| Posted: Aug 31, 2007 | |
An early example of nanotechnology's convergence with other technologies |
|
| (Nanowerk Spotlight) You might have come across the acronym NBIC, which stands for Nanotechnology, Biotechnology, Information technology and new technologies based on Cognitive science. Initially introduced in the U.S. National Science Foundation's Converging Technologies for Improving Human Performance report (pdf download, 5.9 MB) this acronym is often used to describe the basic idea that scientific and technological innovation can be stimulated through the convergence of two, three, or all four fields. At its most radical (and most controversial), proponents of convergence suggest that nanotechnologies will promote the unification of most branches of science and technology, based on the unity of nature at the nanoscale, including cognitive sciences. We'll keep you posted on this over the next few decades and see how it all works out. For the time being, though, it would be nice to be able to report on something more hands-on and - dare I write it - even practical. As it happens, scientists at the University of Toronto have done exactly that. They have demonstrated, for what appears to be the first time, the convergence of nanotechnology, microtechnology, microfluidics, photonics, signal processing, and proteomics to build a medical device that could lead to the development of fast, portable point-of-care diagnostics for infectious disease (IDs) such as HIV, SARS and many others. | |
| Tremendous progress is being made every day in labs around the world on various aspects of the nano-bio-medicine field. In previous studies, researchers have usually avoided a discussion on how to integrate emerging technologies. The discussion and research in many scientific papers also tends to revolve around a single technology. As the fields advance, and in order for all this research to result in practical devices, more integration and convergence will be needed. Some of this work of course is done in industrial labs but you can't read much about it until the finished products hit the market; and so far they haven't. | |
| "Our recent development is that we combined multiple technologies to develop a useful device for monitoring and detection of infectious diseases" Dr. Warren Chan explains to Nanowerk. "We show that with this device we can detect HIV and Hepatitis B and C with greater sensitivity than conventional FDA approved methods." | |
| Chan is an Assistant Professor at the University of Toronto's Institute of Biomaterials and Biomedical Engineering. This work, just published in Nano Letters ("Convergence of Quantum Dot Barcodes with Microfluidics and Signal Processing for Multiplexed High-Throughput Infectious Disease Diagnostics"), represents a major advance because this is one of the first demonstrations of the convergence of many technologies to make a useful medical device. | |
| The Toronto team assembled a high-throughput, high-sensitivity platform capable of detecting multiple biomarkers (multiplexed detection) of ID agents by integrating recent advances in quantum dot QdotB, microfluidic, and photon detection technologies, signal processing, and proteomic biomarkers of infection. | |
| "We showed that this technology can measure some of the most prevalent infectious diseases in serum-spiked sample (a pre-cursor to clinical validation of detection technology) with improved sensitivity" says Chan. | |
| Chan and his collaborators from Toronto General Hospital (led by Dr. Kevin Kain) present a detection system that only requires a few steps (after preparation of so-called quantum dot barcodes – QdotB; not to be confused with the rapper of the same name...), no amplification, and is simpler to use than existing techniques. Furthermore, unlike previous studies, they compare their results to FDA-approved methods and thus assess platform performance against current standards. A major reason for the success of this project lies in the diverse background of this team (lead author Jesse Klostranec ? Aerospace Engineer, Dr. Qing Xiang ? Optical Engineer, Dr. Gabriella Farcas ? Infectious Disease, Dr. Jeongjin Alex Lee ? Electrical Engineer, Dr. Kevin Kain ? Infectious Disease, Steven Perrault and Erin Lafferty ? Biologist, Alex Rhee ? Chemical Engineer). | |
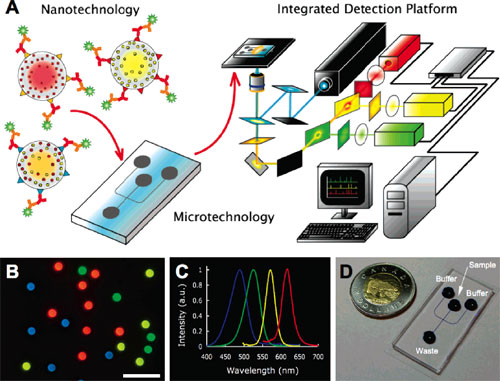 |
Diagnostic scheme. (A) Diagram illustrating the integration of QdotBs, solution-based sandwich assay, microfluidics and fluorescence detection with custom software for high throughput, multiplexed blood-borne pathogen detection (see Supporting Information for details). (B) Fluorescence image of a collection of different color emitting, 5.0 µm diameter polystyrene QdotBs suitable for proteomic or genomic assays (Olympus 40 x objective, 0.75 NA, scale bar = 20 µm). (C) Normalized Qdot emission profiles corresponding to the Qdots used for the barcodes in (B), all excited using 365 nm light. (D) Sample microfluidic chip, fabricated in polydimethylsiloxane with wells labeled. Channel dimensions are 100 µm wide by 15 µm high. Blue dye was used to visualize the channel intersection for electrokinetic focusing. (Reprinted with permission from American Chemical Society) |
