Showing Spotlights 185 - 192 of 262 in category All (newest first):
 So far, there have been no research reports on a graphene-based transistor amplifier and investigations of its in-field controllability for analog, mixed-signal, and radio-frequency applications. Previous work on graphene transistors has largely focused on frequency multiplication near the Dirac point in graphene current-voltage characteristic. But now, a team of researchers has demonstrated the first triple-mode graphene amplifier. They have shown experimentally that by leveraging the ambipolarity of charge transport in graphene, the amplifier can be configured in the common-source, common-drain, or frequency multiplication mode of operation by changing the gate bias. This is the first demonstration of a single-transistor amplifier that can be tuned between different modes of operation using a single three-terminal transistor. Moreover, during its operation, the graphene amplifier was configured in-field to switch between the different modes. The result marks another important step toward graphene applications in electronics.
So far, there have been no research reports on a graphene-based transistor amplifier and investigations of its in-field controllability for analog, mixed-signal, and radio-frequency applications. Previous work on graphene transistors has largely focused on frequency multiplication near the Dirac point in graphene current-voltage characteristic. But now, a team of researchers has demonstrated the first triple-mode graphene amplifier. They have shown experimentally that by leveraging the ambipolarity of charge transport in graphene, the amplifier can be configured in the common-source, common-drain, or frequency multiplication mode of operation by changing the gate bias. This is the first demonstration of a single-transistor amplifier that can be tuned between different modes of operation using a single three-terminal transistor. Moreover, during its operation, the graphene amplifier was configured in-field to switch between the different modes. The result marks another important step toward graphene applications in electronics.
Oct 11th, 2010
 Zinc oxide is considered a workhorse of technological development exhibiting excellent electrical, optical, and chemical properties with a broad range of applications as semiconductors, in optical devices, piezoelectric devices, surface acoustic wave devices, sensors, transparent electrodes, solar cells, antibacterial activity etc. Thin films or nanoscale coating of ZnO nanoparticles are viewed with great interest for their many potential applications as substrates for functional coatings. Researchers in Taiwan have now shown, for the first time, that they can directly grow vertically aligned, highly crystalline and defect-free single-crystalline zinc oxide nanorods and nanoneedles on paper.
Zinc oxide is considered a workhorse of technological development exhibiting excellent electrical, optical, and chemical properties with a broad range of applications as semiconductors, in optical devices, piezoelectric devices, surface acoustic wave devices, sensors, transparent electrodes, solar cells, antibacterial activity etc. Thin films or nanoscale coating of ZnO nanoparticles are viewed with great interest for their many potential applications as substrates for functional coatings. Researchers in Taiwan have now shown, for the first time, that they can directly grow vertically aligned, highly crystalline and defect-free single-crystalline zinc oxide nanorods and nanoneedles on paper.
Oct 6th, 2010
 As technology keeps getting faster and smaller, the computer industry is working towards the end of the Moore's Law roadmap where technology will eventually be designed and created at the atomic level. Rather than working their way down incrementally, some researchers are taking a different approach by exploring what happens at the end of Moore's Law, specifically whether it is possible to do computing and other work at that scale. This means they are asking questions like, 'how many atoms are needed to store information', and 'are there schemes to do computation with magnetic atoms instead of transistors'? An IBM research team has now demonstrated, for the first time, the ability to measure how long an individual iron atom can hold magnetic information. They show how a scanning tunneling microscope can measure electron spin relaxation times of individual atoms adsorbed on a surface with nanosecond time resolution using an all-electronic pump-probe measurement scheme.
As technology keeps getting faster and smaller, the computer industry is working towards the end of the Moore's Law roadmap where technology will eventually be designed and created at the atomic level. Rather than working their way down incrementally, some researchers are taking a different approach by exploring what happens at the end of Moore's Law, specifically whether it is possible to do computing and other work at that scale. This means they are asking questions like, 'how many atoms are needed to store information', and 'are there schemes to do computation with magnetic atoms instead of transistors'? An IBM research team has now demonstrated, for the first time, the ability to measure how long an individual iron atom can hold magnetic information. They show how a scanning tunneling microscope can measure electron spin relaxation times of individual atoms adsorbed on a surface with nanosecond time resolution using an all-electronic pump-probe measurement scheme.
