Showing Spotlights 65 - 72 of 217 in category All (newest first):
 Oxygen reduction reaction (ORR), oxygen evolution reaction (OER), and hydrogen evolution reaction (HER) are among the core electrochemical processes in clean energy conversion and storage devices, such as metal-air batteries, water electrolyzers, and fuel cells. Single-atom catalysts have emerged as a new frontier of heterogeneous catalysts for these reactions due to their highly increased coverage of active sites, enhanced catalytic performance, and maximal metal utilization. By using machine learning algorithms, researchers provide a new paradigm for directly predicting the catalytic performance from physical properties of catalyst candidates.
Oxygen reduction reaction (ORR), oxygen evolution reaction (OER), and hydrogen evolution reaction (HER) are among the core electrochemical processes in clean energy conversion and storage devices, such as metal-air batteries, water electrolyzers, and fuel cells. Single-atom catalysts have emerged as a new frontier of heterogeneous catalysts for these reactions due to their highly increased coverage of active sites, enhanced catalytic performance, and maximal metal utilization. By using machine learning algorithms, researchers provide a new paradigm for directly predicting the catalytic performance from physical properties of catalyst candidates.
Feb 13th, 2020
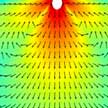 Plasmonic 'nanotweezers' are structures made of noble metals that concentrate and absorb light, resulting in 'plasmonic hotspots' that make it possible to manipulate nanoscale objects suspended in a fluid. Trapping or sensing of nanoparticles using nanotweezers suffers from an intrinsic problem of low throughput as the particle delivery process is often diffusion-limited. Researhers have developed bubble- and convection-assisted trapping techniques that overcome the diffusion-limited trapping in nanoaperture tweezers and enable a reduction of 1-2 orders of magnitude in particle-trapping time compared to that of a diffusion-limited trap.
Plasmonic 'nanotweezers' are structures made of noble metals that concentrate and absorb light, resulting in 'plasmonic hotspots' that make it possible to manipulate nanoscale objects suspended in a fluid. Trapping or sensing of nanoparticles using nanotweezers suffers from an intrinsic problem of low throughput as the particle delivery process is often diffusion-limited. Researhers have developed bubble- and convection-assisted trapping techniques that overcome the diffusion-limited trapping in nanoaperture tweezers and enable a reduction of 1-2 orders of magnitude in particle-trapping time compared to that of a diffusion-limited trap.
Dec 23rd, 2019
 Nanoengineering is a branch of engineering that exploits the unique properties of nanomaterials - their size and quantum effects - and the interaction between these materials, in order to design and manufacture novel structures and devices that possess entirely new functionality and capabilities, which are not obtainable by macroscale engineering. The book 'Nanoengineering: The Skills and Tools Making Technology Invisible' puts a spotlight on some of the scientists who are pushing the boundaries of technology and it gives examples of their work and how they are advancing knowledge one little step at a time.
Nanoengineering is a branch of engineering that exploits the unique properties of nanomaterials - their size and quantum effects - and the interaction between these materials, in order to design and manufacture novel structures and devices that possess entirely new functionality and capabilities, which are not obtainable by macroscale engineering. The book 'Nanoengineering: The Skills and Tools Making Technology Invisible' puts a spotlight on some of the scientists who are pushing the boundaries of technology and it gives examples of their work and how they are advancing knowledge one little step at a time.
Dec 4th, 2019
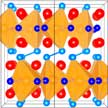 Scientists found that size-confinement - going from macroscopic bulk samples towards nanoparticles - leads to drastic changes in the atomic structure of certain magnetocaloric materials: The change from positive thermal expansion at the macroscale towards a negative thermal expansion (NTE) at the nanoscale. NTE materials are of increasing technological interest mostly because of the idea of mixing them with a symmetric negative thermal expansion material in order to create a composite material which has near-zero thermal expansion. The nanoparticle shape is particularly suitable for the design of such composites.
Scientists found that size-confinement - going from macroscopic bulk samples towards nanoparticles - leads to drastic changes in the atomic structure of certain magnetocaloric materials: The change from positive thermal expansion at the macroscale towards a negative thermal expansion (NTE) at the nanoscale. NTE materials are of increasing technological interest mostly because of the idea of mixing them with a symmetric negative thermal expansion material in order to create a composite material which has near-zero thermal expansion. The nanoparticle shape is particularly suitable for the design of such composites.
