Showing Spotlights 185 - 192 of 338 in category All (newest first):
 When inkjet printing graphene, achieving satisfactory results it is a trade-off between the sizes of the graphene sheets and the chosen printing strategies. Direct ink writing offers an attractive way to break the routine and meet the printability with the demanding sheet sizes. The nozzles' diameters range from sub micrometer to millimeter scale to accommodate the inks. More importantly, the extrusion-based procedure plays a crucial role in directing the orientation of graphene sheets to pass through the nozzle during printing.
When inkjet printing graphene, achieving satisfactory results it is a trade-off between the sizes of the graphene sheets and the chosen printing strategies. Direct ink writing offers an attractive way to break the routine and meet the printability with the demanding sheet sizes. The nozzles' diameters range from sub micrometer to millimeter scale to accommodate the inks. More importantly, the extrusion-based procedure plays a crucial role in directing the orientation of graphene sheets to pass through the nozzle during printing.
May 12th, 2016
 Lithium-sulfur (Li-S) batteries, which employ sulfur as cathode and metallic lithium as anode materials, have been extensively studied as promising alternatives to the widely used lithium-ion batteries because - theoretically - they can render 3-6 times higher energy density. In practice, though, it has proven challenging to approach that theoretical value. Specifically, the rapid capacity fading, low Coulombic efficiency, and irreversible loss of active materials have impeded large-scale commercial use of Li-S batteries. Researchers now have shown that trapping lithium polysulfide species on (nanoscale) host materials is an effective way to overcome these challenges.
Lithium-sulfur (Li-S) batteries, which employ sulfur as cathode and metallic lithium as anode materials, have been extensively studied as promising alternatives to the widely used lithium-ion batteries because - theoretically - they can render 3-6 times higher energy density. In practice, though, it has proven challenging to approach that theoretical value. Specifically, the rapid capacity fading, low Coulombic efficiency, and irreversible loss of active materials have impeded large-scale commercial use of Li-S batteries. Researchers now have shown that trapping lithium polysulfide species on (nanoscale) host materials is an effective way to overcome these challenges.
Apr 14th, 2016
 The scaling up of nanomaterials in the broader context of materials science and engineering is the topic of a Perspective article, where the authors construct a roadmap for assembling nanoscale building blocks into bulk nanostructured materials, and define some of the critical challenges and goals. Two-dimenisonal sheets are uniquely well-suited in this roadmap for constructing dense, bulk-sized samples with scalable material performance or interesting emergent properties. But no matter what structures are used, when nanostructures with better-than-bulk material performances are used in bulk form, it is critical that those extraordinary nanoscale properties can be scaled to the macroscopic level.
The scaling up of nanomaterials in the broader context of materials science and engineering is the topic of a Perspective article, where the authors construct a roadmap for assembling nanoscale building blocks into bulk nanostructured materials, and define some of the critical challenges and goals. Two-dimenisonal sheets are uniquely well-suited in this roadmap for constructing dense, bulk-sized samples with scalable material performance or interesting emergent properties. But no matter what structures are used, when nanostructures with better-than-bulk material performances are used in bulk form, it is critical that those extraordinary nanoscale properties can be scaled to the macroscopic level.
Feb 4th, 2016
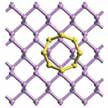
 Hierarchical porous carbon/graphene (HPC/HPG) materials have been intensively investigated over the past decades. These materials are demonstrated as promising electrode materials for various systems, such as lithium-ion batteries, lithium-sulfur batteries, supercapacitors, and fuel cells, with a remarkable capacity, high efficiency, long stability, and excellent rate capability. Researchers have now proposed the employment of hierarchical porous calcium oxide (CaO) particles as effective catalytic template for the facile CVD growth of graphene.
Hierarchical porous carbon/graphene (HPC/HPG) materials have been intensively investigated over the past decades. These materials are demonstrated as promising electrode materials for various systems, such as lithium-ion batteries, lithium-sulfur batteries, supercapacitors, and fuel cells, with a remarkable capacity, high efficiency, long stability, and excellent rate capability. Researchers have now proposed the employment of hierarchical porous calcium oxide (CaO) particles as effective catalytic template for the facile CVD growth of graphene.
Jan 26th, 2016
 Due to the high concentration of silica in rice husks, most of the present research focuses on the preparation of silicon-based materials, which exhibit broad applications in the fields of adsorption, catalysis, energy storage, etc. There is also a large amount of organic components in rice husks, which is typically wasted in the preparation of these silica materials. Researchers now have developed an advanced method for the comprehensive use of rice husks. They fabricated high quality graphene quantum dots from the organic components of rice husks, and simultaneously obtained mesoporous silica nanoparticles with a high surface area from the inorganic content.
