| Aug 09, 2022 | |
Nanobiomimetic strategies for medicine |
|
| (Nanowerk Spotlight) The defenses of the body's immune system tend to destroy synthetic nanoparticles and frequently they are captured and removed from the body within few minutes. This, of course, is a major barrier to the use of nanotechnology in medicine. | |
| In order to evade the host immune response, researchers have demonstrated that the combination of nanomaterials and biomimetic strategies can change diverse nanomaterials from 'distinguishable state' to 'camouflage state'. For instance, by 'cloaking' the therapeutic nanoparticles with cell membranes, e.g., by disguising nanoparticles as red blood cells, they often can survive for hours unharmed (read more in our previous Nanowerk Spotlight: "Cell membrane coating as a platform nanotechnology for medicine"). | |
| The development of this kind of biomimetic strategies has become a highly active research field with the goal of developing clinically valuable materials and technologies for nanomedicine applications. | |
| A recent review in Advanced Functional Materials ("Nanobiomimetic Medicine") systematically summarizes and discusses the latest progresses on this profound technology, ranging from their construction and modification, to their widespread and versatile nanomedical applications including bioimaging, drug delivery, cancer diagnosis and therapeutics, and tissue engineering. | |
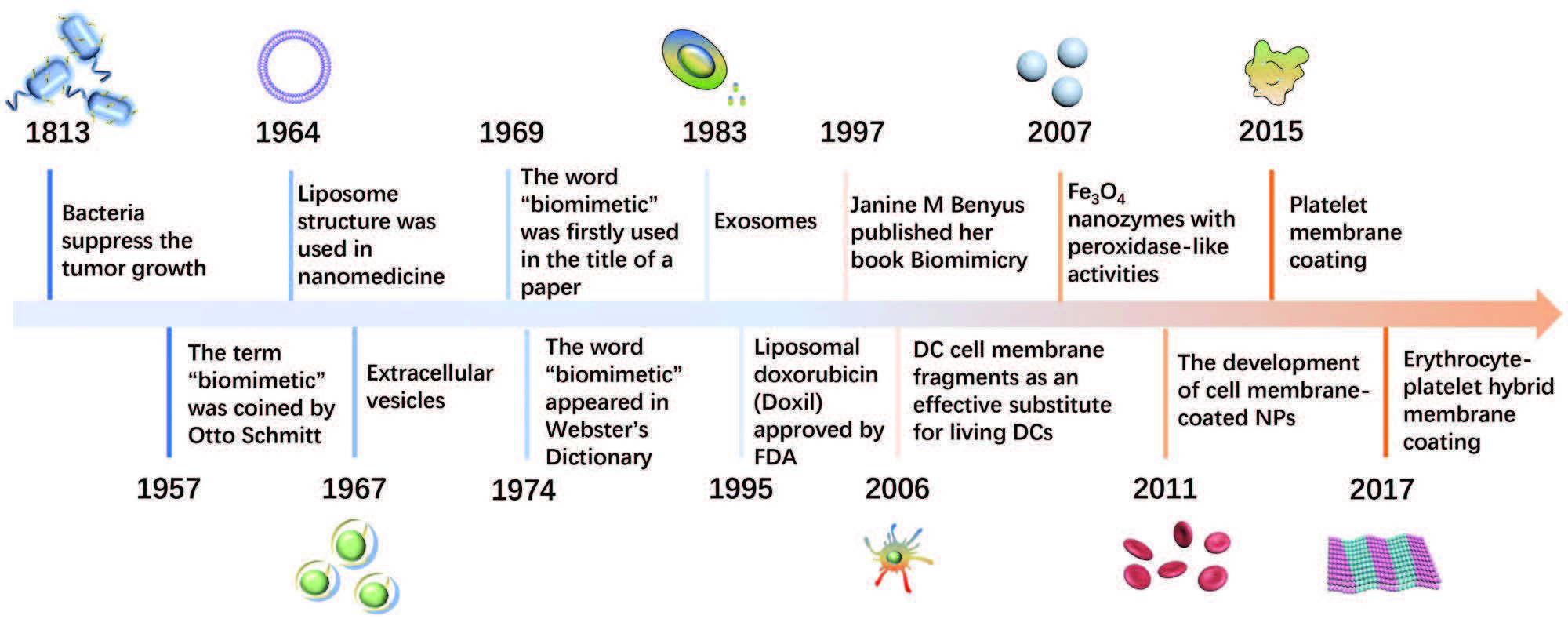 |
|
| A brief timeline for the development of biomimetic technology. (Reprinted with permission by Wiley-VCH Verlag) (click on image to enlarge) | |
| The authors provide an in-depth insight into the relationship between biomimetic preparation strategy (top-down and bottom-up), modification methodology (size, shape, and surface engineering) and biological functions of nanomedicines and biomaterials in order to detail the distinct properties of biomimetic materials for the development of nanobiomimetic medicine. | |
