| Posted: Oct 30, 2008 | |
Real-time observation of crystal growth |
|
| (Nanowerk Spotlight) Crystalline nanoporous compounds have attracted the attention of scientists and materials engineers because of the interest in creating nanoscale spaces and the novel phenomena in them. Nanoporous materials find applications in many chemical processes such as separation and catalysis and are also heavily researched as storage materials, for instance in hydrogen fuel cells (see: Nanotechnology storage tanks). Researchers still lack a complete understanding of the mechanism that leads to, and occurs during, crystal growth. Only once scientists achieve full control of properties such as composition, structure, size, morphology, and the presence and form of defects within the crystals can they fully exploit the benefits of crystalline nanoporous materials for the fabrication of novel materials. | |
| Researchers in the UK have now presented definitive real-time evidence of the crystal growth mechanism in what appears to be the first high-resolution observation of in-situ crystal growth of a crystalline nanoporous material monitored using atomic force microscopy (AFM). | |
| "Real time monitoring of crystal growth provides a unique opportunity to observe intermediate structures of the crystal formed during the crystal growth process" Dr. Martin P. Attfield tells Nanowerk. "Use of in-situ AFM measurements demonstrates that geometric information (i.e. heights) obtained from these images provide additional information on the nature of growth units. This may complement spectroscopic studies in order to gain an overall picture for the crystal growth process of framework materials, a central question in the synthesis of nanoporous solids and covalently or datively bonded solids in general." | |
| Attfield, a Lecturer at the Centre for Nanoporous Materials in the School of Chemistry at the University of Manchester in the UK, together with his colleagues Maryiam Shoaee and the Centre's director Prof. Michael W. Anderson, has published a paper on his findings in the October 8, 2008 online edition of Angewandte Chemie International Edition ("Crystal Growth of the Nanoporous Metal-Organic Framework HKUST-1 Revealed by In Situ Atomic Force Microscopy"). | |
| Attfield says that the crystal growth of frameworks around void space full of solvent or template molecules is still relatively poorly understood but that in-situ measurements such as the ones performed by his team may allow future measurement of fundamental growth parameters. | |
| "These parameters include rate constants and activation energies, in addition to information on the units of growth, all of which will increase our understanding of various aspects of crystal growth of framework materials" he says. "Increase in understanding within this area should aid the design of synthesis techniques and conditions to grow crystallites of these materials in a more controlled manner. Future goals in this area would include being able to grow crystals with a specific size and habit, massive single crystals, single crystal films, defect-free materials or materials with specific defect concentrations." | |
| In his recent work, Attfield and his colleagues studied the crystal growth of a crystalline nanoporous material called HKUST-1, an important member of the nanoporous metal organic framework family that is receiving a lot of attention for, among other things, applications in gas storage and purification. | |
| By taking a series of high-resolution AFM deflection micrographs of the growing face of HKUST-1 over time, the scientists observed how the crystal surface changed dramatically after injection of a growth solution. | |
| "Our observations indicate that growth of the crystal surface is through a two-dimensional surface nucleation or 'birth and spread' crystal growth mechanism" says Attfield. "At the lowest supersaturation levels tested, we found step velocities of ca. 0.4 nanometers per second, corresponding to a growth rate perpendicular to the crystal surface of ca. 0.04 micrometers per hour. Such step velocities are comparable to those reported in other metal-organic extended solids." | |
| One aspect of the growth of this crystal surface revealed exclusively by real-time imaging is the influence of line defects present on the initial substrate surface. | |
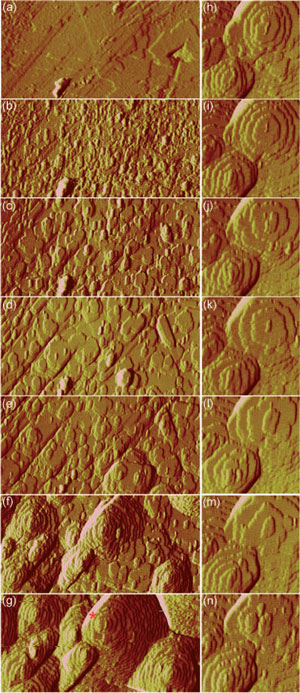
|
|
| Deflection AFM images of the growing {111} facet of a HKUST-1 crystal after a) 0, b) 4, c) 9, d) 17, e) 31, f) 74, g) 111 h) 123, i) 128, j) 132, k) 137, l) 141, m) 145, n) 154 min. The red asterisk in image (g) marks the main growth hillock at the top of images (h)?(n). The image sizes in (a)?(g) and (h)?(n) are 3.0x1.5 µm2 and 1.0x1.0 µm2, respectively. (Reprinted with permission from Wiley) | |
| "The line defects, visible as diagonal lines in the bottom left-hand corner of (a) [in the image above], initially act as boundaries impeding the step flow of adjacent growing nuclei and islands" says Attfield. "This results in the formation of approximately rectangular step bunches, as seen in (d) and (e). Eventually sufficient growth on either side of a line defect enables growth hillocks to form that straddle the area where the line defect was present and the latter is no longer visible as observed in (g). Crystal growth over other large gross defects visible on the substrate surface is also evident in (a)-(g). Growth of the crystal surface over these features will introduce defects into the bulk of the resultant crystal that may have beneficial or detrimental effects on its final properties. It is also interesting to note that no fracturing of the newly formed crystal surface is noticeable which supports the observation that the fractures observed in the ex situ AFM images of {111} faces of HKUST-1 occur after crystallization." | |
| As the University of Manchester team has shown, in-situ AFM monitoring of crystal growth provides a unique opportunity to observe intermediate structures of the crystal structure formed during the crystal growth process and analysis of the geometric information contained within these images indicate that the actual units of growth are more likely to be simpler fragments rather than preformed building blocks such as that shown in the image to the lower left. | |
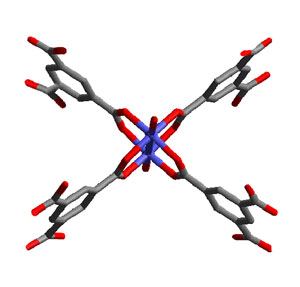
|
|
| The structure of a postulated fundamental building block of HKUST-1. Key: blue - Cu; red - oxygen; grey ? carbon. (Image: Dr. Attfield/University of Manchester) | |
| Attfield notes that findings gained from such observations will affect how the growth of extended and framework materials is considered, particularly with respect to the fundamental building blocks of growth. "It clearly demonstrates the power and potential of in-situ AFM to interrogate the crystal growth of crystalline nanoporous and framework materials." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
