon nanotube coated cotton yarn. All device components are flexible. According to the team, this is the first device that has been proven to be stable under rigorous washing conditions in the presence of hot water, detergents and high torque (spinning action of washing machine). This provides the device with comprehensive mechanical stability.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





