Showing Spotlights 57 - 64 of 203 in category All (newest first):
 Spider silk fibers show nature's most outstanding mechanical properties, for example, excellent tensile strength and elasticity, and remarkable protein sequence structure. So far, it has remained completely unidentified how the mechanical properties of spider silk fibers effectively contribute to the performance of a piezoelectric nanogenerator. Now, researchers have successfully demonstrated by Piezoresponse Force Microscopy experiment that natural spider silk fibers have a definite vertical (out-of-plane) piezoelectric coefficient.
Spider silk fibers show nature's most outstanding mechanical properties, for example, excellent tensile strength and elasticity, and remarkable protein sequence structure. So far, it has remained completely unidentified how the mechanical properties of spider silk fibers effectively contribute to the performance of a piezoelectric nanogenerator. Now, researchers have successfully demonstrated by Piezoresponse Force Microscopy experiment that natural spider silk fibers have a definite vertical (out-of-plane) piezoelectric coefficient.
Jun 14th, 2018
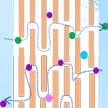 As a promising large-scale energy storage technology, redox flow batteries (RFBs) are attracting increasingly more research attention. For RFB separators, the essential requirement is achieving high ionic conductivity with minimal cross-over at low cost. Researchers now have demonstrated a proof-of-concept graphene oxide (GO) membrane as separator for large-scale energy RFBs. Their work shows that the two-dimensional nanochannel structure and low frictional water flow inside micrometer-thick GO laminates make this material an attractive candidate membrane for large-scale energy storage systems.
As a promising large-scale energy storage technology, redox flow batteries (RFBs) are attracting increasingly more research attention. For RFB separators, the essential requirement is achieving high ionic conductivity with minimal cross-over at low cost. Researchers now have demonstrated a proof-of-concept graphene oxide (GO) membrane as separator for large-scale energy RFBs. Their work shows that the two-dimensional nanochannel structure and low frictional water flow inside micrometer-thick GO laminates make this material an attractive candidate membrane for large-scale energy storage systems.
Apr 17th, 2018
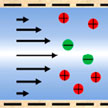 The ability of nanochannels to regulate transported substances in confined spaces is of great research interest in innovative applications, such as high-resolution sensing, filtering, and high-efficiency energy utilization. In the last area, research on nanochannels in energy-related areas continues to face challenges such as low efficiencies, complex preparation processes, and high fabrication costs. Overcoming these challenges is an important and difficult task in the field of energy conversion, energy conservation, and energy recovery.
The ability of nanochannels to regulate transported substances in confined spaces is of great research interest in innovative applications, such as high-resolution sensing, filtering, and high-efficiency energy utilization. In the last area, research on nanochannels in energy-related areas continues to face challenges such as low efficiencies, complex preparation processes, and high fabrication costs. Overcoming these challenges is an important and difficult task in the field of energy conversion, energy conservation, and energy recovery.
Apr 10th, 2018
 Enzymatic biofuel cells (EBFCs) are bioelectronic devices that utilize enzymes as the electrocatalysts to catalyze the oxidation of fuel and/or the reduction of oxygen or peroxide for energy conversion to electricity. EBFCs have already been demonstrated as wearable epidermal tattoo biosensors and in new work, researchers report the fabrication flexible EBFCs with flexible nanoporous gold electrodes that were modified with lactate oxidase and bilirubin oxidase for use as a lactate/O2 biofuel cell.
Enzymatic biofuel cells (EBFCs) are bioelectronic devices that utilize enzymes as the electrocatalysts to catalyze the oxidation of fuel and/or the reduction of oxygen or peroxide for energy conversion to electricity. EBFCs have already been demonstrated as wearable epidermal tattoo biosensors and in new work, researchers report the fabrication flexible EBFCs with flexible nanoporous gold electrodes that were modified with lactate oxidase and bilirubin oxidase for use as a lactate/O2 biofuel cell.
Mar 6th, 2018
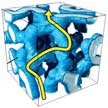 Finding low-cost solid materials capable of efficiently and safely replacing liquid electrolytes in lithium-ion batteries has been a considerable research interest over the past years. Of the various types of solid electrolytes that have been developed so far, composite polymer electrolytes exhibit acceptable Li-ion conductivity due to the interaction between nanofillers and polymer. By fabricating a pre-percolated network of ceramic filler instead of distributing particles in polymer, a 3D interconnected ceramic framework provides continuous pathways for ion conduction. This novel method will help to develop composite materials in a different but much improved way than conventional particle distributions.