Oct 10th, 2019
 2019 marks the 150th anniversary of the Periodic Table, a unique tool, enabling scientist to predict the appearance and properties of matter on the Earth and in the the Universe. Different elements have played critical roles in different periods of human activities, with silicon (Si) being a key element, at present. However, the nanotechnology age has brought different elements into the limelight and transformed their roles in science and technology. A recent article discusses the most relevant elements from a nanotechnology perspective and their applications in nanomaterials.
2019 marks the 150th anniversary of the Periodic Table, a unique tool, enabling scientist to predict the appearance and properties of matter on the Earth and in the the Universe. Different elements have played critical roles in different periods of human activities, with silicon (Si) being a key element, at present. However, the nanotechnology age has brought different elements into the limelight and transformed their roles in science and technology. A recent article discusses the most relevant elements from a nanotechnology perspective and their applications in nanomaterials.
Oct 9th, 2019
 The precise mechanisms underlying friction at the nanoscale are still not completely understood. While gears, bearings, and liquid lubricants can reduce friction in the macroscopic world, the origins of micro-and nanoscale friction for small devices require other solutions. This primer explains the concept of friction, in nature and man-made systems, especially at the nanoscale. It then discusses a new class of 'smart' materials with the ability to modulate the friction of a surface.
The precise mechanisms underlying friction at the nanoscale are still not completely understood. While gears, bearings, and liquid lubricants can reduce friction in the macroscopic world, the origins of micro-and nanoscale friction for small devices require other solutions. This primer explains the concept of friction, in nature and man-made systems, especially at the nanoscale. It then discusses a new class of 'smart' materials with the ability to modulate the friction of a surface.
Aug 29th, 2019
 Nanoparticle characterization is a broad and complex discipline. Even today, researchers are still challenged by the task of determining the physicochemical properties of nanoparticles and exploring their structure-function relationships. A recent review article provides a set of guidelines to investigate and characterize the key parameters defining a nanoparticle sample, namely size, shape, surface charge, and porosity. It also provides recommendations of how the physicochemical parameters of nanoparticles should be investigated, and how to characterize these key properties in different environments according to the intended nanoparticle use.
Nanoparticle characterization is a broad and complex discipline. Even today, researchers are still challenged by the task of determining the physicochemical properties of nanoparticles and exploring their structure-function relationships. A recent review article provides a set of guidelines to investigate and characterize the key parameters defining a nanoparticle sample, namely size, shape, surface charge, and porosity. It also provides recommendations of how the physicochemical parameters of nanoparticles should be investigated, and how to characterize these key properties in different environments according to the intended nanoparticle use.
Jun 5th, 2019
 Superlubricity is a phenomenon where two surfaces slide over each other with barely any resistance (a state that is called near-zero friction). This is a very special case where friction almost vanishes between two surfaces. The difficulty of achieving superlubricity in mechanical systems is due to the very complex physical, chemical, and mechanical interactions that occur simultaneously at the sliding interfaces of these systems. Superlubricity is one of the most promising properties of functional nanomaterials for energy saving applications.
Superlubricity is a phenomenon where two surfaces slide over each other with barely any resistance (a state that is called near-zero friction). This is a very special case where friction almost vanishes between two surfaces. The difficulty of achieving superlubricity in mechanical systems is due to the very complex physical, chemical, and mechanical interactions that occur simultaneously at the sliding interfaces of these systems. Superlubricity is one of the most promising properties of functional nanomaterials for energy saving applications.
May 10th, 2019
 Oxygen reduction reaction (ORR), oxygen evolution reaction (OER), and hydrogen evolution reaction (HER) are among the core electrochemical processes in clean energy conversion and storage devices, such as metal-air batteries, water electrolyzers, and fuel cells. Single-atom catalysts have emerged as a new frontier of heterogeneous catalysts for these reactions due to their highly increased coverage of active sites, enhanced catalytic performance, and maximal metal utilization. By using machine learning algorithms, researchers provide a new paradigm for directly predicting the catalytic performance from physical properties of catalyst candidates.
Oxygen reduction reaction (ORR), oxygen evolution reaction (OER), and hydrogen evolution reaction (HER) are among the core electrochemical processes in clean energy conversion and storage devices, such as metal-air batteries, water electrolyzers, and fuel cells. Single-atom catalysts have emerged as a new frontier of heterogeneous catalysts for these reactions due to their highly increased coverage of active sites, enhanced catalytic performance, and maximal metal utilization. By using machine learning algorithms, researchers provide a new paradigm for directly predicting the catalytic performance from physical properties of catalyst candidates.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





