| Posted: Aug 06, 2009 | |
Improving solid oxide fuel cells with nanostructured electrolyte layers |
|
| (Nanowerk Spotlight) In today's Spotlight we take a look at a specific example of the challenges researchers face in improving fuel cell technology and the important role that modern laboratory instruments such as electron microscopes play in their work. | |
| Fuel cells have gained a lot of attention because they provide a potential solution to our addiction to fossil fuels. Energy production from oil, coal and gas is an extremely polluting, not to mention wasteful, process that consists of heat extraction from fuel by burning it, conversion of that heat to mechanical energy, and transformation of that mechanical energy into electrical energy. In contrast, fuel cells are electrochemical devices that convert a fuel's chemical energy directly to electrical energy with high efficiency and without combustion (although fuel cells operate similar to batteries, an important difference is that batteries store energy, while fuel cells can produce electricity continuously as long as fuel and air are supplied). Read more here: Building better fuel cells with nanotechnology. | |
| The scientific activities of the Institute of Energy Research at the Research Center Jülich (FZJ) – with a staff of about 4400 one of the largest research centers in Europe – are focused on the development of materials, structural elements and components for innovative systems of energy conversion, particularly in the area solid oxide fuel cell (SOFC) and thermal barrier coatings for advanced power stations. In this context, powder synthesis and production of ceramic and metallic materials, as well as mixed materials for bulk and layered elements, is carried out. | |
| The FZJ scientists are also working in fields that are either closely related to these main research areas – like the high-temperature stable composites C/SiC as a possible alternative material for new generations of gas turbines – or those based on the central area of powder technology and coating process, like the manufacturing of powder-metallic elements consisting of NiTi shape memory alloys or the development of metals with improved functional porosity. | |
| The Jülich Research Center has been working on SOFCs since 1989. This includes all research fields like materials development, processing and coating techniques, stack assembling, and system analysis of SOFC power plants. The major task of the institute is the materials synthesis and the development and application of the processing and coating techniques. | |
|
|
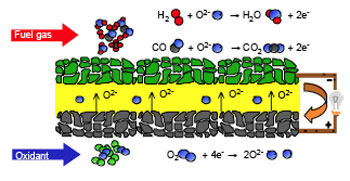
| Principle of a solid oxide fuel cell. A SOFC is based on two electrodes and an electrolyte. The reduction of air is assigned to the cathode electrode, while the fuel gas, e.g. hydrogen, is oxidized on the anode side. The two electrodes are separated by the gas tight electrolyte. In the SOFC the electrolyte is based on an oxygen ion-conducting ceramic material in which oxygen ions are diffusing. On reaching the anode side the ions react with the fuel gas to form water. This migration of ions constitutes a current pushing through an external load. (Images: FZJ) | |
| A recent research focus in the development of SOFCs at the FZJ has been the lowering of the fuel cell's operating temperature from above 800°C down to 600°C in order to achieve cost reduction and better long-term thermal stability. Specifically, this requires a further reduction of the thickness of the cell's electrolyte layer and an optimization of its electrochemical properties with respect to higher ionic conductivity at intermediate temperature are the actual fields of research. | |
| A team at FZJ has developed a planar anode supported SOFC – a concept that is based on the anode substrate as the mechanical supporting component. The sandwich structure of this SOFC stacks | |
|
|
|
| So far, the researchers have achieved an average power output of 1.4 W/cm2 at 750°C and 0.7 V with their fuel cell. | |
| The manufacturing of the electrolyte layer is usually done by vacuum slip-casting or screen-printing. Thinning of this layer needs another deposition technique like the sol-gel process: In a liquid solution of an organometallic precursor a new phase – the sol – is formed by hydrolysis and condensation. This sol is a stable mixture of a solid phase dispersed in a liquid, in which the dispersed phase is much smaller than a micrometer, so that gravitational force is negligible and particle interactions are dominated by short range forces. The dispersed particles in the sol can condense in a gel, in which the solid is still immersed like in a liquid. By drying and firing of the gel during a subsequent low temperature thermal treatment, it is possible to obtain solid matrices with a tailored microstructure. | |

|
|
| SEM images of a layer from a colloidal sol deposited onto a tape-casted substrate sintered at 1400°C.(Images: FZJ) | |
| A fundamental property of the sol-gel process is the generation of a ceramic material at rather low temperature compared to traditional routes. By tuning hydrolysis and condensation reactions, the desired nano-sized ceramic particles are obtained for making advanced layers. | |
| Such thin layers can be deposited by spraying, spin coating, dip coating or even painting. The as-deposited gel layers contain still a large solvent amount. Drying (evaporation of the solvent) creates the final amorphous or crystalline layer, which is further processed by calcination (decomposition of starting materials and formation of reaction products) and sintering (densification). | |
| There are several problems that need to be investigated and solved when taking the sol-gel layer fabrication route: homogeneity of multi-layers by repeated coating; soaking of the sol into the substrate grain assembly due to capillary tension; and crack formation and their growth. | |
| The lab in Jülich uses a high end scanning electron microscopy (SEM) and several optical microscopes for studying the layer formation. The information retrieved from optical microscopy was not sufficient with respect to detail resolution. A high end SEM can overcome this problem, but the workload on it results in a long sample throughput. The way-out offers a microscope which imaging capabilities exceed those of an optical one and which allows quick access to shorten cycle time between coating manufacturing and microstructure evaluation. The team is using the table top scanning electron microscope Phenom from FEI for studying 1) improvement of the substrate surface quality, 2) optimization of coating process, and 3) evaluation of layer quality. | |
| A key argument for using the table top SEM has been a shortening of the cycle time while still being able to perform structural sample characterization at reasonable costs. Yet – with a magnification of 5000-10000x, a low accelerating voltage of 5 kV together with the high-sensitive backscattered electron detector – the instrument is powerful enough to generate detailed surface-enhanced images. | |
|
|
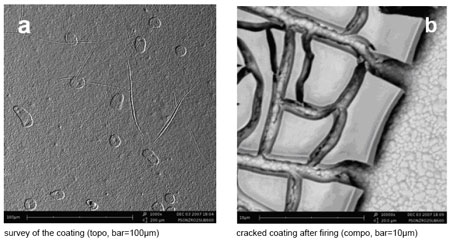
| Polymeric sol dip-coated and fired at 600°C on tape-casted 3YSZ substrate with a well finished surface. (Images: FZJ) | |
| The SOFC researchers operated their table top Phenom in its two imaging modes – compositional (compo) and topographical (topo) contrast mode. In compo contrast mode, differences in the average atomic number of different phases and early stages in grain formation can be seen (see images above). Topo contrast mode shows that topographic features of the substrate prevent an even and homogenous layer. This leads to the conclusion that high quality thin electrolyte layers can be made only on well finished substrate surfaces and with a particle size of the solid constituent in the gel slightly larger than the size of remaining defects. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