| Biomimetic materials have been extensively explored in the field of diagnostics, therapeutics, and tissue engineering including biomimetic hydrogels, biomimetic liposomes, biomimetic micelles, biomimetic polymer, biomimetic ceramics (hydroxyapatite), high-density lipoproteins (HDLs), and biomimetic nanostructures such as nanoparticles, nanotubes, and nanofibers. | |
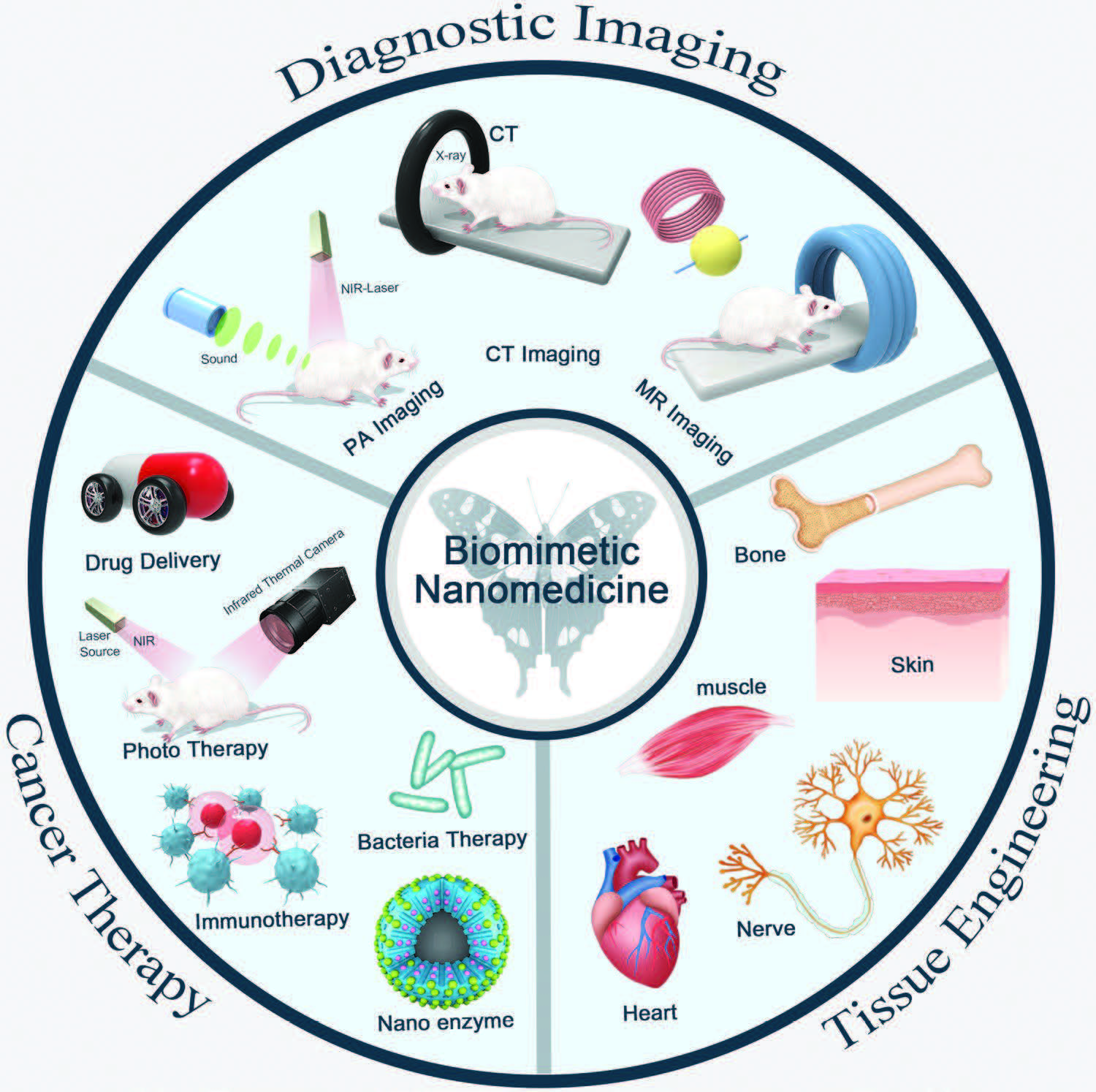 |
|
| Schematic illustration of nanobiomimetic medicine in diagnostic imaging, cancer therapy, and tissue engineering. (Reprinted with permission by Wiley-VCH Verlag) | |
| Scientists consider the precise modulation of the crucial structural parameters – particle size, shape, and surface status – as the most effective strategies for the development of desirable biomimetic nanosystems. The review addresses all three parameters in detail. | |
| Biomimetic strategies have emerged as promising approaches to nanomedicine drug delivery in cancer treatments. Nanodrug delivery systems including nanoparticles, liposomes, and micelles are able to achieve precise drug delivery through controllable release and passively target processes utilizing the enhanced permeability and retention (EPR) effect. | |
| Clinical applications of these systems are hampered, though, by a number of obstacles such as immune clearance, biocompatibility, toxicity and biodistribution profiles. Addressing strategies to overcome these challenges, this review comprehensively summarizes recent advances in the research of nanobiomimetic medicine, highlighting the relationship between biomimetics and nanomedicine, and deepening the understanding of biomimetic strategies. | |
Biomimetic drug delivery |
|
| This is very biomimetic cancer drug delivery shows its advantages. They include biomimetic membrane encapsulation approaches containing erythrocyte membrane-based, platelet membrane-based, cancer cell membrane-based, macrophage membrane-based, and stem cell membrane-based biomimetic drug delivery systems. Again, details for each approach are included in the review. | |
Biomimetic nanozyme therapy |
|
