Showing Spotlights 1545 - 1552 of 2781 in category All (newest first):
 This article summarizes the progress, products outlook, advantages, and limitations of nanocomposites in the automotive industry. Polymer nanocomposites represent a new class of multiphase materials containing dispersion of nano-sized filler materials such as nanoparticles, nanoclays, nanotubes, nanofibers etc. within the polymer matrices. Owing to their nanoscale size features and very high surface-to-volume ratios, they possess unique combination of multifunctional properties not shared by their more conventional composite counterparts reinforced with micro-sized fillers. These multifunctional nanocomposites not only exhibit excellent mechanical properties, but also display outstanding combination of optical, electrical, thermal, magnetic and other physico-chemical properties.
This article summarizes the progress, products outlook, advantages, and limitations of nanocomposites in the automotive industry. Polymer nanocomposites represent a new class of multiphase materials containing dispersion of nano-sized filler materials such as nanoparticles, nanoclays, nanotubes, nanofibers etc. within the polymer matrices. Owing to their nanoscale size features and very high surface-to-volume ratios, they possess unique combination of multifunctional properties not shared by their more conventional composite counterparts reinforced with micro-sized fillers. These multifunctional nanocomposites not only exhibit excellent mechanical properties, but also display outstanding combination of optical, electrical, thermal, magnetic and other physico-chemical properties.
Jan 11th, 2012
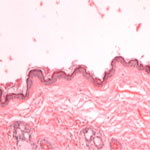 Nitric oxide (NO) is known to possess impressively broad antimicrobial activity due to both its inherent ability to inhibit growth and kill pathogens as well as its function as a potent immunostimulatory signaling molecule. Research data shows that NO is a potentially powerful therapeutic for serious skin and soft-tissue infections, including MRSA (methicillin-resistant S. aureus) infected wounds. However, as a highly reactive gas, NO has proven difficult to deliver in a convenient and cost effective therapeutic format. This limitation has largely precluded its routine use, even in hospital settings. In new work, researchers have now demonstrated the potential application of NO as an antimicrobial agent in the setting of skin and soft tissue infections.
Nitric oxide (NO) is known to possess impressively broad antimicrobial activity due to both its inherent ability to inhibit growth and kill pathogens as well as its function as a potent immunostimulatory signaling molecule. Research data shows that NO is a potentially powerful therapeutic for serious skin and soft-tissue infections, including MRSA (methicillin-resistant S. aureus) infected wounds. However, as a highly reactive gas, NO has proven difficult to deliver in a convenient and cost effective therapeutic format. This limitation has largely precluded its routine use, even in hospital settings. In new work, researchers have now demonstrated the potential application of NO as an antimicrobial agent in the setting of skin and soft tissue infections.
Jan 4th, 2012
 Vanadium dioxide (VO2) has long been recognized as a a material of significant technological interest for optics and electronics and a promising candidate for making 'smart' windows: it can transition from a transparent semiconductive state at low temperatures, allowing infrared radiation through, to an opaque metallic state at high temperatures, while still allowing visible light to get through. So far, VO2 hasn't been considered to be particularly suited for large-scale practical smart-window applications due to its low luminous transmittance and solar modulating ability. Researchers in China have now developed a process that can prepare VO2 thin-films with a controllable polymorph and morphology. Their results show that with increased porosity and decreased optical constants the performance of the VO2 films is enhanced, leading to a higher transmittance of visible light and improved solar modulating ability.
Vanadium dioxide (VO2) has long been recognized as a a material of significant technological interest for optics and electronics and a promising candidate for making 'smart' windows: it can transition from a transparent semiconductive state at low temperatures, allowing infrared radiation through, to an opaque metallic state at high temperatures, while still allowing visible light to get through. So far, VO2 hasn't been considered to be particularly suited for large-scale practical smart-window applications due to its low luminous transmittance and solar modulating ability. Researchers in China have now developed a process that can prepare VO2 thin-films with a controllable polymorph and morphology. Their results show that with increased porosity and decreased optical constants the performance of the VO2 films is enhanced, leading to a higher transmittance of visible light and improved solar modulating ability.
