Showing Spotlights 121 - 128 of 544 in category All (newest first):
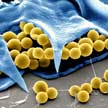 New work on self-propelled biohybrid microrobots has been inspired by recent developments of biohybrid cyborgs that integrate self-propelling bacteria with functionalized synthetic nanostructures to transport materials. Taking inspiration from the science fiction concept of a cybernetic organism, or cyborg, researchers developed a self-propelled biohybrid microrobot, named rotibot, employing the marine microorganism rotifer as their engine.
New work on self-propelled biohybrid microrobots has been inspired by recent developments of biohybrid cyborgs that integrate self-propelling bacteria with functionalized synthetic nanostructures to transport materials. Taking inspiration from the science fiction concept of a cybernetic organism, or cyborg, researchers developed a self-propelled biohybrid microrobot, named rotibot, employing the marine microorganism rotifer as their engine.
Apr 3rd, 2019
 Alpha-synuclein is a protein whose function in the healthy brain is currently unknown. It is of great interest to Parkinson's researchers because it is a major constituent of Lewy bodies, protein clumps that are the pathological hallmark of Parkinson's disease (PD). Scientists believe that the self-assembly of alpha-synuclein into oligomers and fibrils is linked to progress and pathogenesis of the disease. A new study suggest that important characteristics of the fibrillation process, such as surface charge and surface functional group, should be considered in the development of nanotechnology-based therapeutic approaches.
Alpha-synuclein is a protein whose function in the healthy brain is currently unknown. It is of great interest to Parkinson's researchers because it is a major constituent of Lewy bodies, protein clumps that are the pathological hallmark of Parkinson's disease (PD). Scientists believe that the self-assembly of alpha-synuclein into oligomers and fibrils is linked to progress and pathogenesis of the disease. A new study suggest that important characteristics of the fibrillation process, such as surface charge and surface functional group, should be considered in the development of nanotechnology-based therapeutic approaches.
Mar 19th, 2019
 Nanoparticles that mimic the complexity and function of natural enzymes can act as effective peroxidase to catalyse for the oxidization of 3,3,5,5-tetramethylbenzidine (TMB), generating an oxidised blue-coloured product. For the first time scientists found that when the surface of gold nanoparticle (AuNPs) is coated with casein, the intrinsic peroxidase-mimicking activity of AuNPs is suppressed strongly, i.e. by up to 77.1%, due to the surface shielding effects.
Nanoparticles that mimic the complexity and function of natural enzymes can act as effective peroxidase to catalyse for the oxidization of 3,3,5,5-tetramethylbenzidine (TMB), generating an oxidised blue-coloured product. For the first time scientists found that when the surface of gold nanoparticle (AuNPs) is coated with casein, the intrinsic peroxidase-mimicking activity of AuNPs is suppressed strongly, i.e. by up to 77.1%, due to the surface shielding effects.
Dec 6th, 2018
 The idea has been around for a while that selected segments of RNA or DNA could be used therapeutically to affect gene or cell function. The attraction for researchers is the flexibility that these therapeutic nucleic acids' (TNAs) versatility, programmability, and modularity affords them and shows a promising route towards treatment for a wide variety of disorders such as cancer, metabolic disorders, viral infections, cardiovascular and inflammatory diseases. Due to the programmability of RNA and DNA, scientists now are able to embed functional assemblies with controllable immunogenic potential into nucleic acid-based nanoparticles to eliminate the immune response and control the timing of their therapeutic activation.
The idea has been around for a while that selected segments of RNA or DNA could be used therapeutically to affect gene or cell function. The attraction for researchers is the flexibility that these therapeutic nucleic acids' (TNAs) versatility, programmability, and modularity affords them and shows a promising route towards treatment for a wide variety of disorders such as cancer, metabolic disorders, viral infections, cardiovascular and inflammatory diseases. Due to the programmability of RNA and DNA, scientists now are able to embed functional assemblies with controllable immunogenic potential into nucleic acid-based nanoparticles to eliminate the immune response and control the timing of their therapeutic activation.
Dec 5th, 2018
 Researchers investigated the potentially protective role of nanoceria against oxidative stress associated with microgravity and cosmic radiations in space. Their findings support the application of antioxidant nanomaterials to skeletal muscle tissue culture for protection from the noxious effects of microgravity and cosmic radiations, which result in muscle mass and force loss and limit human operations and permanence in space. On Earth, these deteriorations are usually associated with aging or pathologies and are exhibited over longer time intervals compared to those occurring in space.
