| Posted: Aug 17, 2016 | |
2D electrode materials enable high performance sodium ion full cells |
|
| (Nanowerk Spotlight) Sodium-ion batteries (SIBs) represent an attractive alternative to lithium-ion batteries, owing to the fact that sodium resources are practically inexhaustible and evenly distributed around the world while the ion insertion chemistry is largely identical to that of lithium. Moreover, although lithium ion batteries have been rapidly developed and are widely used, the easily accessible global lithium reserves largely exist in politically sensitive or remote areas. | |
| "However, the larger sodium ion and its higher ionization potential require a more open framework for Na-ion intercalation and deintercalation," Guihua Yu, an Assistant Professor of Materials Science at the University of Texas at Austin, explains to Nanowerk. "Searching for suitable electrode materials is still a challenge for the development of the SIBs. The most exciting result of our recent research in this area is that an advanced sodium ion full battery was rationally designed and fabricated and both of the cathode and anode materials possessed very unique two-dimensional (2D) nanostructured architecture." | |
| The findings by Yu's group have been published in Energy & Environmental Science ("An advanced high-energy sodium ion full battery based on nanostructured Na2Ti3O7/VOPO4 layered materials"). | |
| In comparison with the electrodes made from the bulk, the 2D nanostructured electrodes have several advantages: They provide high surface area and large open space for fast charge and mass transport; and they show the ability to better withstand structural damage/change caused by sodium insertion and extraction, leading to better reversible capacities of the electrodes. | |
| Yu points out that these unique features enabled by 2D nanostructured architecture, result in excellent rate capability (fast charge/discharge) and stable cycling performance of the assembled sodium ion full batteries. | |
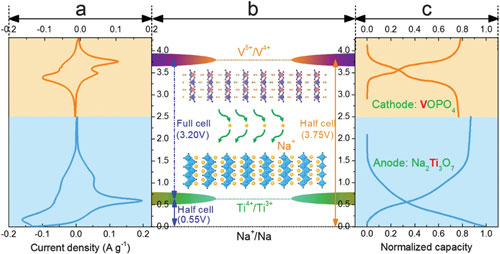 |
|
| The working principle of the designed sodium ion full battery based on nanostructured Na2Ti3O7/VOPO4 layered materials. (© The Royal Society of Chemistry) (click on image to enlarge) | |
| Most of the current research concentrates on sodium ion half-cells, and the development of sodium ion full cells with high performance has remained a critical challenge. Previous reports on sodium ion full batteries are typically based on a sodium storage cathode and a nonmetallic sodium anode, and their battery performance has been rather disappointing. | |
| The few examples of sodium ion full cells that have been developed are mainly fabricated using hard carbon as the anode material. But this material suffers from inferior rate capability and the voltage plateau related to most capacities is too close to the sodium plating voltage, causing a serious safety concerns. | |
| In order to achieve higher energy density and better safety, better sodium ion full batteries with both novel cathodes and anodes need to be designed and studied. | |
| This is where the research by Yu's group comes in. The novel sodium ion full batteries that they designed utilize nanostructured Na2Ti3O7 and VOPO4 as the anode and cathode, respectively. | |
| "Both of the electrode materials possess a layered crystal structure, enabling excellent rate capability and cycling performance," says Yu. "Moreover, the temperature-dependent battery tests that we carried out for our sodium ion full battery demonstrated excellent capacity retentions in a wide temperature range of -20 to 55°C. Therefore, our results represent an important step forward in understanding the battery characteristics of sodium ion full batteries, and showing great potential of nanostructured layered electrode materials for building high-performance sodium ion batteries." | |
| Thanks to the advantageous electrochemical features of both anodes and cathodes, these sodium ion full batteries exhibit excellent electrochemical characteristics. The full battery outputs one of the highest operating voltages ~3V and delivers a large reversible capacity of 114 mA h g-1 at a rate of 0.1C with outstanding rate capability (fast charge/discharge possible) and excellent cycling stability (~92.4% capacity retention after 100 cycles). A high energy density of 220 W h kg-1 is achieved, which is comparable to the state-of-the-art lithium ion batteries. | |
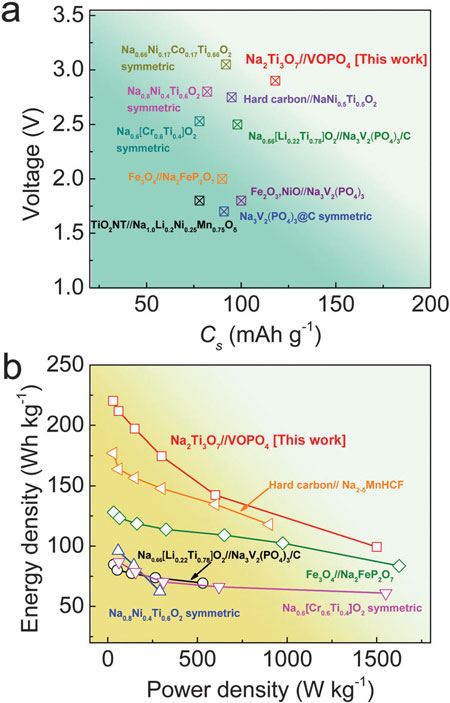 |
|
| Sodium ion storage properties of the Na2Ti3O7/VOPO4 full cells and other recently reported literatures. (© The Royal Society of Chemistry) | |
| Because of sodium's high abundance and low cost, sodium ion full batteries have great potential in large-scale energy storage, such as stationary grid storage. They may also serve as power supply for electronic devices. | |
| The researchers point out that, as demonstrated in their paper, the designed SIBs show good mechanical flexibility – performing well under different mechanical states, e.g. bending, folding, rolling – and therefor may even serve as energy devices to power flexible/wearable electronics. | |
| Going forward, the team is continuing their search for suitable electrode materials with novel nanostructured designs that can accommodate large volumetric strains to enable much longer cycle life SIBs. | |
| In addition, replacing the high volatile and flammable organic electrolytes with solid-state electrolytes or quasi-solid-state electrolytes provides another exciting opportunity to build a safer sodium ion battery. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
