| Posted: Feb 14, 2012 | |
Tissue engineering of 3D tubular structures |
|
| (Nanowerk Spotlight) Tissue engineering is a difficult task where living cells must be organized into tissues with structural and physiological features resembling actual structures in the body (read more: "Tissue engineering meets nanotechnology: A look at tomorrow's medicine"). The ultimate goal of tissue engineering as a medical treatment concept is to replace or restore the anatomic structure and function of damaged, injured, or missing tissue – ultimately providing doctors the ability to replace entire organs. | |
| At the core of tissue engineering is the construction of three-dimensional scaffolds out of biomaterials to provide mechanical support and guide cell growth into new tissues or organs. In another advance for the field, researchers have now demonstrated a strategy to fabricate tubular structures with multiple types of cells as different layers of the tube walls. This method may be widely used in simulation of many tubular tissues and enriches the toolbox for 3D micro/nanofabrication by initially patterning in 2D and transforming it into 3D. | |
| "Tubular tissues such as the trachea, blood vessels, lymph vessels, and intestines, have two distinguishing features" Xingyu Jiang, a professor in chemical biology at the National Center for Nanoscience and Technology in Beijing, explains to Nanowerk: "They have specific 3D shapes;and they have different types of cells at specific locations, i.e. different parts of the tube wall are made up of different cells. Mimicking both of these features is a prerequisite for fabricating functional tubular tissues in vitro, and the realization of structural-tissue mimicry may have wide applications, but remains challenging." | |
| In a paper in a recent issue of Advanced Materials ("A Strategy for Depositing Different Types of Cells in Three Dimensions to Mimic Tubular Structures in Tissues"), Jiang and his team report a new fabrication strategy that results in stable tubular tissue with a high structural similarity to many biological tubular tissues. | |
| Using a stress-induced rolling membrane (SIRM) technique, the researchers used two fabrication steps for their tubular structures. First, different types of cells were delivered and patterned on a 2D SIRM using micro- and nanofluidic channels. Then, the SIRM was released to roll up into a 3D tube. | |
 |
|
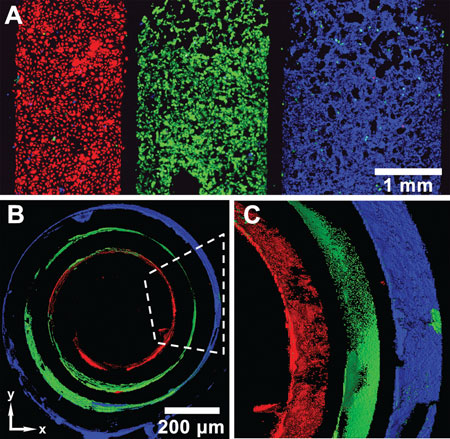
| Three types of cells on a SIRM before and after rolling. Each color indicates a different type of cell. Red are endothelial cells (HUVECs); green: smooth muscle cells; blue: fibroblasts (NIH 3T3). A) Three types of cells are patterned on the surface of a SIRM before rolling, after removing microfluidic channels. B) After rolling of the SIRM, different cells distribute in 3D as the wall of the tubes, in a fashion similar to blood vessels: ECs, SMCs, and fibroblast cells from inside out. Scale bar in (B) only is used on xy plane. C) Magnified and side view of a portion of the tube in (B), indicated by the dashed line. The scanning thickness of the tube is 112 µm in both (B) and (C). (reprinted with permission from Wiley-VCH Verlag) | |
| "We were able to control not only the diameter of the tube and the thickness of the tubular wall, but also the cell orientation inside the tube via the control of cell adhesion on the 2D membrane," says Jiang. "This enables the more precise mimicking of tubular tissues compared with existing approaches." | |
| He points out that, compared with previous methods for the fabrication of vessel-like structures, the advantages of this new method are unique: | |
| 1) Each kind of cell is precisely distributed at microscale resolution in 3D space, and cell orientation can be precisely controlled inside the tubes. | |
| 2) The size of the tubes can be adjusted because the driving force for rolling is internal stress, a parameter that the researchers have complete control over. | |
| 3) The rolling time of the tube is short (within minutes), and the tubular structure can last for several months in tissue-like conditions without any external device. | |
| To demonstrate the capability of their method, the scientists successfully simulated the structure of a human vessel-like structure – the tubular wall has three layers, and in each layer there is one representative type of cells: endothelial cells, smooth muscle cells and fibroblast cells (from inside out). | |
| This kind of tubular structure with multiple types of cells can be applied in tissue engineering such as arterial and venous grafts in vivo. And, as Jiang notes, the preparation method of stress-induced rolling membrane can be applied to fabricate other self-assembled 3D structures. | |
| "I think the future directions for our research field are mapped out in two areas" says Jiang: "One is the study of cell growth, migration and differentiation in 3D structures; the other is the functional simulation of tissue engineering scaffolds. Tissues or organs of animal are extraordinarily complex, so it will be a big challenge to really mimic the function of tissues or organs." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
