| Posted: Dec 29, 2008 | |
Nanotechnology to repair the brain |
|
| (Nanowerk Spotlight) Neural engineering is an emerging discipline that uses engineering techniques to investigate the function and manipulate the behavior of the central or peripheral nervous systems. Neural engineering is highly interdisciplinary and relies on expertise from computational neuroscience, experimental neuroscience, clinical neurology, electrical engineering and signal processing of living neural tissue, and encompasses elements from robotics, computer engineering, neural tissue engineering, materials science, and nanotechnology. | |
| In order for neural prostheses to augment or restore damaged or lost functions of the nervous system they need to be able to perform two main functions: stimulate the nervous system and record its activity. To do that, neural engineers have to gain a full understanding of the fundamental mechanisms and subtleties of cell-to-cell signaling via synaptic transmission, and then develop the technologies to replicate these mechanisms with artificial devices and interface them to the neural system at the cellular level. A group of European researchers has now shown that carbon nanotubes may become the ideal material for repairing damaged brain tissue. | |
| "Our findings show that carbon nanotubes, which, like the nervous cells of our brain, are excellent electrical signal conductors and form intimate mechanical contacts with cellular membranes, thereby establishing a functional link to neuronal structures," Laura Ballerini, a professor of physiology, together with Maurizio Prato, professor of organic chemistry, both at the University of Trieste, Italy, explain to Nanowerk. "Such a functional and mechanical link might favor electrical shortcuts between the proximal and distal compartments of the neuron, therefore improving neuronal performance." | |
| The study was conducted in Prato's and Ballerini's laboratories at the University of Trieste, Italy, in collaboration with Henry Markram's Laboratory of Neural Microcircuitry at Ecole Polytechnique Federale de Lausanne (EPFL) in Switzerland, and Michel Giugliano (now an assistant professor at the University of Antwerp). The team has published their findings in the December 21, 2008 online edition of Nature Nanotechnology ("Carbon nanotubes might improve neuronal performance by favouring electrical shortcuts"). | |
| These findings represent one of the earliest attempts at linking electrical phenomena in nanomaterials to neuronal excitability. | |
| For their experiments, the team deposited single-wall or multi-wall carbon nanotubes on a glass substrate and subsequently defunctionalized them by thermal treatment to form glass slides covered with a purified and mechanically stable thin film of about 50-70 nm thickness. This dense network of nanotubes acts as a largely resistive network that permits long-range electrical connectivity. | |
| The effect of nanotubes on neuronal integrative properties was then investigated by comparing the electrophysiology of rat hippocampal cells cultured on control substrates to those grown on a thin film of purified nanotubes for 8 to 12 days. | |
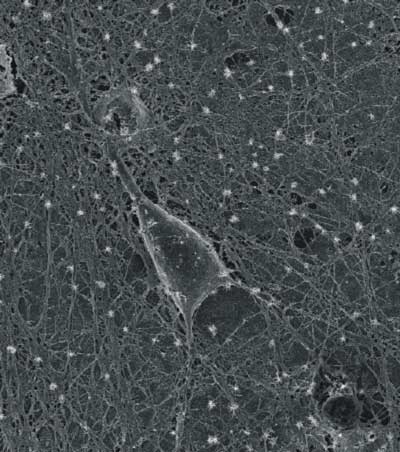 |
|
| TSEM micrograph of a cultured rat hippocampal neuron grown on a layer of purified carbon nanotubes. (Image: Laura Ballerini, University of Trieste) | |
| "In a sample of cultures grown on nanotubes we quantified – in terms of postsynaptic currents frequency – the presence of a significant increase in synaptic activity, compared to control cultures," says Ballerini. "This increased activity represents a typical feature of neurons grown on nanotube substrates." | |
| During the last few years, several research groups supported the use of carbon nanotubes substrates as potential biocompatible materials that promote cell attachment, differentiation, growth and long term neuronal survival. | |
| Ballerini and Giugliano, together with their colleagues at the University of Trieste and the Brain Mind Institute at EPFL, have been working on interfacing neurons with carbon nanotubes for a while now and we have reported on some of their earlier findings in a previous Nanowerk Spotlight ("Nanotechnology coming to a brain near you"). | |
| In this previous work, they reported, for the first time, the effects of carbon nanotubes substrates on the electrical behavior of neuronal networks in vitro. | |
| Ballerini notes that the growth of functional brain circuits on a conductive carbon nanotubes meshwork was always accompanied by powerful physiological changes, in the form of a significant enhancement in the efficacy of neural signal transmission, suggesting that the electrical conductivity of the carbon nanotube-substrate might be underlying these physiological effects. "Yet, the specific functional mechanisms leading to carbon nanotubes-driven neuronal potentiation were largely unexplored," she says. | |
| This new work now considerably widens the perspectives of employing conductive nanomaterials for neuroengineering applications. It proposes carbon nanotubes not only as ideal probes for bidirectional interfaces in neuroprosthetics but also as nanotools to endogenously (re)engineer single-neuron excitability and network connectivity. | |
| "Our results are extremely relevant for the emerging field of neuro-engineering and neuroprosthetics," explains Giugliano, who hypothesizes that the nanotubes could be used as a new building block of novel "electrical bypass" systems for treating traumatic injury of the central nervous system. "Carbon nanotube electrodes could also be used to replace metal parts in clinical applications such as deep brain stimulation for the treatment of Parkinson's disease or severe depression. And they show promise as a whole new class of smart materials for use in a wide range of potential neuroprosthetic applications." | |
| Markram, who heads of the Laboratory of Neural Microcircuitry and an author on the paper, adds: "There are three fundamental obstacles to developing reliable neuroprosthetics: 1) stable interfacing of electromechanical devices with neural tissue, 2) understanding how to stimulate the neural tissue, and 3) understanding what signals to record from the neurons in order for the device to make an automatic and appropriate decision to stimulate. The new carbon nanotube-based interface technology discovered together with state of the art simulations of brain-machine interfaces is the key to developing all types of neuroprosthetics – sight, sound, smell, motion, vetoing epileptic attacks, spinal bypasses, as well as repairing and even enhancing cognitive functions." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
