| Posted: Jan 11, 2007 | |
Nanotechnology could offer an alternative to brain surgery |
|
| (Nanowerk News) If you had brain tumor, would you rather receive your medicine through an injection in the arm or have a hole drilled in your skull? Even if you opted for the 'hole-in-the-skull' method, brain cancers are often inoperable due to their location within critical brain regions or because they are too small to detect. | |
| Nanotechnology offers a vision for a 'smart' drug approach to fighting tumors: the ability of nanoparticles to locate cancer cells and destroy them with single-cell precision. One of the most important applications for such nanoparticulate drug delivery could be the delivery of the drug payload into the brain. However, crossing the brains protective shield, the blood-brain barrier, is a considerable challenge. | |
| Novel targeted nanoparticulate drug delivery systems that are able to cross this barrier bring us closer to this vision of brain cancer destroying drugs. | |
| A tight seal of endothelial cells lines the blood vessels in the brain and acts as a barrier to protect its cells. This is known as the blood-brain barrier (BBB). BBB strictly limits transport into the brain through both physical (tight junctions) and metabolic (enzymes) barriers and keeps most substances, such as chemicals and large biomolecules, out of the brain. | |
| The challenge in treating most brain disorders is overcoming the difficulty of delivering therapeutic agents to specific regions of the brain by crossing the BBB. The problem is the BBB does not differentiate what it keeps out. With very few exceptions, only nonionic and low molecular weight molecules soluble in fat clear the BBB. For instance, alcohol, caffeine, nicotine and antidepressants meet these criteria. However, large molecules needed to deliver drugs cannot cross it. | |
| "The delivery of peptides into the brain is a challenge that is not adressed with classical pharmaceutical formulations, since the blood brain barrier doesn't allow the easy diffusion of such molecules." Dr. Patrick Couvreur explained to Nanowerk. | |
| Couvreur, the director of the Center for Pharamceutical Studies at the Université Paris Sud, together with an international group of researchers, describes novel nanoparticles that are able to transport drugs into the brain ("Development and Brain Delivery of Chitosan-PEG Nanoparticles Functionalized with the Monoclonal Antibody OX26"). | |
Nanoparticle delivery to the brain |
|
| The development of carrier nanoparticles able to penetrate the BBB, as done by this research group, is an important step towards detection and therapeutic treatment of diseases in the brain. BBB investigation is an ever growing and dynamic field studied by pharmacologists, neuroscientists, pathologists, physiologists, and clinical practitioners. | |
| "The chitosan-based immuno-nanoparticles we have developed" says Couvreur "allow transport across the BBB by combining two factors: 1) the ability of cationic (with a full positive charge) chitosan to interact with the negative charges of the brain endothelium and 2) the affinity of the monoclonal antibody OX26 for the transferrin receptor." | |
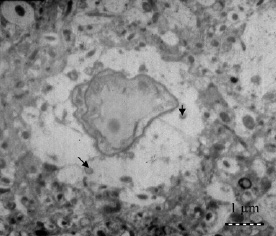 | |
| Transmission electron micrographs showing nanoparticles in the brain interstitium (original magnification 5000); (Reprinted with permission from American Chemical Society) | |
| The researchers hypothized that transferrin receptor (TfR), which is highly concentrated in the BBB, may trigger receptor-mediated transport across the BBB. Transferrin itself is limited as a brain drug transport vector since the transferrin receptors are almost saturated under physiologic conditions. However, the antibodies that bind to TfR have been shown to selectively target BBB endothelium due to the high levels of TfR expressed by these cells. Therefore, these antibodies are potential carriers for the delivery of therapeutic agents to the central nervous systems. In particular, a mouse monoclonal antibody, OX26, with a high abundance of transferrin receptors at the brain microvascular endothelium has been detected and was used in the experiments. | |
| Consequently, the researchers designed chitosan (CS) nanospheres conjugated with poly(ethylene glycol) (PEG) bearing the OX26 monoclonal antibody. The nanoparticles were injected intravenously to mice. The results showed that an important amount of nanoparticles were located in the brain. | |
| Raoul Kopelman and his group at the University of Michigan designed nanoparticles they dubbed Probes Encapsulated by Biologically Localized Embedding (PEBBLEs) to carry a variety of agents on their surface, each with a unique function. The particles consist of an iron oxide core that serves as an MRI contrast agent. Attached to them are copies of a cancer-targeting peptide called F3, as well as a light-absorbing compound called photofrin that kills cells when hit with red light. When Kopelman?s team used their PEBBLEs to treat rats previously injected with cancer cells inside their brains they saw very clear results: A single IV injection of a targeted therapeutic nanoparticle dose, coupled with a single 5 min photodynamic therapy treatment, leads not only to an increase in the rat survival rate but accomplishes complete tumor remission. | |
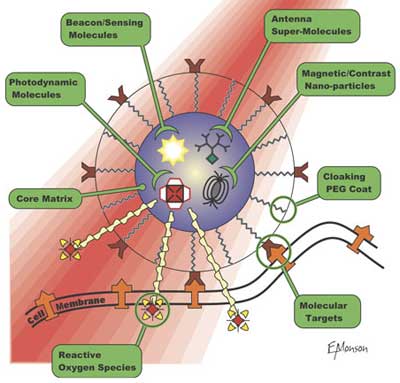 | |
| The PEBBLE platform for the detection and treatment of cancer (Image: Kopelman Laboratory) | |
| The targeted treatments using nanoparticles may offer a number of advantages, not only over surgery but also over traditional chemotherapy. In chemotherapy, the drugs indiscrimenately permeate cells throughout the body to damage their DNA and prevent rapid growth, and are only moderately more toxic to cancer cells over normal cells. Nanoparticulate drug delivery, on the other hand, is highly localized to the cancer target, and does no or very little damage to surrounding healthy tissue. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
