| Posted: Mar 12, 2009 | |
Brain implants improved by nanotechnology coatings |
|
| (Nanowerk Spotlight) In previous Spotlights we have addressed the numerous benefits that nanotechnology materials and applications could bring to the field of neural engineering and neural prostheses (see for instance: Nanotechnology to repair the brain). Different biomedical devices implanted in the central nervous system, so-called neural interfaces, already have been developed to control motor disorders or to translate willful brain processes into specific actions by the control of external devices. | |
| Examples of existing brain implants include brain pacemakers, to ease the symptoms of such diseases as epilepsy, Parkinson's Disease, dystonia and recently depression; retinal implants that consist of an array of electrodes implanted on the back of the retina, a digital camera worn on the user's body, and a transmitter/image processor that converts the image to electrical signals sent to the brain. | |
| As promising as these new devices are, the reliability and robustness of neural interfaces is a major challenge due to the way brain tissue responds to the implant. | |
| Mohammad Reza Abidian, a post-doctoral researcher in the Department of Biomedical Engineering at the University of Michigan, says the reliability of today's brain-penetrating microelectrodes often begins to decline after they're in place for only a few months. | |
| Abidian, together with David Martin, professor of macromolecular science and engineering and biomedical engineering, has established methods for the fabrication of multifunctional nanobiomaterials that can be used for coating neural microelectrode arrays that increases their biocompatibility and performance, and is also capable of controlled drug release. | |
 |
|
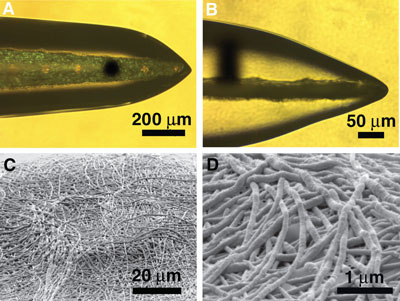
| A) Optical micrograph of deposited PEDOT (black color) on electrode site (top view), B) side view of (A) showing vertical growth of PEDOT from an electrode site and through the alginate hydrogel scaffold (black color), C) SEM images of electrode site after dissolving the alginate coating and electrospun nanofibers. This image reveals that PEDOT was grown around the electrospun nanofibers to form PEDOT NTs. D) Higher magnification image of (C). (Reprinted with permission from Wiley). | |
| This coating is made of three components that together allow electrodes to interface more smoothly with the brain. The nanobiomaterial consists of a special electrically-conductive nanoscale polymer (PEDOT); a natural, gel-like buffer (alginate hydrogel); and biodegradable nanofibers loaded with a controlled-release anti-inflammatory drug. | |
| "The process includes electrospinning of dexamethasone (DEX)-incorporated biodegradable nanofibers, encapsulation of these nanofibers by an alginate hydrogel layer, and then electrochemical polymerization of the conducting polymer PEDOT on the electrode site, around the DEX-loaded nanofibers, and within the hydrogel matrix" Abidian explains to Nanowerk. "We have shown that electrical properties of neural microelectrodes have been significantly improved with these coatings." | |
| The two researchers have reported their findings in the January 29, 2009 online edition of Advanced Functional Materials (Multifunctional Nanobiomaterials for Neural Interfaces). | |
| Abidian describes the three components of their hybrid nanostructured neural interface: | |
| 1) The PEDOT in the coating enables the electrodes to operate with less electrical resistance than current models, which means they can communicate more clearly with individual neurons. | |
| 2) The biodegradable, drug-loaded nanofibers fight the 'encapsulation' that occurs when the immune system tells the body to envelop foreign materials. Encapsulation is another reason these electrodes can stop functioning properly. The nanofibers fight this response well because they work with the alginate hydrogel to release the anti-inflammatory drugs in a controlled, sustained fashion as the nanofibers themselves break down. | |
| 3) The hydrogel, partially derived from algae, gives the electrodes mechanical properties more similar to actual brain tissue than the current technology. That means coated neural electrodes would cause less tissue damage. "These hydrogel coatings provide a mechanical buffer layer between the hard silicon-based probe and the soft brain tissue, a scaffold for growing the conducting polymer within the hydrogel matrix, and a diffusion barrier for controlling drug release," says Abidian. | |
| Especially the last point is of importance since Abidian and Martin demonstrated that alginate hydrogel coatings could decrease the burst effect of the drug (DEX) release for controlling the long-term release patterns. According to Abidian, this controlled release should reduce the risk of exposure to high systemic doses of DEX that are associated with serious side effects such as diabetes, hemorrhagic ulcer, skin atrophy, and osteoporosis. | |
| "We believe that our method provides a generally useful means for creating soft, low impedance, high charge density, controlled releasing for neural prostheses, and other biosensor applications" says Abidian. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