Researchers investigated the potentially protective role of nanoceria against oxidative stress associated with microgravity and cosmic radiations in space. Their findings support the application of antioxidant nanomaterials to skeletal muscle tissue culture for protection from the noxious effects of microgravity and cosmic radiations, which result in muscle mass and force loss and limit human operations and permanence in space. On Earth, these deteriorations are usually associated with aging or pathologies and are exhibited over longer time intervals compared to those occurring in space.
Oct 29th, 2018
 Investigating the potential of DNA to form self-assembled injectable hydrogels via physical crosslinking with silicate nanodisks, researchers have utilized DNA as a high molecular weight polymeric chain in order to form hydrogel networks for tissue regeneration and drug delivery applications. They have designed shear thinning hydrogels, which can be passed through a 22-gauge syringe by taking advantage of the native chemical structure of DNA and its specific base pairing interactions.
Investigating the potential of DNA to form self-assembled injectable hydrogels via physical crosslinking with silicate nanodisks, researchers have utilized DNA as a high molecular weight polymeric chain in order to form hydrogel networks for tissue regeneration and drug delivery applications. They have designed shear thinning hydrogels, which can be passed through a 22-gauge syringe by taking advantage of the native chemical structure of DNA and its specific base pairing interactions.
Sep 24th, 2018
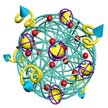 Development of an effective approach for delivering a new drug is as important as inventing a new drug. Even if a developed new drug has excellent pharmacokinetic and therapeutic properties, it shows its potential activity in the body effectively only when it is exactly targeted to specific molecules. Various nanotechnological approaches for effective drug delivery have been developed and some of them have already been successfully commercialized. Most prominent nano drug delivery systems that are in market place are oncology related and based on liposomal, solid nanoparticle based, protein polymer conjugates and polymer-drug conjugate based delivery platforms.
Development of an effective approach for delivering a new drug is as important as inventing a new drug. Even if a developed new drug has excellent pharmacokinetic and therapeutic properties, it shows its potential activity in the body effectively only when it is exactly targeted to specific molecules. Various nanotechnological approaches for effective drug delivery have been developed and some of them have already been successfully commercialized. Most prominent nano drug delivery systems that are in market place are oncology related and based on liposomal, solid nanoparticle based, protein polymer conjugates and polymer-drug conjugate based delivery platforms.
Sep 18th, 2018
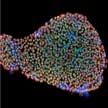 Researchers have developed a way to achieve an ultra-high bioelectric signal from human embryonic stem cells using direct current-voltage measurements facilitated by few-layered 2D molybdenum disulfide sheets. This method, which produces cell signals 2 orders of magnitude higher than previous electrical-based detection methods, paves the way for the development of a broadly applicable, fast, and damage-free stem cell detection method capable of identifying pluripotency with virtually any complementary metal-oxide-semiconductor circuits.
Researchers have developed a way to achieve an ultra-high bioelectric signal from human embryonic stem cells using direct current-voltage measurements facilitated by few-layered 2D molybdenum disulfide sheets. This method, which produces cell signals 2 orders of magnitude higher than previous electrical-based detection methods, paves the way for the development of a broadly applicable, fast, and damage-free stem cell detection method capable of identifying pluripotency with virtually any complementary metal-oxide-semiconductor circuits.
Sep 17th, 2018
 New work on self-propelled biohybrid microrobots has been inspired by recent developments of biohybrid cyborgs that integrate self-propelling bacteria with functionalized synthetic nanostructures to transport materials. Taking inspiration from the science fiction concept of a cybernetic organism, or cyborg, researchers developed a self-propelled biohybrid microrobot, named rotibot, employing the marine microorganism rotifer as their engine.
New work on self-propelled biohybrid microrobots has been inspired by recent developments of biohybrid cyborgs that integrate self-propelling bacteria with functionalized synthetic nanostructures to transport materials. Taking inspiration from the science fiction concept of a cybernetic organism, or cyborg, researchers developed a self-propelled biohybrid microrobot, named rotibot, employing the marine microorganism rotifer as their engine.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





