| Posted: Mar 10, 2011 | |
Biomedical applications of nanodiamonds might require more caution |
|
| (Nanowerk Spotlight) Owing to their large surface area, strong infrared (NIR) photoluminescence and magnetic properties, nanodiamonds are promising for various biomedical applications, including as drug/gene carriers and alternatives to the current bio-imaging platforms (see for instance: "Game-changing nanodiamond discovery for magnetic resonance imaging"). However, the biomedical applications will hardly be realized unless the potential hazards of nanodiamonds to humans and other biological systems are ascertained. | |
| In a previous Nanowerk Spotlight from four years ago ("Diamond nanoparticles appear not to be cytotoxic"), we reported on investigations that indicated that nanodiamonds indeed are very biocompatible, certainly more so than other carbon nanomaterials. These studies showed that nanodiamonds did not produce significant amount of reactive oxygen species (ROS) and did not affect the mitochondrial function and ATP production of these cells. | |
| The biocompatibility of nanodiamonds at the cellular level has been confirmed by many independent studies (e.g. "Peptide-Grafted Nanodiamonds: Preparation, Cytotoxicity and Uptake in Cells" or "The Biocompatibility of Fluorescent Nanodiamonds and Their Mechanism of Cellular Uptake"). | |
| Following these earlier cytotoxicity studies, many groups have used nanodiamonds and their functionalized derivatives for drug/gene deliveries (see for instance: "Nanomedicine promises safer gene therapy with nanodiamonds"). | |
| In spite of the earlier reports that nanodiamonds are biocompatible at the cellular level, researchers have now demonstrated in a new study ("DNA Damage in Embryonic Stem Cells Caused by Nanodiamonds") that nanodiamonds can activate DNA repair proteins in embryonic stem cells, suggesting possible DNA damages. | |
| "Although the observed DNA damage for nanodiamonds is much less severe than other carbon nanomaterials – e.g., multiwalled carbon nanotubes (MWCNTs) – our new findings do suggest some cautions need to be taken in future biomedical applications of nanodiamonds," Liming Da, Kent Hale Smith Professor in the Department of Chemical Engineering at Case Western Reserve University, tells Nanowerk. | |
| Following their first study on the cytotoxicity of nanodiamonds ("Are Diamond Nanoparticles Cytotoxic?"), this is the first genotoxicity study of nanodiamonds. | |
| "We have already demonstrated that DNA damage is possible even for those materials, like multiwalled carbon nanotubes, that have been previously demonstrated to have limited or no toxicity at the cellular level ("DNA Damage Induced by Multiwalled Carbon Nanotubes in Mouse Embryonic Stem Cells")" says Dai. "As far as we are aware, this is the first genotoxicity study on carbon nanomaterials. Our earlier work on genotoxicity of MWCNTs, together with the well-established methodologies developed in our previous study for characterizing DNA damages of embryonic stem cells, prompted us to study the genotoxicity of nanodiamonds in mouse embryonic stem cells." | |
 |
|
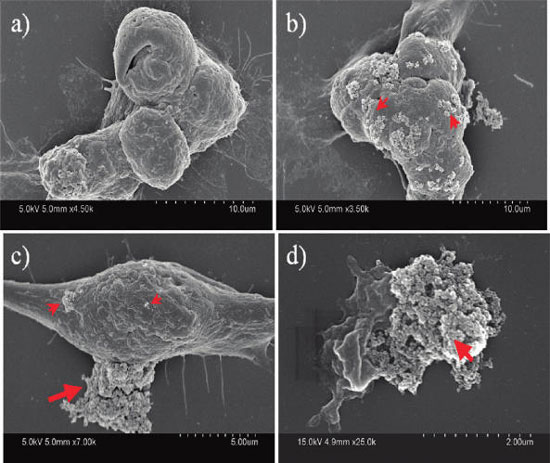
| The nanodiamonds formed clusters of a few hundred nanometers to several micrometers in size (indicated by arrows) and stuck onto the cell membrane. The fact that these clusters stay attached to the cells after many rounds of washing during the sample preparation implies strong interactions between the nanoparticle and the cell membrane. SEM images show that (a) the cell with no nanodiamonds attached, (b) a cell extensively covered with nanodiamonds clusters, (c) a cell with nanodiamonds clusters of various sizes, and (d) a collapsed cell under the huge nanodiamonds cluster. Arrows indicate nanodiamonds cluster; not all clusters are indicated. (Reprinted with permission from American Chemical Society). | |
| Dai explains that embryonic stem cells are a unique cell population with the ability to undergo both self-renewal and differentiation with the potential to give rise to all cell lineages and an entire organism. "It has been shown that embryonic stem cells are highly sensitive to DNA damages, possibly caused by UV irradiation or exposure to nanoparticles. The sensitivity of embryonic stem cells to DNA damage prompted us to study the genotoxicity of nanodiamonds in mouse embryonic stem cells, while the well-established methodologies reported in our previous paper for characterizing DNA damages of embryonic stem cells made the embryonic stem cell as the cell of choice." | |
| The team's results showed that nanodiamonds caused increased expression of DNA repair proteins in mouse embryonic stem cells, indicating the occurrence of DNA damage. | |
| "We found that the pristine as well as oxidized nanodiamonds caused a slight and transient increase in p53 expression within 2-4 hours of cell incubation" says Dai. "However, no significant change in downstream biomarkers has been found in embryonic stem cells treated with the nanodiamonds, suggesting that the damage is minor and might have been repaired very quickly." | |
| Additional experiments on apoptosis and embryonic stem cell differentiation showed that the pristine nanodiamonds were quite benign to embryonic stem cells and did not cause increased differentiation or noticeable apoptosis. However, embryonic stem cells treated with the oxidized nanodiamonds showed signs of differentiation, as well as apoptosis. | |
| "Our genotoxicity results from nanodiamonds and MWCNTs suggest that careful scrutiny of the genotoxicity of nanomaterials is needed even for those materials that have been previously demonstrated to have limited or no toxicity at the cellular level" cautions Dai. "In our present study, oxidized nanodiamonds were demonstrated to cause more DNA damage than the pristine/raw nanodiamonds, showing surface chemistry specific genotoxicity." | |
| He notes that – knowing that the genotoxicity of nanomaterials is surface chemistry specific – there will be considerable room for controlling/minimizing the genotoxicity by functionalization of nanomaterials. | |
| "For this purpose, the most challenging problem that needs to be addressed is to elucidate the genotoxicity mechanism" Dai describes the required next steps in this research. "Our results from both the nanodiamonds and MWCNT studies point to ROS induced DNA damages. However, the detailed mechanisms governing the ROS generation and diffusion into the cell nuclei to cause the DNA damage are still largely unknown." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