| Chan says that developing this platform into a point-of-care device would overcome many of the problems associated with current state-of-the-art detection platforms used in ID diagnostics, such as high operating costs, low sensitivity, and limited multiplexing ability, while improving the speed of diagnosis. | |
| To demonstrate the working of their new device, the scientists chose three diagnostic targets – hepatitis B virus, HBV; hepatitis C virus, HCV, and human immunodeficiency virus, HIV. The detection system itself consists of four major components: | |
| 1) QdotBs conjugated to targeting molecules provide selectivity and multiplexing capabilities. The unique fluorescence signature from the barcode identifies the targeting molecule. | |
| 2) Electrokinetically driven microfluidics enables sequential, high-throughput readout of single barcodes with no movable parts and portability potential. | |
| 3) Photon counting detection systems enable real-time readout of flowing barcodes. | |
| 4) Signal processing enables deconvolution of QdotB optical signals. | |
| "In this proof-of-concept experiment we used the Nie method to prepare three different QdotBs (QdotB1 contained 570 nm emitting ZnS-capped CdSe Qdots; QdotB2 contained 615 nm emitting Qdots; QdotB3 contained both 570 and 615 nm emitting Qdots)" explains Klostranec. "It has been speculated that as many as 1 million unique barcodes can be prepared using this approach because of the narrow spectral line widths and tunable fluorescence emissions of Qdots. If 1 million barcodes are created, 1 million biomarkers could in theory be identified simultaneously." | |
| After the three pathogen biomarkers (i.e., antigens) were covalently bound to the QdotB surface using carbodiimide chemistry, the QdotBs were loaded into the sample well of a microfluidic chip, with the resulting fluorescent emission signals then passing through an optical train of dichroic mirrors, bandpass filters, and focusing lenses before illuminating solid-state photodetectors. | |
| Chan notes that these detectors have response times several orders of magnitude less than the time scale of a flowing bead, allowing for discrimination of single versus aggregated beads in the flow. In a typical experiment, QdotBs are read at a rate of 70 barcodes per minute. | |
| "Fluorescence is a popular detection method because it is typically visible to the naked eye and its signal is more sensitive than most other spectroscopy-based analytical detection methods such as absorbance" says Klostranec. "Although we demonstrated only 50 times greater sensitivity than current FDA-approved diagnostic methods, the sensitivity could be further enhanced in the future. In these experiments, we used organic dyes and polarized laser light in the detection system, where the anisotropy of the dye molecule dipole moment on the surface of the QdotBs results in less efficient excitation of detection fluorophores and hence a lower detection signal." | |
| The researchers hope that by conjugating individual, water solubilized Qdots to the detection antibodies, which possess isotropic dipole moments and larger absorption cross sections than organic dyes, the detection signal should be significantly increased. | |
| Nevertheless, the achieved detection results already were better than for ELISA kits (enzyme-linked immunosorbent assay – a technology to test for the presence of various antigen and antibody in human serum or plasma and widely used in diagnosis of infectious disease, cancer, diabetes, etc.). Not restricted to biomarkers for only IDs, this platform could also be adapted toward screening for biomarkers of other diseases such as cancer or heart disease. | |
| There are a number of major challenges ahead of the Toronto research team but they are determined to perfect the manufacturing and development of each of the device components so that it can be commercialized. | |
| "A major challenge in the medical field lies in the ability to diagnose any kind of infection fast, locally and accurately" says Chan. "This unmet need is what motivates our work. We have witnessed the impact of infectious disease on society and economy in both the developing and developed world. Without the ability to rapidly diagnose a dangerous infection we cannot treat the disease and we cannot minimize it's spread (e.g., SARS and Avian Flu). Our technology hopefully will lead to novel, high-throughput point-of-care diagnostic devices for many serious blood-borne illnesses and help address the diagnostic problems doctors in the field are facing today." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