Due to the high concentration of silica in rice husks, most of the present research focuses on the preparation of silicon-based materials, which exhibit broad applications in the fields of adsorption, catalysis, energy storage, etc. There is also a large amount of organic components in rice husks, which is typically wasted in the preparation of these silica materials. Researchers now have developed an advanced method for the comprehensive use of rice husks. They fabricated high quality graphene quantum dots from the organic components of rice husks, and simultaneously obtained mesoporous silica nanoparticles with a high surface area from the inorganic content.
Jan 12th, 2016
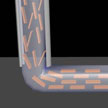
 A scientist has discovered a previously unknown three-dimensional nanostructure consisting of graphene sheets. Graphene is a single monolayer of carbon atoms forming a hexagonal two-dimensional crystal lattice. The discovered nanostructure is a multilayer system of parallel hollow channels with quadrangular cross-section extending along the surface. The discovered nanostructure looked so extraordinary that it took some time to understand what it actually was. The structure was dramatically different from whatever had previously been observed on graphite.
A scientist has discovered a previously unknown three-dimensional nanostructure consisting of graphene sheets. Graphene is a single monolayer of carbon atoms forming a hexagonal two-dimensional crystal lattice. The discovered nanostructure is a multilayer system of parallel hollow channels with quadrangular cross-section extending along the surface. The discovered nanostructure looked so extraordinary that it took some time to understand what it actually was. The structure was dramatically different from whatever had previously been observed on graphite.
Jan 11th, 2016
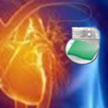 Adding to the options for wirelessly powering implants from outside the body, researchers are proposing a light-driven powering device using near infrared rays (nIR). Flashing light impulses, which are absorbed by the device, induce temperature fluctuation, thus generating voltage/current pulses which can be used for charging a battery or biological stimulations. This flexible and compact device can generate electrical pulses with controllable amplitude and width when remotely irradiated by nIR. Not only can it supply power to implantable bioelectronics, but it also provides adjustable electrical pulses for nerve stimulation.
Adding to the options for wirelessly powering implants from outside the body, researchers are proposing a light-driven powering device using near infrared rays (nIR). Flashing light impulses, which are absorbed by the device, induce temperature fluctuation, thus generating voltage/current pulses which can be used for charging a battery or biological stimulations. This flexible and compact device can generate electrical pulses with controllable amplitude and width when remotely irradiated by nIR. Not only can it supply power to implantable bioelectronics, but it also provides adjustable electrical pulses for nerve stimulation.
Nov 6th, 2015
 The complexity of the microenvironment of a biological cell is influenced by many factors, including surface topography and chemistry; matrix stiffness; mechanical stress; molecular liquid composition and other physiochemical parameters. However, most artificial biointerfaces are developed based on just a single chemical or physical factor to direct cell behaviors. The functions performed by these artificial biointerfaces are far simpler than those performed in the natural cell microenvironment. In an effort to more closely mimic a cell's natural environment, researchers have fabricated an antibody modified reduced graphene oxide platform and used it to significantly improve the efficiency for capturing circulating tumor cells.
The complexity of the microenvironment of a biological cell is influenced by many factors, including surface topography and chemistry; matrix stiffness; mechanical stress; molecular liquid composition and other physiochemical parameters. However, most artificial biointerfaces are developed based on just a single chemical or physical factor to direct cell behaviors. The functions performed by these artificial biointerfaces are far simpler than those performed in the natural cell microenvironment. In an effort to more closely mimic a cell's natural environment, researchers have fabricated an antibody modified reduced graphene oxide platform and used it to significantly improve the efficiency for capturing circulating tumor cells.
Oct 14th, 2015
 When inkjet printing graphene, achieving satisfactory results it is a trade-off between the sizes of the graphene sheets and the chosen printing strategies. Direct ink writing offers an attractive way to break the routine and meet the printability with the demanding sheet sizes. The nozzles' diameters range from sub micrometer to millimeter scale to accommodate the inks. More importantly, the extrusion-based procedure plays a crucial role in directing the orientation of graphene sheets to pass through the nozzle during printing.
When inkjet printing graphene, achieving satisfactory results it is a trade-off between the sizes of the graphene sheets and the chosen printing strategies. Direct ink writing offers an attractive way to break the routine and meet the printability with the demanding sheet sizes. The nozzles' diameters range from sub micrometer to millimeter scale to accommodate the inks. More importantly, the extrusion-based procedure plays a crucial role in directing the orientation of graphene sheets to pass through the nozzle during printing.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