Sep 28th, 2010
 Electron pumps are devices that can transfer a certain number of electrons during each pumping cycle. Besides being of fundamental interest to physicists, single-electron pumps have a potential for practical application in metrology, acting as an accurate frequency-current converter. The general goal of this field is to build a current standard based on the electrical charge of a single electron in order to achieve high accuracy for current measurement. A device called single-electron transistor (SET) can confine charges down to single electron level and hence is applicable for quantized current generation. Attempts to generate quantized current in nanotubes have been made with various methods over the past few years, but were not very successful in obtaining a high degree of current quantization. A research team in Germany has now demonstrated the feasibility of using a single molecule - in this case, a single-walled carbon nanotube - for the generation of quantized electric current.
Electron pumps are devices that can transfer a certain number of electrons during each pumping cycle. Besides being of fundamental interest to physicists, single-electron pumps have a potential for practical application in metrology, acting as an accurate frequency-current converter. The general goal of this field is to build a current standard based on the electrical charge of a single electron in order to achieve high accuracy for current measurement. A device called single-electron transistor (SET) can confine charges down to single electron level and hence is applicable for quantized current generation. Attempts to generate quantized current in nanotubes have been made with various methods over the past few years, but were not very successful in obtaining a high degree of current quantization. A research team in Germany has now demonstrated the feasibility of using a single molecule - in this case, a single-walled carbon nanotube - for the generation of quantized electric current.
Sep 9th, 2010
 Single-walled carbon nanotube (SWCNT) based thin film transistors (TFTs) could be at the core of next-generation flexible electronics - displays, electronic circuits, sensors, memory chips, and other applications that are transitioning from rigid substrates, such as silicon and glass, to flexible substrates. What's holding back commercial applications is that industrial-type manufacturing of large scale SWCNT-based nanoelectronic devices isn't practical yet because controlling the morphology of single-walled carbon nanotubes is still causing headaches for materials engineers. In an effort to develop a new and effective solution process of isolated SWCNTs, researchers in Japan have now demonstrated a novel solution process to fabricate high-performance TFTs of individual SWCNTs using DNA.
Single-walled carbon nanotube (SWCNT) based thin film transistors (TFTs) could be at the core of next-generation flexible electronics - displays, electronic circuits, sensors, memory chips, and other applications that are transitioning from rigid substrates, such as silicon and glass, to flexible substrates. What's holding back commercial applications is that industrial-type manufacturing of large scale SWCNT-based nanoelectronic devices isn't practical yet because controlling the morphology of single-walled carbon nanotubes is still causing headaches for materials engineers. In an effort to develop a new and effective solution process of isolated SWCNTs, researchers in Japan have now demonstrated a novel solution process to fabricate high-performance TFTs of individual SWCNTs using DNA.
Sep 7th, 2010
 Biomechanical energy is one of the main energy components in biological systems. Developing an effective technique that can convert biomechanical energy into electricity is important for the future of in vivo implantable biosensors and other nanomedical devices. Researchers have already shown the conversion of biomechanical energy into electricity by a muscle-movement-driven nanogenerator to harvest mechanical energy from body movement under in vitro conditions. In a first demonstration of using nanotechnology to convert tiny physical motion into electricity in an in vivo environment, the same team has now reported the implanting of a nanogenerator in a live rat to harvest energy generated by its breath and heartbeat.
Biomechanical energy is one of the main energy components in biological systems. Developing an effective technique that can convert biomechanical energy into electricity is important for the future of in vivo implantable biosensors and other nanomedical devices. Researchers have already shown the conversion of biomechanical energy into electricity by a muscle-movement-driven nanogenerator to harvest mechanical energy from body movement under in vitro conditions. In a first demonstration of using nanotechnology to convert tiny physical motion into electricity in an in vivo environment, the same team has now reported the implanting of a nanogenerator in a live rat to harvest energy generated by its breath and heartbeat.