Dec 28th, 2011
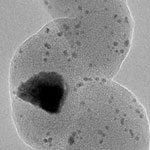 A number of parameters are known to affect the synthesis of carbon nanomaterials, such as the composition and size of the catalysts, type of hydrocarbon gas, temperature, and reaction time. Different carbon nanomaterials having various carbon atomic configurations demonstrate different physical and chemical properties. As a result, it is critical to synthesize carbon nanomaterials with controlled morphology and internal structures for their potential applications as building blocks for nanoscale electronics and photonics, catalyst supports for fuel cells, non-viral carriers for delivering biomolecules into cells, biomedical imaging, and additives for reinforced composite materials. In order to overcome these barriers, researchers need to investigate the interactions between catalysts and carbon nanomaterials to understand how the catalyst facilitate the growth of carbon nanomaterials and, thereby, obtain carbon nanomaterials with controlled properties through tailoring of their catalyst parameters.
A number of parameters are known to affect the synthesis of carbon nanomaterials, such as the composition and size of the catalysts, type of hydrocarbon gas, temperature, and reaction time. Different carbon nanomaterials having various carbon atomic configurations demonstrate different physical and chemical properties. As a result, it is critical to synthesize carbon nanomaterials with controlled morphology and internal structures for their potential applications as building blocks for nanoscale electronics and photonics, catalyst supports for fuel cells, non-viral carriers for delivering biomolecules into cells, biomedical imaging, and additives for reinforced composite materials. In order to overcome these barriers, researchers need to investigate the interactions between catalysts and carbon nanomaterials to understand how the catalyst facilitate the growth of carbon nanomaterials and, thereby, obtain carbon nanomaterials with controlled properties through tailoring of their catalyst parameters.
Dec 23rd, 2011
 Curcumin is the star bioactive component responsible for turmeric's antioxidant, anti-inflammatory and anticancer properties. Recently, it has emerged as one of the most potent chemo-preventive and chemotherapeutic agents. Consequently, there has been a steep rise in the number of research publications and patents starting from the year 2000 onwards. This article presents the findings of a literatre survey and patent analysis on nano-enabled curcumin. There is an upward trend in patenting and publishing activities, which is especially noteworthy from 2007 onwards. One intriguing fact is that the patenting activity is showing a dominating trend in comparison with the scientific research activity suggesting the growing commercial importance of nano-enabled curcumin.
Curcumin is the star bioactive component responsible for turmeric's antioxidant, anti-inflammatory and anticancer properties. Recently, it has emerged as one of the most potent chemo-preventive and chemotherapeutic agents. Consequently, there has been a steep rise in the number of research publications and patents starting from the year 2000 onwards. This article presents the findings of a literatre survey and patent analysis on nano-enabled curcumin. There is an upward trend in patenting and publishing activities, which is especially noteworthy from 2007 onwards. One intriguing fact is that the patenting activity is showing a dominating trend in comparison with the scientific research activity suggesting the growing commercial importance of nano-enabled curcumin.
Dec 22nd, 2011
 Implantable devices like pace makers or neurostimulators are powered by lithium batteries whose service life is as low as 10 years. Hence, many patients must undergo a major surgery to check the battery performance and replace the batteries as necessary. A team in Japan has now reported an interesting strategy that would keep using batteries but provides a mechanism for remotely recharging them from outside the body by converting laser light into thermal energy and subsequently to electricity. The main purpose of this study was to show that it is possible to remotely control electrical energy generation by laser light that can be transmitted through living tissue in order to target various bionic applications implanted in the body.
Implantable devices like pace makers or neurostimulators are powered by lithium batteries whose service life is as low as 10 years. Hence, many patients must undergo a major surgery to check the battery performance and replace the batteries as necessary. A team in Japan has now reported an interesting strategy that would keep using batteries but provides a mechanism for remotely recharging them from outside the body by converting laser light into thermal energy and subsequently to electricity. The main purpose of this study was to show that it is possible to remotely control electrical energy generation by laser light that can be transmitted through living tissue in order to target various bionic applications implanted in the body.