| Another recent approach in cancer medicine are nanozymes – nanoparticles that mimic catalytic sites of natural enzymes and are able to selectively triggering specific catalytic reactions. So far, researchers have successfully developed peroxidase-mimic, catalase-mimic, oxidase-mimic, and superoxide dismutase-mimic nanozymes for biomedical applications. | |
Biomimetic chemotherapy |
|
| Chemotherapy, notwithstanding its severe systemic toxicity and low therapeutic efficiency due to inefficient targeting features, remains one of the most common ways for cancer treatment. Biomimetic therapeutic approaches based on cell membrane coating have emerged as promising alternatives. | |
Biomimetic phototherapy |
|
| As a typical exogenous stimulation approach, the precise and effective tumor treatment could be achieved by biomimetic phototherapy including biomimetic photothermal therapy and biomimetic photodynamic therapy. These techniques employ phototherapeutic agents to realize the generation of heat or reactive oxygen species (ROS) and apply biomimetic strategies to overcome the inherent low targeting limitations. | |
Biomimetic sonodynamic therapy |
|
| As a noninvasive and novel therapeutic modality, biomimetic sonodynamic therapy (SDT) can achieve the purpose of cancer treatment through the triggering of sonosensitizers by ultrasound irradiation to generate ROS. Different from conventional therapies, the advantages of SDT including high spatiotemporal selectivity, minimal invasiveness and deeper tissue penetrating capability. | |
| In order to enhance the effectiveness of SDT, researchers have developed various biomimetic membrane coating systems. | |
Bacteria therapy |
|
| Tumor-targeting bacteria therapy dates back to 1813, when Vautier discovered that gas gangrene could suppress the tumor growth. In the mid-1990s, the number of studies on live bacteria treatment increased because of the generation of effective and less toxic bacterial strains. To date, several bacteria-based therapies have advanced to clinical trials for cancer therapy. Nevertheless, potential toxicities associated with infection remain a major obstacle to clinical development and studies have shown that it is difficult to reduce the tissue toxicity while maintaining the therapeutic effect, whether using domesticated, screened natural strains or simple genetically modified bacteria. | |