Finding low-cost solid materials capable of efficiently and safely replacing liquid electrolytes in lithium-ion batteries has been a considerable research interest over the past years. Of the various types of solid electrolytes that have been developed so far, composite polymer electrolytes exhibit acceptable Li-ion conductivity due to the interaction between nanofillers and polymer. By fabricating a pre-percolated network of ceramic filler instead of distributing particles in polymer, a 3D interconnected ceramic framework provides continuous pathways for ion conduction. This novel method will help to develop composite materials in a different but much improved way than conventional particle distributions.
Feb 28th, 2018
 Scientists report a systematic study involving theoretical and experimental approaches to evaluate the Li-ion storage capability in 2D atomic sheets of nonlayered MoO2. They describe a new process - polymer-assisted reduction - to make atomically flat 2D sheets of MoO2. Since MoO2 is not a 2D material, this process opens a new pathway to make 2D nanostructures from non-layered materials. By successfully making 2D atomic sheets of MoO2, the researchers fabricated a battery electrode in which the Li ion diffusion and electron transport are exceptionally fast.
Scientists report a systematic study involving theoretical and experimental approaches to evaluate the Li-ion storage capability in 2D atomic sheets of nonlayered MoO2. They describe a new process - polymer-assisted reduction - to make atomically flat 2D sheets of MoO2. Since MoO2 is not a 2D material, this process opens a new pathway to make 2D nanostructures from non-layered materials. By successfully making 2D atomic sheets of MoO2, the researchers fabricated a battery electrode in which the Li ion diffusion and electron transport are exceptionally fast.
Feb 15th, 2018
 Inspired by the designs printed on T-shirts, researchers recently reported a new class of wearable power sources. To explore the feasibility of power sources directly printed on cotton T-shirts, which look like letters or symbols, they chose electric double layer supercapacitors based on activated carbon materials as a model electrochemical system. These T-shirts look and behave like a normal T-shirt but feature printed supercapacitors in the shape of letters and symbols.
Inspired by the designs printed on T-shirts, researchers recently reported a new class of wearable power sources. To explore the feasibility of power sources directly printed on cotton T-shirts, which look like letters or symbols, they chose electric double layer supercapacitors based on activated carbon materials as a model electrochemical system. These T-shirts look and behave like a normal T-shirt but feature printed supercapacitors in the shape of letters and symbols.
Jan 19th, 2018
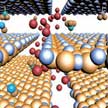 Motived by the large number of non-layered materials, e.g. transition metal oxides, which hold great promise in battery applications, scientists aim to extend the concept of nanofluidic channels into intrinsically non-layered materials and improve their electrochemical properties. In a new study, researchers have developed a Co3O4-based two-dimensional nano-architecture possessing nanofluidic channels with specially designed interlayer characteristics for fast lithium ion transport, leading to exceptional performance in lithium ion batteries ever reported for this material.
Motived by the large number of non-layered materials, e.g. transition metal oxides, which hold great promise in battery applications, scientists aim to extend the concept of nanofluidic channels into intrinsically non-layered materials and improve their electrochemical properties. In a new study, researchers have developed a Co3O4-based two-dimensional nano-architecture possessing nanofluidic channels with specially designed interlayer characteristics for fast lithium ion transport, leading to exceptional performance in lithium ion batteries ever reported for this material.
Dec 8th, 2017
 Spider silk fibers show nature's most outstanding mechanical properties, for example, excellent tensile strength and elasticity, and remarkable protein sequence structure. So far, it has remained completely unidentified how the mechanical properties of spider silk fibers effectively contribute to the performance of a piezoelectric nanogenerator. Now, researchers have successfully demonstrated by Piezoresponse Force Microscopy experiment that natural spider silk fibers have a definite vertical (out-of-plane) piezoelectric coefficient.
Spider silk fibers show nature's most outstanding mechanical properties, for example, excellent tensile strength and elasticity, and remarkable protein sequence structure. So far, it has remained completely unidentified how the mechanical properties of spider silk fibers effectively contribute to the performance of a piezoelectric nanogenerator. Now, researchers have successfully demonstrated by Piezoresponse Force Microscopy experiment that natural spider silk fibers have a definite vertical (out-of-plane) piezoelectric coefficient.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