Dec 20th, 2011
 Block copolymer lithography is a cost-effective, parallel, and scalable nanolithography for the densely packed periodic arrays of nanoscale features, whose typical dimension scale is beyond the resolution limit of conventional photolithography. So far, it has been impossible to utilize block copolymer lithography on low surface energy materials such as Teflon, graphene or gold, where block copolymer thin film generally de-wets. To address this technological challenge, researchers in South Korea introduced block copolymer lithography that employs polydopamine coating - inspired by the adhesive proteins secreted by mussels - as a surface pretreatment for universal wettability generally applicable to arbitrary surfaces.
Block copolymer lithography is a cost-effective, parallel, and scalable nanolithography for the densely packed periodic arrays of nanoscale features, whose typical dimension scale is beyond the resolution limit of conventional photolithography. So far, it has been impossible to utilize block copolymer lithography on low surface energy materials such as Teflon, graphene or gold, where block copolymer thin film generally de-wets. To address this technological challenge, researchers in South Korea introduced block copolymer lithography that employs polydopamine coating - inspired by the adhesive proteins secreted by mussels - as a surface pretreatment for universal wettability generally applicable to arbitrary surfaces.
Dec 15th, 2011
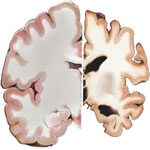 Alzheimer's disease is among the most common brain disorders affecting the elderly population the world over, and is projected to become a major health problem with grave socio-economic implications in the coming decades. The total number of people afflicted by Alzheimer's disease (AD) worldwide today is about 15 million people, a number expected to grow by four times by 2050. This review looks at some of the nanotechnology-enabled approaches that are being developed for early detection and accurate diagnosis of Alzheimer's, its therapeutic treatment, and prevention. These potential solutions offered by nanotechnology exemplify the growing significance that it holds for dealing with brain ailments in general.
Alzheimer's disease is among the most common brain disorders affecting the elderly population the world over, and is projected to become a major health problem with grave socio-economic implications in the coming decades. The total number of people afflicted by Alzheimer's disease (AD) worldwide today is about 15 million people, a number expected to grow by four times by 2050. This review looks at some of the nanotechnology-enabled approaches that are being developed for early detection and accurate diagnosis of Alzheimer's, its therapeutic treatment, and prevention. These potential solutions offered by nanotechnology exemplify the growing significance that it holds for dealing with brain ailments in general.
Dec 13th, 2011
 This article summarizes the progress, products outlook, advantages, and limitations of nanocomposites in the automotive industry. Polymer nanocomposites represent a new class of multiphase materials containing dispersion of nano-sized filler materials such as nanoparticles, nanoclays, nanotubes, nanofibers etc. within the polymer matrices. Owing to their nanoscale size features and very high surface-to-volume ratios, they possess unique combination of multifunctional properties not shared by their more conventional composite counterparts reinforced with micro-sized fillers. These multifunctional nanocomposites not only exhibit excellent mechanical properties, but also display outstanding combination of optical, electrical, thermal, magnetic and other physico-chemical properties.
This article summarizes the progress, products outlook, advantages, and limitations of nanocomposites in the automotive industry. Polymer nanocomposites represent a new class of multiphase materials containing dispersion of nano-sized filler materials such as nanoparticles, nanoclays, nanotubes, nanofibers etc. within the polymer matrices. Owing to their nanoscale size features and very high surface-to-volume ratios, they possess unique combination of multifunctional properties not shared by their more conventional composite counterparts reinforced with micro-sized fillers. These multifunctional nanocomposites not only exhibit excellent mechanical properties, but also display outstanding combination of optical, electrical, thermal, magnetic and other physico-chemical properties. 
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