| Engineering of bacteria with nanotechnology is an effective way to improve the anticancer ability of attenuated bacteria. For instance, it was reported that DNA delivery and expression of target genes have been accomplished by combining Listeria monocytogenes with DNA-containing nanoparticles. | |
Biomimetic theranostics |
|
| Theranostics is as a promising modality of nanomedicine and can achieve diagnosis and image-guided therapy through diagnostic-therapeutic integrated functions. To date, the development of biomimetic theranostic agents optimized the diagnosis and therapeutic efficiencies, which is attributed to their camouflage and homology performance conferred by biomimetic platforms. | |
Biomimetic tissue engineering |
|
| In addition to cancer therapy, tissue engineering research – the development of artificial matrix substitutes that mimic or improve tissue function – has implemented biomimetic strategies for instance for the engineering of mesenchymal stem cells, bone, blood vessel, and nerves. | |
Other diseases |
|
| Considering the universality and diversity of biomimetic strategies, researchers have also developed strategies to address diseases such as antibacterial therapy, anti-HIV infection, and rheumatoid arthritis. | |
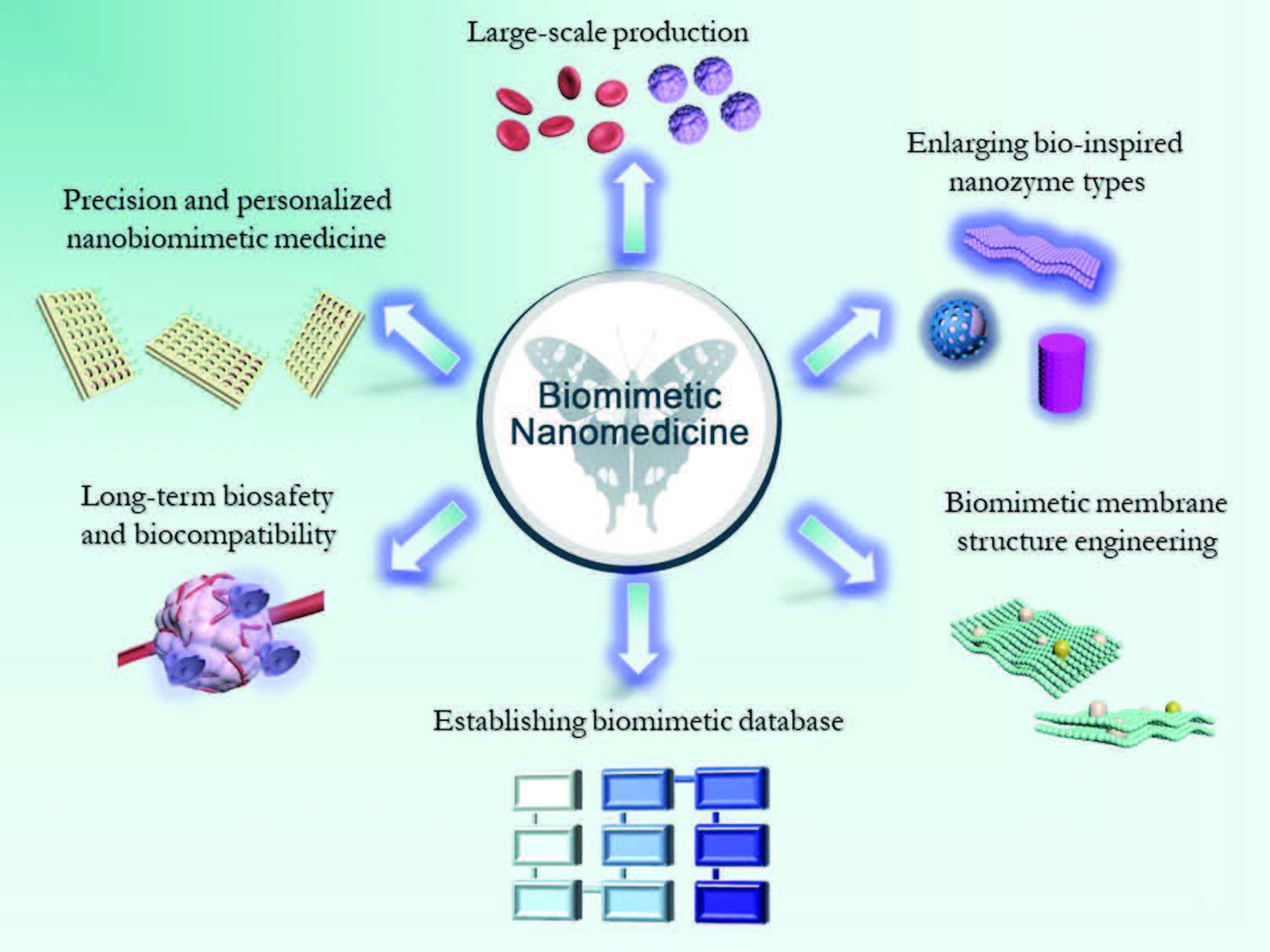 |
|
| Schematic illustration of the development and challenges of nanobiomimetic medicine, including large-scale production, precision and personalized nanobiomimetic medicine, long-term biosafety, and biocompatibility, the creation of biomimetic database, the engineering of biomimetic membrane structure and the expansion bio-inspired nanozyme types. (Reprinted with permission by Wiley-VCH Verlag) | |
| In concluding their review, the authors list a number of challenges and several critical issues, which they suggest should be clarified and addressed for further clinical translation (illustrated above). | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