Jul 9th, 2010
 Before the superior electronic properties of graphene can be utilized in industrial products, researchers must find a way that allows the mass production of graphene-based devices. New work by a European research team now demonstrates the feasibility of graphene synthesis on commercially available cubic SiC/Si substrates of 300 mm and greater in diameter, which result in graphene flakes electronically decoupled from the substrate. This work demonstrates that it is possible to grow high-quality graphene layers on beta-SiC(001), i.e. on the cubic modification of this material. This is a very important step, since beta-SiC is commercially available and it can be well integrated into present electronic production processes.
Before the superior electronic properties of graphene can be utilized in industrial products, researchers must find a way that allows the mass production of graphene-based devices. New work by a European research team now demonstrates the feasibility of graphene synthesis on commercially available cubic SiC/Si substrates of 300 mm and greater in diameter, which result in graphene flakes electronically decoupled from the substrate. This work demonstrates that it is possible to grow high-quality graphene layers on beta-SiC(001), i.e. on the cubic modification of this material. This is a very important step, since beta-SiC is commercially available and it can be well integrated into present electronic production processes.
Jun 18th, 2010
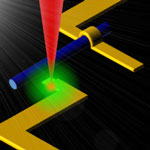 Current fabrication technologies for nanoscale devices include deep-UV or electron-beam (e-beam) lithography. Both of these techniques involve successive deposition of metal or insulating layer and a resist layer, which is patterned using a UV source or a scanning electron beam. The process needs to be repeated for each layer of the architecture while the sample is taken out of the high vacuum chamber. Thus, multilayer lithography processes seriously compromise throughput and cost. In addition, the resolution is limited in the sub-10 nm regime. Researchers at the Institute of Bioengineering and Nanotechnology (IBN) in Singapore have now successfully demonstrated, for the first time, a lithography-free, direct-write technique for fabricating discrete field-effect transistors, as well as digital logic gates on a single nanowire.
Current fabrication technologies for nanoscale devices include deep-UV or electron-beam (e-beam) lithography. Both of these techniques involve successive deposition of metal or insulating layer and a resist layer, which is patterned using a UV source or a scanning electron beam. The process needs to be repeated for each layer of the architecture while the sample is taken out of the high vacuum chamber. Thus, multilayer lithography processes seriously compromise throughput and cost. In addition, the resolution is limited in the sub-10 nm regime. Researchers at the Institute of Bioengineering and Nanotechnology (IBN) in Singapore have now successfully demonstrated, for the first time, a lithography-free, direct-write technique for fabricating discrete field-effect transistors, as well as digital logic gates on a single nanowire.
Jun 16th, 2010
 So far, there have been no research reports on a graphene-based transistor amplifier and investigations of its in-field controllability for analog, mixed-signal, and radio-frequency applications. Previous work on graphene transistors has largely focused on frequency multiplication near the Dirac point in graphene current-voltage characteristic. But now, a team of researchers has demonstrated the first triple-mode graphene amplifier. They have shown experimentally that by leveraging the ambipolarity of charge transport in graphene, the amplifier can be configured in the common-source, common-drain, or frequency multiplication mode of operation by changing the gate bias. This is the first demonstration of a single-transistor amplifier that can be tuned between different modes of operation using a single three-terminal transistor. Moreover, during its operation, the graphene amplifier was configured in-field to switch between the different modes. The result marks another important step toward graphene applications in electronics.
So far, there have been no research reports on a graphene-based transistor amplifier and investigations of its in-field controllability for analog, mixed-signal, and radio-frequency applications. Previous work on graphene transistors has largely focused on frequency multiplication near the Dirac point in graphene current-voltage characteristic. But now, a team of researchers has demonstrated the first triple-mode graphene amplifier. They have shown experimentally that by leveraging the ambipolarity of charge transport in graphene, the amplifier can be configured in the common-source, common-drain, or frequency multiplication mode of operation by changing the gate bias. This is the first demonstration of a single-transistor amplifier that can be tuned between different modes of operation using a single three-terminal transistor. Moreover, during its operation, the graphene amplifier was configured in-field to switch between the different modes. The result marks another important step toward graphene applications in electronics.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





